Breast Augmentation
William P. Adams Jr., M.D.
Patrick Mallucci, M.D.
Dallas, Texas; and London, United Kingdom
From the University of Texas Southwestern Medical Center and private practice, and the Royal Free and University College Hospitals.
Received for publication November 23, 2011; accepted April 30, 2012.
Copyright ©2012 by the American Society of Plastic Surgeons
DOI: 10.1097/PRS.0b013e318262f607
Disclosure: Dr. Adams is an investigator for Allergan and Mentor, a member of the Education Advisory Council for Allergan, a royalty recipient for Atlas McGraw-Hill, and chief medical officer of the Plastic Surgery Channel. Dr. Mallucci lectures and teaches on behalf of Allergan, but has no financial or contractual relationship with them.
Learning Objectives: After reading this article, the participant should be able to: 1. Cite the key concepts in the process of breast augmentation that optimize outcomes; 2. Cite key components of tissue-based planning for implant selection; 3. Discuss complications and risks of breast augmentation.
Summary: Breast augmentation remains one of the top surgical procedures performed by plastic surgeons. Current literature supports the concept that breast augmentation outcomes are optimized using a concept of “the process of breast augmentation.” Breast augmentation is often thought of as a surgical procedure; however, the nonsurgical aspects of the procedure are more important for optimizing outcomes and minimizing reoperation and complications. The process of breast augmentation includes patient education, tissue-based preoperative planning, refined surgical technique, and defined postoperative management. This CME article reviews and discusses the current relevant topics and issues surrounding breast implants. There are also supporting videos to enhance the reader’s experience. CME questions are present at the end for a self-assessment. (Plast. Reconstr. Surg. 130: 598e, 2012.)
Breast augmentation is the most popular cosmetic surgery procedure performed worldwide.1,2 This procedure has evolved greatly since the inception of the first implants in the early 1960s and has undergone continued refinement in patient management techniques and implant technology.
The first attempts at augmentation of the breast date back to the late 1800s. Vincenz Czerny, in 1895, used a lipoma from a woman’s back to augment her breast.3 The early 1900s through the 1950s saw the development of injectable substances ranging from glass, rubber, Silastic, paraffin, liquid silicone to almost any material imaginable, with disastrous results—many women ended up with mastectomies to remove impregnated, inflamed, painful beasts.4–8
In 1961, Cronin and Gerow developed the first silicone prosthesis with the Dow Corning Corporation9 that consisted of a teardrop shape with a silicone rubber envelope filled with viscous liquid silicone and a Dacron patch at the back to prevent rotation. These were known as the first-generation implants. Subsequent development over the past 40 to 50 years has led to the development of fifth-generation implants, which are used today (Table 1).
Types of Breast Implants
Implant Filler
The two main types of breast implants in use today are silicone or saline, both with outer silicone elastomer shells. Saline has been used predominantly in the United States because of the silicone controversy of the early 1990s and the moratorium on the use of silicone implants between 1992 and 2006. In Europe and the rest of the world, the vast majority of implants used have been silicone, as they are generally regarded as superior in terms of feel and durability. Changes in the gel fill of
modern implants have led to the development of form-stable gels that are highly cohesive through increased cross-linking of silicone.
modern implants have led to the development of form-stable gels that are highly cohesive through increased cross-linking of silicone.
Table 1. Generations of Silicone Gel-Filled Breast Implants | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Silicone Gel Implant Moratorium in the United States in 1992
In 1992, silicone implants were effectively removed from use other than for reconstructive or revision purposes in a trial setting. The silicone controversy arose as a series of high-profile media cases alluded to complications following breast augmentation with silicone implants. The complications reported were largely alleged systemic autoimmune diseases; however, extensive population studies worldwide since have failed to prove any link between silicone implants and any autoimmune or other disease process. The silicone implants were available for reconstruction and revision/replacement subsequently as multiple U.S. Food and Drug Administration clinical trials were developed. After U.S. Food and Drug Administration panel hearings and multiple other hurdles, the implants were approved again for general use in 2006. Ongoing U.S. Food and Drug Administration core group studies by Allergan Medical and Mentor continue to collect data for a 10-year span,10 and new data are being collected in postapproval studies that were required by the U.S. Food and Drug Administration.
Interestingly, in the recent U.S. Food and Drug Administration silicone breast implant follow-up panel in August of 2011, there were still questions raised by anti-implant groups and individuals citing anecdotal reports of autoimmune diseases caused by breast implants. Despite over 25 high-level scientific studies and over 10 meta-analyses, all of which demonstrate no link between breast implants and any autoimmune disease, there curiously remains a question of this association.11–18 With the degree of science currently available, it would be difficult to conceive of what other evidence could be added to answer this question in the eyes of some individuals.
Nevertheless, the silicone moratorium in the United States prompted the widespread use of saline implants not seen in other parts of the world where silicone had not been banned. Saline has long been seen as second choice to silicone because of problems with deflation and underfilling and overfilling, giving rise to unnatural feel and texture.
Other implant types emerged as alternatives to silicone as a result of the silicone scare. These include Trilucent (soybean oil) implants, marketed as unique because of their “natural” composition and radiographic translucency.19–22 These were withdrawn because of elevated carcinogenic levels in the breakdown product of the soya. Hydrogel implants, also came and went rapidly because of the inability to demonstrate safety data.23
Modern Implant Characteristics
Modern Generation Silicone Implants
The main advances in implant technology have consisted of changes to both the shell and the silicone gel. The features of most modern shells lie in their barrier layer technology and surface texturing options. The barrier layer has reduced significantly the high incidence of silicone bleeding seen in earlier generation implants.24,25 The surface texturing has minimized implant rotation in anatomical implants.
The gel has become progressively more cohesive through more extensive crosslinking of the silicone polymer. Cohesive gels are referred to as form stable (i.e., able to maintain their shape without collapsing under their normal weight or being deformed by the surrounding soft-tissue envelope). There is some evidence that form stability has some long-term advantages in minimizing implant-related complications.28–30
Implant Shell
Essentially, there are two types of implant shell: smooth and textured. Textured devices were originally developed to mimic the surface of polyurethane implants that had a rough porous surface and were known to have very low capsular contracture rates of approximately 1 to 2 percent31; however, level I studies have not supported the lower capsular contracture rates in textured implants.26,32–44 In fact, there are as many level I studies showing no difference as there are that show a benefit, likely indicating that surface texture does not have a role in capsular contracture, especially in the subpectoral pocket plane. The studies
are less conclusive for subglandular pocket plane, with a possible benefit of texture in this position.27,34,39,40 Coming full circle, the contracture benefit seen with polyurethane implants was most likely a biochemical effect not attributable to the surface texture. The current generation implant shells are much more durable, and the introduction of barrier layers has reduced the incidence of gel bleed significantly, a problem so often associated with early generation prostheses.
are less conclusive for subglandular pocket plane, with a possible benefit of texture in this position.27,34,39,40 Coming full circle, the contracture benefit seen with polyurethane implants was most likely a biochemical effect not attributable to the surface texture. The current generation implant shells are much more durable, and the introduction of barrier layers has reduced the incidence of gel bleed significantly, a problem so often associated with early generation prostheses.
Implant Shape
Implants are either round or anatomical. There is a wide variety within these shapes. Most round implants come in different projections for a given volume, allowing for better customization.
Anatomical implants come with even more variability of dimension because of their naturally asymmetric shape. Therefore, width, height, and projection can all be varied to optimally select a “best fit” implant.
There is much debate as to the relative merits of using round over anatomical devices and vice versa. Many surgeons will tend to use one form over the other out of habit and familiarization with a particular product. Our view is that surgeons should be comfortable in moving from one form to the other according to patient desire and the anatomy of the individual. Some situations will dictate a preference for anatomical and others for round.
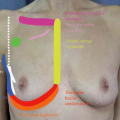
Indications for round implants include the following (Fig. 1):
Fuller appearance.
Very small volumes (shape has less impact at small volumes).
Good basic anatomy and skin quality.
Secondary surgery to avoid rotation.
Athletic individuals because of worry about rotation.
Certain breast/chest wall shapes best suited.
Desiring an overfilled “Baywatch” appearance—best achieved with round implant 50 to 100 cc above optimal fill volume.
Indications for anatomical implants include the following (Fig. 2):
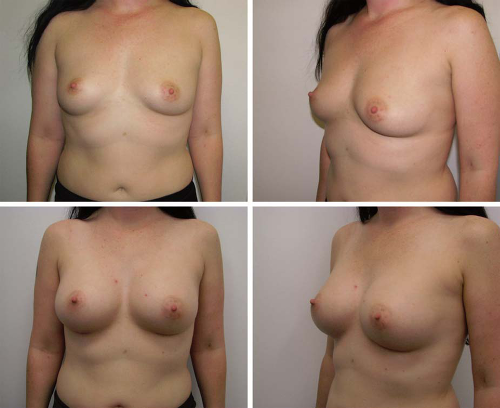
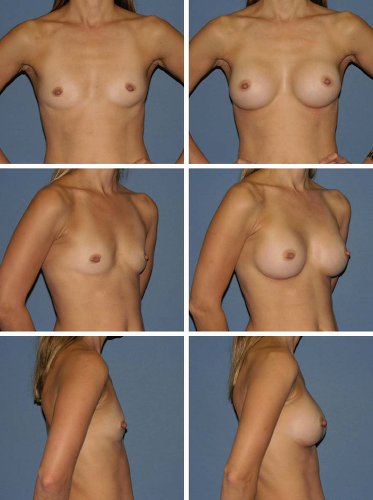 Fig. 2. Preoperative (left) and postoperative (right) views of breast augmentation with anatomical 295-cc implants. |
Patients who want a natural appearance and implant that “fits” their breast.
Constricted lower pole breast (anatomical form-stable implants).
Thoracic hypoplasia.
Breast reconstruction.
Mild ptosis or pseudoptosis, although the rotation risk increases with increasing envelope laxity.
The Process of Breast Augmentation
Breast augmentation is not simply putting an implant into a pocket. The concept of breast augmentation being practiced as a process has revolutionized this procedure.45–47 (See Video, Supplemental Digital Content 1, which introduces the process of breast augmentation, available in the “Related Videos” section of the full-text article on PRSJournal.com or, for Ovid users, at.)
A process is defined as a group of practices that are completed successively to reach a goal. For 45 years, breast augmentation has been thought of as an isolated surgical procedure; however, well-documented elevated reoperation rates of 15 to 24 percent over 2 years in successive premarket approval studies have resulted in a critical analysis of this procedure.10 Factors that impact outcomes have been identified and practice recommendations have been established and refined.48
This analysis has resulted in a redefinition of this procedure to a much broader process beyond the actual surgical placement of the implant. Essential components include comprehensive patient education that enhances informed consent, tissue-based preoperative planning, refined surgical technique and rapid recovery, and a strictly defined postoperative patient management plan. Although each component may exist individually, the combination of these steps in succession has resulted in enhanced outcomes for patients far better than any one component practiced in isolation. In recent years, as key components of this process have been elucidated, it has been demonstrated that the process is transferable and reproducible.45–47 The goals of breast augmentation are as follows:
 Video 1. Supplemental Digital Content 1, which introduces the process of breast augmentation, is available in the “Related Videos” section of the full-text article on PRSJournal.com or, for Ovid users, at http://links.lww.com.easyaccess2.lib.cuhk.edu.hk/PRS/A543. |
To enhance the breast, producing a result that is pleasing and in proportion with the individual.49
To minimize distortion through careful implant selection.
To avoid complications through good technique.
To achieve results with longevity.
Patient Education
The most essential part of breast augmentation is ensuring that patients have a good understanding of the process. This includes an explanation of the principles of implant selection—why a particular type, shape, dimension, or size is used. (See Video, Supplemental Digital Content 2, which discusses patient education and informed consent, available in the “Related Videos” section of the full-text article on PRSJournal.com or, for Ovid users, at http://links.lww.com.easyaccess2.lib.cuhk.edu.hk/PRS/A544.)
It is important for patients to have realistic expectations and to understand the limitations of the procedure and both short- and long-term consequences; this includes knowledge of possible complications and the need for reoperation. Patients will often arrive “well informed” from Internet searches, and it is important for the physician to add context and prioritize this often random information. The goal of the surgeon and the surgical team is to provide the patient with the knowledge to make good decisions about their breast augmentation.
 Video 2. Supplemental Digital Content 2, which discusses patient education and informed consent, is available in the “Related Videos” section of the full-text article on PRSJournal.com or, for Ovid users, at http://links.lww.com.easyaccess2.lib.cuhk.edu.hk/PRS/A544. |
Essentials of Preoperative Assessment
History
This should include the patient’s motives and motivation for surgery. Establish whether goals are realistic. Try to understand what appearance the patient is hoping to achieve—natural versus obvious. This will affect implant choice. It is also important to be cautious about specifying cup size, as this is often inaccurate. Concepts about shape and form are more useful.
A pregnancy history (i.e., if family planning is complete or if in the middle of trying to have a family) should be obtained. In general, the best advice is to wait for the completion of the family before proceeding with breast surgery. It is also advisable to wait for 3 to 6 months following the cessation of breast feeding for breasts to return to normal before embarking on breast augmentation or usually a minimum of 3 months of stable breast size after breast feeding.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree