Basic Science and Management of Postsurgical Lymphedema and Vascularized Lymph Node Transplant
Joseph H. Dayan
History
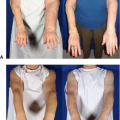
Lymphedema continues to be a common and debilitating disease affecting millions of women with breast cancer worldwide. Managing lymphedema can be onerous with lifelong compression, massage, and physiotherapy. Despite 100% compliance, lymphedema often progresses. Many patients experience a delay in referral because mild swelling may not appear very impressive on physical examination. However, these patients often experience poor quality of life because they have an incurable disease, avoid many activities they enjoy, and are committed to hours of daily care. Additionally, approximately one-third of patients develop cellulitis which can spread rapidly and lead to sepsis (1).
While there are clear guidelines for breast cancer screening, surveillance for lymphedema in high-risk patients (axillary dissection and radiotherapy) is not yet standard practice. It is also not uncommon for mild limb swelling to be observed as opposed to immediate referral for lymphedema therapy. Waiting for a 10% or greater difference in limb volume is akin to waiting for a heart attack to diagnose coronary artery disease. Lymphatic dysfunction starts before visible swelling. Symptoms of pain, heaviness, and transient swelling are best referred early to a certified lymphedema therapist.
Fortunately, there has recently been significant interest in lymphedema, improved understanding of the disease, and promising treatments. Surgery has become a viable option not only for appropriately selected patients with lymphedema but also for those patients undergoing axillary dissection who are at risk. Effective medical therapies for lymphedema may soon become a reality. This chapter does not cover the entirety of this subject, but aims to provide a modern and practical understanding of lymphedema, and describe the current role of lymphatic surgery. Undoubtedly, the indications and surgical options for this disease will evolve significantly in years to come.
Pathophysiology of Lymphedema
A common misconception about lymphedema is that it is purely a plumbing issue: removal of axillary lymph nodes causes a mechanical obstruction that leads to backup of lymph and swelling of the upper limb. While there is certainly an initial mechanical obstruction, not all patients develop lymphedema immediately after axillary dissection. Most develop lymphedema months or years later, so there is something else that happens in addition to the surgical injury. Another peculiarity of lymphedema is that over 80% of patients develop some degree of fibrofatty accumulation, and one-third of patients have more fat than fluid in their limb (2). These bizarre manifestations of this disease are a result of the immune response to the lymphatic injury which can result in this chronic and progressive condition. One current hypothesis proposed is that the initial lymphadenectomy leads to lymph stasis, creating an inflammatory environment which then leads to scarring of the lymphatics, compounding the lymph stasis which continues to amplify the inflammatory process (3). Authors have demonstrated that a lymphatic injury–induced inflammatory cascade leads to increased levels of IL-4 and IL-13, in turn leading to lymphatic vessel injury and lymphedema (4). Administering antibodies specific to cytokines which promote T helper 2 cell differentiation associated with IL-4 and IL-13 production effectively reversed lymphedema in a mouse model (4). It still remains a mystery why some patients have a fluid-dominant limb while others have a fat-dominant limb, some have intractable cellulitis and others have never had a single episode. These different patterns of lymphedema are not on the same continuous spectrum. Medical therapies have aimed to target the inflammatory cascade and block it, or to stimulate lymphangiogenesis (5,6,7).
Diagnosis of Lymphedema
Lymphedema is most commonly diagnosed by clinical examination. Upper limb swelling is typically first evaluated with duplex ultrasound to rule out deep vein thrombosis, and if negative, the patient is referred for lymphedema therapy. Historically, a 10% difference in limb volume was diagnostic for lymphedema. However, significant swelling is a late finding of lymphatic compromise. Patient symptoms of heaviness, transient swelling, and pain typically precede swelling and should be referred for lymphedema therapy. There are technologies that may improve early detection of lymphedema. Bioimpedance spectroscopy (BIS) is a noninvasive test that can detect abnormally high extracellular volume in a limb, consistent with lymphedema. Imaging of the lymphatic system is traditionally performed with lymphoscintigraphy. While not particularly sensitive, a finding of dermal backflow in the limb is diagnostic (1).
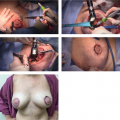
More recently and currently off-label but commonly used among lymphatic surgeons, is indocyanine green (ICG) lymphography (8). This involves subdermal injection of ICG which is taken up by the lymphatic vessels and using a near-infrared camera, a high-resolution image of the lymphatic system itself is seen. Abnormalities of the lymphatic vessels are directly and clearly visualized. A complete absence of lymphatic vessels of the lymphatic system is seen in Figure 25-1. Imaging studies are best used to confirm a diagnosis in cases which are unclear or in preparation for lymphatic surgery. In summary, patient-reported symptoms often present much earlier than visible swelling and should prompt referral for lymphedema therapy. Screening with symptoms and/or BIS at the patient’s postop oncologic follow-up appointment every 6 months is a reasonable starting point for lymphedema surveillance in high-risk patients.
Indications for Lymphatic Surgery
Our understanding of which lymphatic procedure is appropriate for which patient continues to evolve. Currently, the three most common procedures are lymphovenous bypass (LVB), lymph node transplant, and liposuction (9,10,11,12,13). The first two aim to provide an exit for lymph, while liposuction is reserved for the more advanced cases of lymphedema which are fat dominant. These procedures can also be combined in patients who have a significant degree of both fluid and fat accumulation.
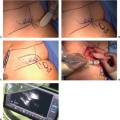
LVB is a minimally invasive outpatient procedure where an obstructed lymphatic vessel is divided and anastomosed to a neighboring vein through one or several small incisions (Fig. 25-2). These vessels are typically 0.3 to 0.8 mm
in diameter making LVB technically challenging. In general, distal LVB is most effective in patients with early lymphedema evidenced by presence of linear lymphatics on ICG. An LVB performed on a heavily scarred lymphatic vessel is not going to be as effective. More recently, immediate lymphatic reconstruction with LVB in the axilla at the time of lymphadenectomy is being performed in the hopes of reducing the risk of lymphedema (Fig. 25-3) (14). Data in this field is still preliminary and also under study protocol at our institution.
in diameter making LVB technically challenging. In general, distal LVB is most effective in patients with early lymphedema evidenced by presence of linear lymphatics on ICG. An LVB performed on a heavily scarred lymphatic vessel is not going to be as effective. More recently, immediate lymphatic reconstruction with LVB in the axilla at the time of lymphadenectomy is being performed in the hopes of reducing the risk of lymphedema (Fig. 25-3) (14). Data in this field is still preliminary and also under study protocol at our institution.
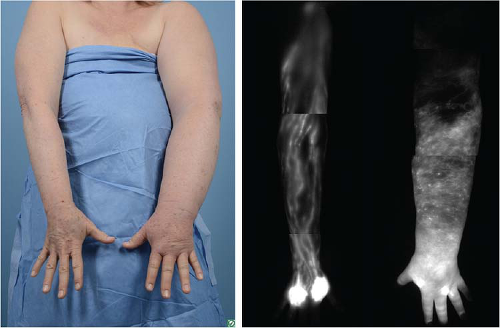 FIGURE 25-1 Clinical photo compared to ICG lymphangiography demonstrating advanced lymphedema with no lymphatic vessels visualized. Not an ideal candidate for lymphovenous bypass. |
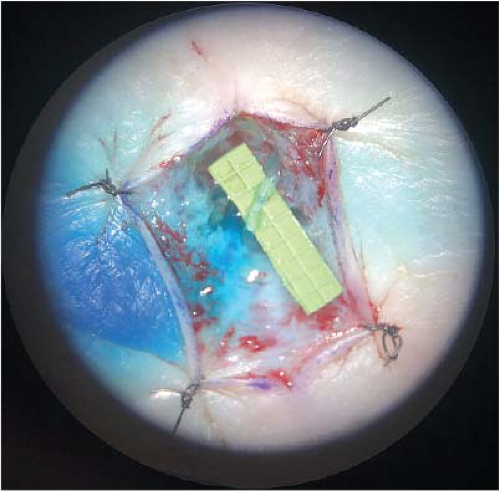 FIGURE 25-2 Distal lymphovenous bypass demonstrating patent anastomosis with clear lymph flowing into the vein. |
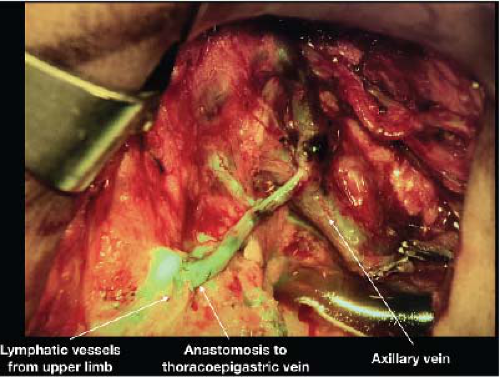 FIGURE 25-3 Immediate lymphatic reconstruction at the time of axillary dissection with lymphovenous bypass of several lymphatic vessels telescoped into the thoracoepigastric vein. |
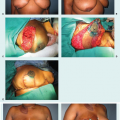
In contrast, a vascularized lymph node transplant (VLNT) only requires an arterial and venous anastomosis and does not depend on the state of the lymphatic vessels. This allows one to perform a viable VLNT in someone with not only mild but also moderate lymphedema. However, there is no data to guide us regarding which patients are most likely to respond to this procedure. VLNT depends on the lymphangiogenic growth factors within the transplanted nodes which have been shown to induce ingrowth of lymphatic vessels (15). The proposed mechanism includes shunting of lymph into the venous system through interconnections between the lymphatic sinuses and venules within the lymph node (16).
A variety of donor sites have been described including groin, axillary, supraclavicular, and intra-abdominal lymph nodes (17,18,19,20). One of the concerns regarding this procedure, particularly for groin and axillary-based lymph node transplant is the risk of donor-site lymphedema (21,22). Dayan et al. first described reverse lymphatic mapping for VLNT to significantly reduce these risks (17,23). Currently, we favor the omentum based on the right gastroepiploic vessels because it eliminates any risk of donor-site lymphedema and also provides abundant lymphatic tissue with consistent anatomy. In our practice, lymph node transplant to the axilla is indicated in patients who already have secondary issues such as axillary contracture, pain, or axillary vein stenosis. If there is profound swelling in the hand and forearm and there is no migration of ICG above the elbow, then a distal lymph node transplant or two lymph nodes transplants (one placed in the wrist or forearm and the other placed in the axilla) are offered (19). Our group will also perform VLNT at the time of axillary dissection in select cases where the axilla is expected to be radiated in addition to the chest wall. This is done not only to reconstruct the lymphatic system but also replace the soft tissue to mitigate potentially significant contracture and pain.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree