Recessive dystrophic epidermolysis bullosa (RDEB) is a severe inherited blistering disease caused by mutations in the type VII collagen gene, resulting in defective anchoring fibrils at the epidermal-dermal junction. At present, no curative treatment for RDEB exists. Mounting evidence on reprogramming of bone marrow stem cells into skin has prompted the authors and others to develop novel strategies for treatment of RDEB. The rationale for bone marrow stem cell therapies for RDEB is based on the evidence that bone marrow–derived cells are guided into becoming skin cells, given the right microenvironment. Preclinical studies in mouse models have shown that wild-type bone marrow–derived cells can ameliorate the phenotype of RDEB and improve survival by restoring the expression of type VII collagen and the anchoring fibrils. At present, several clinical studies are ongoing around the world to study the therapeutic effects of bone marrow stem cell transplantation for RDEB. These studies provide a framework for future development of standardized, effective methods for stem cell transplantation to cure severe inherited skin diseases, including RDEB.
The dystrophic forms of epidermolysis bullosa are a group of inherited blistering disorders caused by mutations in the type VII collagen gene (COL7A1). Recessive dystrophic epidermolysis bullosa (RDEB) is characterized by severely reduced or complete lack of type VII collagen protein production, frequently resulting from premature termination codon mutations. Type VII collagen, produced by both keratinocytes and fibroblasts, is the predominant component of anchoring fibrils, which connect the cutaneous basement membrane to the dermis, thereby maintaining the integrity of the epidermal-dermal connection. Severe defects of anchoring fibrils in RDEB result in impaired dermal-epidermal cohesion as well as defective adhesion of mucosal surfaces in the gastrointestinal tract. Clinically, the patients show tense blisters and erosions that heal with extensive and mutilating scarring. Scarring eventually leads to joint contractures and pseudosyndactyly. Repeated lifelong blistering and aberrant tissue repair results in aggressive and metastatic squamous cell carcinoma, one of the causes of early death in these patients.
At present, there is no definitive treatment for RDEB. Clinical management is mainly supportive, aiming at protection of skin from friction, prevention of infections and loss of body fluids, analgesia, and optimal nutritional status. Tremendous progress in understanding the molecular basis of RDEB has provided the basis for development of novel genetic and cellular therapies targeted at restoring the defective anchoring fibrils. The techniques include local or systemic delivery of collagen type VII gene using viral and nonviral vehicles, collagen type VII protein, genetically modified autologous cells, and allogeneic cells from unaffected donors.
This review focuses on the development of bone marrow stem cell therapies for RDEB. The rationale for bone marrow stem cell therapies is based on the capacity of bone marrow–derived cells to differentiate into skin cells given the right microenvironment. The hypothesis is that chronic skin injury in RDEB generates a microenvironment that promotes homing of bone marrow–derived stem cells, which can then differentiate into skin cells, produce type VII collagen, and restore the anchoring fibrils defective in RDEB. Bone marrow cell therapy provides several advantages compared with other genetic and cellular therapies. Transplantation of wild-type allogeneic stem cells obviates the need for exogenous viral gene delivery and the risks associated with it, including insertional mutagenesis and the eventual loss of gene expression. Furthermore, because bone marrow cell therapy can be delivered systemically via bone marrow transplantation, the therapy enables targeting of extensively affected skin as well as internal organs in RDEB. Here we review the data on the capacity of bone marrow–derived cells to become skin cells, the substantial progress both in preclinical and ongoing clinical studies, and the future directions in developing bone marrow stem cell therapies as a potential cure for RDEB.
Bone marrow stem cell reprogramming into skin cells
Pluripotent embryonic stem cells are capable of differentiating into all cell types of the body. By contrast, adult stem cells are capable of differentiating into some or all major cell types of the tissue or organ in which they reside. Bone marrow is the major source of adult hematopoietic stem cells (HSCs), which are capable of reconstituting the entire circulating population of hematopoietic cells. The bone marrow also contains mesenchymal stem cells (MSCs), which contribute to the regeneration of mesenchymal tissues, including bone, cartilage, muscle, and adipose tissues. In addition, bone marrow stem cells appear to have the capacity to differentiate into other cell types such as endoderm (eg, liver) and ectoderm (eg, epidermis). This property has been attributed to bone marrow stem cell reprogramming or heterogeneity of stem cell types within bone marrow, although debate existed initially on the contribution of cell fusion and technical limitations to these observations. Of importance is that the property of bone marrow cells differentiating into other cell types is thought to be relatively rare, guided by signals from a given microenvironment and promoted by selective advantage, as in the case of tissue injury.
Bone Marrow–Derived Stem Cells Exhibit Homing to the Skin
Bone marrow–derived stem cells have the capacity to migrate to the skin and become skin cells ( Table 1 ). The studies are largely retrospective data on transplantation of genetically marked or sex-mismatched donor bone marrow into wild-type recipients. Engraftment and differentiation of donor-derived cells has been shown by detection of donor-derived DNA, mRNA, or protein. Substantial variability in study design exists, most importantly in methods used for isolation of donor cells and characterization of engrafted donor cells. In some studies the transplant has been enriched for HSCs or MSCs, whereas others have used total bone marrow. Furthermore, the studies have focused on detection of either bone marrow–derived keratinocytes or fibroblasts, or both (see Table 1 ).
| References | Model | Isolation of Cells | Wound | Results |
|---|---|---|---|---|
| Krause et al | Mice; BMT; Sex-mismatched; Transplantation of a single cell after serial transplantation | Lineage depletion, elutriation, bone marrow homing, separation of small G0/G1 cells | No | Bone marrow–derived cells in variable epithelial tissues including the skin; 1%–3% cytokeratin + Y + cells localizing to the bulge and epidermis |
| Korbling et al | Human; PB HSC transplantation to treat malignancy (leukemia/lymphoma etc); Retrospective | CD34 + cells | No | Transplant-derived cells in various organs; Cytokeratin + XY + CD45 − epithelial cells in the skin in stratum spinosum, granulosum and close to dermal-epidermal junction |
| Hematti et al | Human; PB HSC transplantation to treat hematologic malignancy; Sex-mismatched | CD34 + cells | No | No donor –derived DNA in keratinocyte cultures analyzed by PCR for Y chromosome markers |
| Badiavas & Falanga | Mice; BMT; Donor GFP transgenic mice | Unfractionated | Full-thickness wound with/without G-CSF pre-treatment | No wound: Scattered GFP + cells in dermis Wound ± G-CSF: GFP + inflammatory cells on day 2, GFP + keratin + cells in the hair follicle, sebaceous glands, epidermis on day 21 |
| Badiavas et al | Human; Transplantation of autologous bone marrow cells to chronic wounds | Unfractionated | Chronic wound | Wound closure and dermal rebuilding by inspection and histology |
| Kataoka et al | Mice; Transplantation of a mixture of GFP + bone marrow cells and embryonic dermal and epidermal cells into a full-thickness skin defect | Unfractionated | Wound | Bone marrow cells only: Wound not epithelialized Bone marrow cells with embryonic dermal and epidermal cells: Wound epithelialized, GFP + cells in hair follicles (K6 + ), sebaceous glands (Oil red O + ), epidermis (K1 + ), dermis |
| Borue et al | Mice; BMT; Sex-mismatched | Unfractionated | Wound | Increase from <1% to 8% in cytokeratin + XY + epithelial cell number; No evidence of fusion |
| Fathke et al | Mice; BMT; Donor GFP transgenic mice | Unfractionated | No/wound | Bone marrow–derived cells contribute to the skin and express both collagen types I and III |
| Rovo & Gratwohl | Human; PB Allogenic HSC transplantation | Not specified | No | PCR of microsatellite markers revealed no evidence of donor DNA in plucked hairs |
| Fan et al | Mice; BMT; Donor LacZ or GFP transgenic mice | Unfractionated | No/wound | No wound: Bone marrow–derived keratinocytes could not be detected in the epidermis Wound: epidermis contained rare bone marrow–derived keratinocytes |
| Inokuma et al | Mice; BMT; Donor GFP transgenic mice | Unfractionated | Full-thickness wound ± CCL27 intradermal injection | GFP + keratin14 + CD11c − CD45 − cells in basal epidermis and around hair follicle bulge; Intradermal injection of CCL27 chemokine enhanced bone marrow –derived keratinocyte migration and accelerated wound healing; Neutralizing antibody inhibited migration |
| Sasaki et al | Mice; MSC transplantation; Donor GFP transgenic mice | Cultured adherent cells from bone marrow; CD29 + , CD44 + , CD90 + , CD34 + , CD31 + ; adipogenic/osteogenic/chondrogenic potential | Full-thickness wound ± CCL21 intradermal injection | GFP + pancytokeratin + cell 0.14%, GFP + CD31 + cells 4.7%, GFP + SMA + cells 0.2%, GFP + CD11b + cells 1.5%; Intradermal injection of chemokine CCL21 increased GFP + cell migration and accelerated wound repair |
In their pioneering work, Krause and colleagues demonstrated engraftment of bone marrow–derived stem cells to multiple organs, including the skin, after sex-mismatched transplants of a single bone marrow–derived stem cell harvested after serial transplantation. To isolate the HSC population, the donor cells were enriched through multiple sophisticated steps including lineage depletion, elutriation (a process involving gas or liquid that separates particles based on their size), bone marrow homing, and separation of G0/G1 cells. The frequency of donor-derived XY-positive cells with characteristics of keratinocytes, determined by expression of keratins and lack of expression of lymphocyte and macrophage markers, was 1% to 3%. In a later study, the investigators further demonstrated that the event was not due to cell fusion. Instead, the finding was argued to result from either reprogramming or the presence of an uncharacterized epithelial or multipotent bone marrow stem cell population.
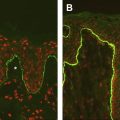
The concept of engraftment of bone marrow–derived cells to the skin was further proven in a small retrospective clinical study using skin from bone marrow transplant recipients. The study included 11 patients who had received high-dose chemotherapy either alone or in combination with radiotherapy, followed by transplantation of allogeneic peripheral blood stem cells (PBSCs) in 10 patients or autologous PBSCs in 1 patient. Long-term engraftment and hematopoietic chimerism after transplantation was demonstrated by microsatellite marker analysis. Skin, liver, and gastrointestinal tract biopsies were studied for the presence of Y-chromosome–containing cells in the tissues of female recipients. Donor-derived keratin-positive cells were found in the epidermis of the skin as far as 3 years after PBSC transplantation, demonstrating that bone marrow cells can be reprogrammed into skin cells in human.
Keratin-positive bone marrow–derived donor cells are often found scattered in the skin, including the epidermis, hair follicles, and sebaceous glands, as keratin-positive cells or adjacent cells. Of note, each of these locations harbor a specified skin stem cell niche for the different epithelial lineages, specifically epidermis, hair follicle, and sebaceous gland. Whereas some studies do not report donor-derived cells in the vicinity of the hair follicles, others show preferential localization of the cells to the hair follicle bulge, one of the specialized skin stem cell niches. Tamai transplanted newborn mouse skin with developing hair follicles onto the back of a mouse with prior green fluorescent protein (GFP)-positive bone marrow transplantation. A significant number of GFP-positive bone marrow–derived cells engrafted into the hair follicle bulge region and the epidermis, supporting the hypothesis that ectopic cells home to one of the skin stem cell niches.
Role of the Microenvironment in Promoting Homing, Engraftment, and Differentiation of Bone Marrow–Derived Stem Cells into Skin Cells
The microenvironment has a critical role in regulating the differentiation potential of stem cells. Stem cell microenvironments, or niches, are specific anatomic locations that regulate the function of stem cells during tissue generation, maintenance, and repair. A variety of factors contribute to the niche composition, including anatomic structures, cellular interactions, extracellular matrix proteins, products of cellular metabolism, and secreted factors. The niche provides the physical interaction, and the inhibitory and stimulatory signals required to maintain stem cell numbers, and to modulate their response to changes in physiologic conditions, such as in wound repair. A commonly used example of modification of the microenvironment is conditioning for bone marrow transplantation, which creates “space” for the donor stem cells by freeing the stem cell niches.
In Drosophila , an empty stem cell niche was shown to reactivate the proliferation of ectopic cells. The investigators postulated that after loss of their normal stem cells, the stem cell niches will likely maintain the potential to stimulate the proliferation of foreign stem cells or other cell types for a defined period. Related findings were reported by Booth and colleagues, who demonstrated that neural stem cells and their progeny enter mammary epithelium–specific niches and adopt the function of mammary cells. Intriguingly, Lako and colleagues reported that hair follicle dermal cells have the capacity to repopulate the mouse hematopoietic system in a lethally irradiated animal, demonstrating an unexpected differentiation potential of these cells guided by the signals of an empty, irradiated hematopoietic stem cell niche. Together, these studies support the concept that tissue-specific signals originating from the stroma and the differentiated somatic cells of a given niche can redirect ectopic stem cells to produce cellular progeny committed to cell fates of the tissue in which the niche resides. The authors hypothesize that the skin microenvironment in RDEB, characterized by severe recurrent blistering and erosions, may function in a related fashion by providing a depleted skin stem cell niche for ectopic stem cells, thereby improving their engraftment and proliferation.
In addition to the role of bone marrow–derived inflammatory cells in the initial phases of wound repair, other bone marrow–derived cells are known to participate in the homeostasis and regeneration of skin during wound healing. With few exceptions, most studies show that wounded skin stimulates engraftment of bone marrow–derived cells to the skin and induces them to incorporate and differentiate into nonhematopoietic skin structures. In comparison with skin at homeostasis, wounding increases the percentage of bone marrow–derived cells with keratinocyte characteristics by more than twofold.
One of the studies showing the differentiation potential of bone marrow–derived cells into the components of the skin and the role of the microenvironment during wound repair was performed by Kataoka and colleagues, who mixed fresh, unfractionated bone marrow from a GFP-transgenic mouse with embryonic epidermal and dermal cells and transplanted this mixture onto skin defects of nude mice. GFP-positive cells costained with keratinocyte, sebaceous gland, and endothelial cell markers and respective cell types were detected in the suprabasal layers of the epidermis, hair follicles, sebaceous glands, and in the dermis associated with blood vessels. Bone marrow cells alone were unable to reconstitute the skin defect, emphasizing the importance of the interactions in the microenvironment necessary for bone marrow cell engraftment. Different populations of bone marrow cells were not tested separately, but these investigators proposed that the unfractionated marrow may contain a common progenitor population producing endodermal, mesodermal, and ectodermal progeny.
In humans, topical application of bone marrow cells to chronic wounds similarly led to closure of the wounds. Autologous bone marrow cells were applied directly to chronic wounds in 3 patients. These patients had not previously responded to standard or advanced therapies, including bioengineered skin application and grafting with autologous skin. Complete closure and evidence of dermal rebuilding was observed in all 3 patients. Findings suggestive of engraftment of applied cells were observed in biopsy specimens of treated wounds. Clinical and histologic evidence of reduced scarring was also observed. These studies suggest that bone marrow–derived cells can lead to dermal rebuilding and closure of nonhealing chronic wounds.
Woodley and colleagues further demonstrated the role of the wounded microenvironment in promoting homing of transplanted cells and the use of fibroblasts as a vehicle to deliver type VII collagen to the wounded skin. The investigators used murine skin defects or grafted human skin as a model system, and showed that systemically delivered fibroblasts, derived from an RDEB patient and engineered to overexpress type VII collagen, have the capacity to home to skin wounds, deliver type VII collagen to the wound, and promote wound healing.
To search for factors contributing to homing of bone marrow–derived cells to the skin, Inokuma and colleagues hypothesized that recruitment of bone marrow–derived cells to the skin is regulated by chemokine-chemokine receptor interactions. CCL27 (chemokine, C-C motif, ligand 27) is a small cytokine belonging to the CC chemokine family, and its receptor is CCR10 (chemokine, C-C motif, receptor 10). CCL27 is predominantly expressed by epidermal keratinocytes, and is associated with infiltration of circulating cells in immunologic disease. Capitalizing on this property, Inokuma and colleagues showed that it can also increase recruitment of bone marrow–derived cells to the epidermis after introduction into the wound bed and accelerate wound healing. Sasaki and colleagues similarly injected CCL21 (chemokine, C-C motif, ligand 21) intradermally to mice transplanted with GFP-positive MSCs. CCL21 increased migration of MSCs to the skin and accelerated wound healing. These findings suggest that specific chemokines such as CCL27 and CCL21 may facilitate chemoattraction of circulating stem cells into the skin for therapeutic purposes.
Correction of the RDEB phenotype in mice by bone marrow transplantation
Stemming from the data on reprogramming of marrow cells into skin cells, several groups have conducted bone marrow transplantation to treat RDEB in an animal model of RDEB ( Table 2 ). Chino and colleagues investigated whether bone marrow–derived fibroblasts can ameliorate RDEB phenotype in a mouse model using embryonic bone marrow cell transplantation. These investigators performed embryonic bone marrow cell transplantation into RDEB mice with T-cell–depleted GFP-transgenic bone marrow cells via the vitelline vein, which is connected to the fetal circulation. The study showed that type VII collagen was detected in the basement membrane zone in the vicinity of the GFP-positive bone marrow–derived fibroblasts positive for fibroblast markers fibronectin, vimentin, type I collagen, and type VII collagen. Embryonic bone marrow cell transplantation also significantly ameliorated the lethal pathology in the neonate mice, with average survival of 15 to 20 days in the treated group and 2 days in the nontreated group.