Summary and Key Features
- •
Autologous cosmeceuticals that use the patient’s biologics offer a paradigm shift for skincare.
- •
These biologics such as adipose tissue, fibroblast cells, and platelet-rich plasma offer a unique cocktail of biomolecules for autologous skin rejuvenation.
- •
Autologous cosmeceuticals have shown to diminish the appearance of fine lines and wrinkles and acne scars.
- •
They offer a new method of post-care treatment after ablative laser or other damage to the skin.
- •
The Food and Drug Administration regulates human cells, tissues, and cellular and tissue-based products (HCT/Ps).
Introduction
The concept of autologous cosmeceuticals began in the latter part of the 20th century. The 1990s ushered in the genesis of tissue engineering. Early pioneer companies such as Advanced Tissue Sciences, Inc. and Organogenesis, Inc. had developed the process to manufacture full three-dimensional skin constructs. These products were used to treat diabetic foot ulcers and burns, as well as serving as alternatives to animal testing. The initial source for manufacturing these products was neonatal foreskin cells. What scientists quickly learned was that the cell culture medium that the foreskin cells were growing had a plethora of growth factors, amino acids, and peptides.
These cells, whether botanical, embryonic fibroblasts, cord blood, various tissue stem cells (e.g., fat, bone, embryonic), or American Type Culture Collection (ATCC) cultured stem cells, have served as a source for multiple advertised growth factors. The allogenic sources can be fraught with obvious pathogen-transmissible concerns, but those concerns have largely been successfully managed by the industry. Still, sources for these cells and bioactive molecules remain controversial. Reproducibility, potency, and culturing reliability to ensure that each production batch has the same concentration, functional performance of growth factors, and so on still represent a challenge today regarding commercial release criteria for these ingredients.
Although these products have brought significant advances in skin care including in aesthetic medicine, they do not entirely represent the personalized, customized products that consumers are requesting. Fully autologous products represent the most personalized skin care products for the foreseeable future.
This chapter provides a review of autologous-derived therapeutic approaches for facial regeneration, specifically addressing adipose tissue, fibroblasts, and platelet-rich plasma (PRP).
Adipose Tissue Source
Adipose-derived stem cells (ADSCs) have become one of the most versatile adult stem cells due to their abundance and relative accessibility. Paracrine cytokines, exosomes, and other bioactive molecules play a critical role in ADSCs’ ability to exert their biologic effects. ADSCs are derived from white fat, the heterologous collection of cells within adipose tissue, the stromal vascular fraction (SVF), which consists of mesenchymal pluripotent cells that can differentiate into adipocytes, neural cells, cardiomyocytes, and hepatocytes.
The paracrine factors—epidermal growth factor (EGF), basic fibroblastic growth factor (bFGF), transforming growth factor beta (TGF-β), vascular endothelial growth factor (VEGF), and others—signal fibroblasts to produce new extracellular matrix (ECM), including collagen and elastin, resulting in sustained clinical improvement in aging skin over weeks.
Preparation
The tissue extraction/preparation procedure remains critical for all the current autologous products in routine use to ensure reproducibility and reliability of yield. Poor isolation and separation of adipose, fibroblast, and PRP tissue results in poor clinical aesthetic efficacy and, at times, discomfort to the patient.
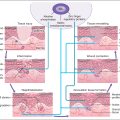
Among the autologous therapeutic products, harvesting of autologous fat is the most challenging, requiring some surgical expertise. However, subcutaneous fat tissue contains 500 times more stem cells than bone marrow does. Depending on the anatomical site of the body, there may be varying amounts of subcutaneous adipose tissue and therefore different amounts of ADSCs. A schematic diagram showing the preparation of adipose-derived stem cells is depicted in Fig. 15.1 .
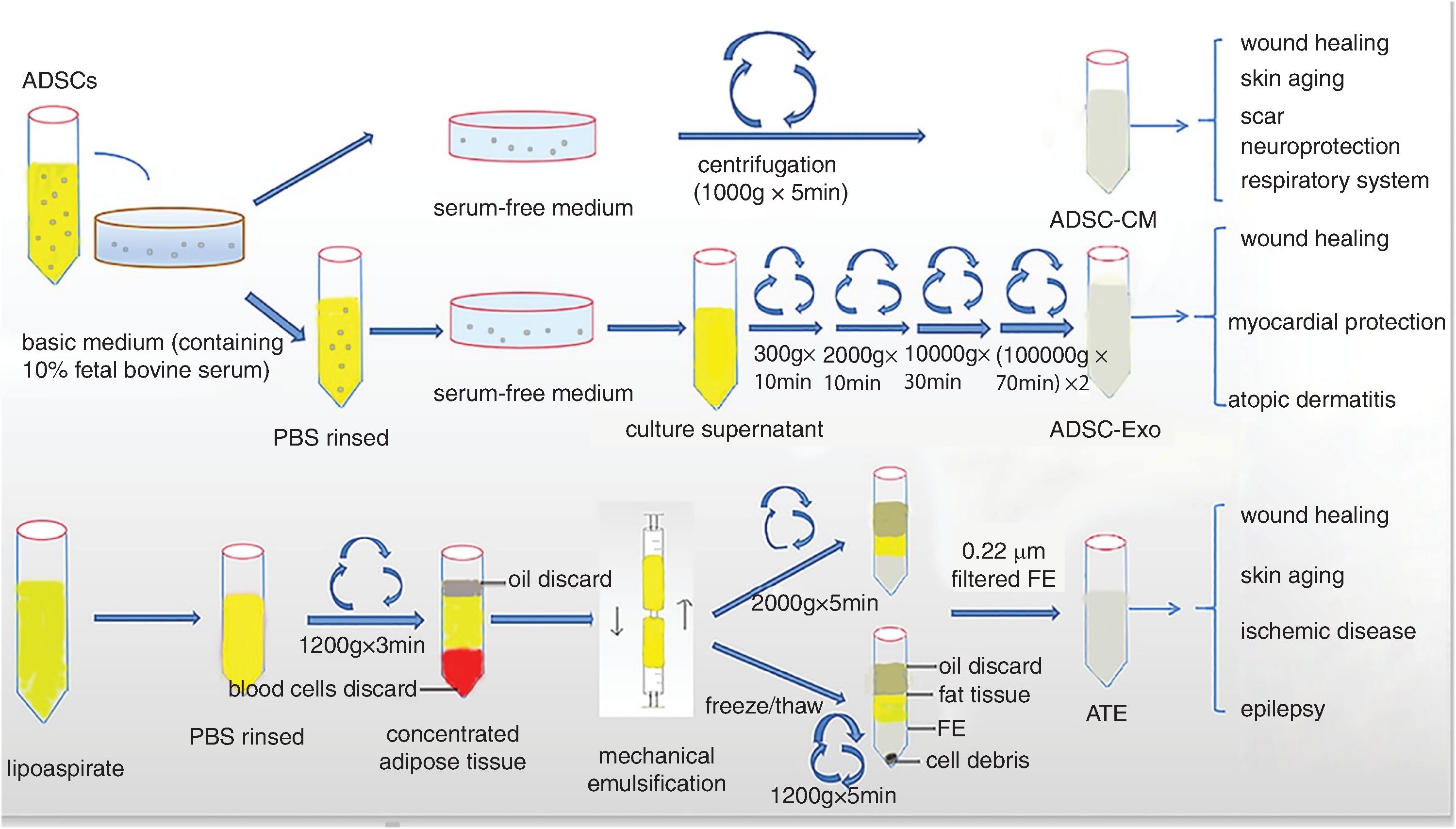
Successful harvesting of fat requires that the provider has an approach that is consistent, reliable, and reproducible. The wrong-size cannula, incorrect hole sizes, too much anesthetic injection, or suction that is too strong can present mechanical shearing forces and other ill effects that in turn produce poor lots of functional fatty tissue and ADSC. The most popular collection technique involves gravity separation centrifugation, which requires no special equipment. Collection of ADSCs requires micronization of the fat particles. Several commercial centrifuge companies have specific swing-out rotors that lead to well-separated viable SVF after two centrifugation cycles. The SVF fraction contains preadipocytes (ADSCs), immune cells (e.g., those that help maintain local immune homeostasis), and ECM proteins.
Use Cases
ADSCs are multipotent in function, but generally they do not proliferate in vivo and instead act as regulatory cells. They have high proliferation ability and can promote angiogenesis in the dermis.
Using harvested fat in the face requires the proper location and depth of the various fat compartments on the face, including the superficial fat space, superficial musculoaponeurotic system, and deep fat space. It also requires considerable experience to effectively inject the fat into the accurate anatomical spaces. Facial areas amendable to autologous fat filling include any hollowing areas, malar bags, and nasolabial folds (NLFs).
Studies from Surowiecka and Struzyna (2022) reported that ADSCs can mitigate the effects of ultraviolet (UV) light photoaging. ADSCs secrete many antioxidants, including cytokines that reduce the effects of UVB irradiation and resultant reactive oxygen species (ROS). ROS triggers multiple damaging cellular pathways, general inflammation, damage to the cell membrane, and alterations in DNA and RNA proteins, including the ECM. Secretion of various adipose bioactive factors, including hepatocyte growth factor, protects the cells from the UV-induced oxidative stress. UV-associated hyperpigmentation triggers melanogenesis and dilated vessels due to increase dermal angiogenesis. ADSCs secrete TGF-β1, which suppresses the tyrosinase enzyme, mitigating UV irradiation-induced hyperpigmentation.
Although there are multiple reports using adipose tissue as injectable filler, there is a paucity of publications on any topical approach for ADSCs. Ohashi (2020) demonstrated that following ablative laser treatment, ADSCs coupled with niacinamide could readily penetrate the skin, bringing improvement in fine lines, wrinkles, roughness, laxity, and increased barrier function.
Complications
Clearly, fat tissue including ADSC has a future in aesthetic medicine. However, there are several potential side effects that the provider needs to be aware of. Bruising, edema, and mild-to-moderate pain can occur with the harvesting procedure. Fat embolism, albeit rare, has been reported in cases, and studies have reported patient deaths of thromboembolism. Importantly, ADSCs do not proliferate in vivo or undergo any neoplastic changes. They do promote neoangiogenesis, and therefore a small risk of neoplastic vascular network does exist. With all autologous cosmeceutical products, it is important for the provider to do a complete medical history and examination prior to beginning an autologous aesthetic procedure.
In summary, ADSCs are mesenchymal pluripotent cells that can differentiate into various cell lineages, including adipocytes and their role in skin regeneration. ADSCs are relatively easy to extract and provide a minimally invasive collection method versus bone marrow–derived stem cells. There is no risk of immune rejection, unlike with embryonic stem cells. Further studies are needed to better understand the molecular factors including homeostatic regulation of bioactive molecules to ensure optimal successful use of ADSCs for aesthetic uses. Improvements in extraction techniques, including new methods such as microporing, will be critical for continuing widespread routine aesthetic use in physician practices.
Fibroblast Source
Part of the complexity of the skin aging process results in a decline in the number of fibroblasts and associated extracellular matrix. Increasing senescence leads to a decrease in tone and elasticity, leading to visible fine lines, wrinkles, and sallowness.
Historically, various implants of silicone, bovine collagen, polymethyl acrylate, polylactic acid, and others have in some patients resulted in allergic dermatitis, nodules, abscesses, and fulminant necrosis. This increases the desire to find other implantable materials with an excellent tolerability profile.
During the past 10 years, there has been increasing use of autologous fibroblasts for treatment of scars, wrinkles, and subcutaneous atrophy. The fibroblasts are cultivated in human serum (bovine serum is no longer used). Autologous fibroblasts are the first and only cell therapy approved by the FDA for aesthetic use from the patient’s own biopsy tissue.
The extraction procedure for fibroblasts typically involves a 4-mm punch biopsy in the periauricular region of the face using local anesthetic. The specimen is sent to a commercial laboratory or Clinical Laboratory Improvement Amendments (CLIA) laboratory for cell expansion and storage of the fibroblast culture. A sample of the patient’s cells undergoes strict mandatory safety testing to exclude any potential carcinogenic, viral, fungal, bacterial, or prion contaminants.
The thawed cultured cells can be injected into the hands, NLFs, scars, stretch marks, wrinkles, and periorbital and perioral areas of the face. An identifiable positive effect is noticed after the first treatment, with improvement continuing over several months.
The exact mechanism of action of injectable autologous fibroblasts remains unknown. Studies have shown that the fibroblasts may operate through direct synthesis of increased amounts of collagen and elastin, inducing proliferation of native fibroblast population or stimulating various growth factors and cofactors to increase the population of fibroblasts.
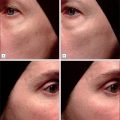
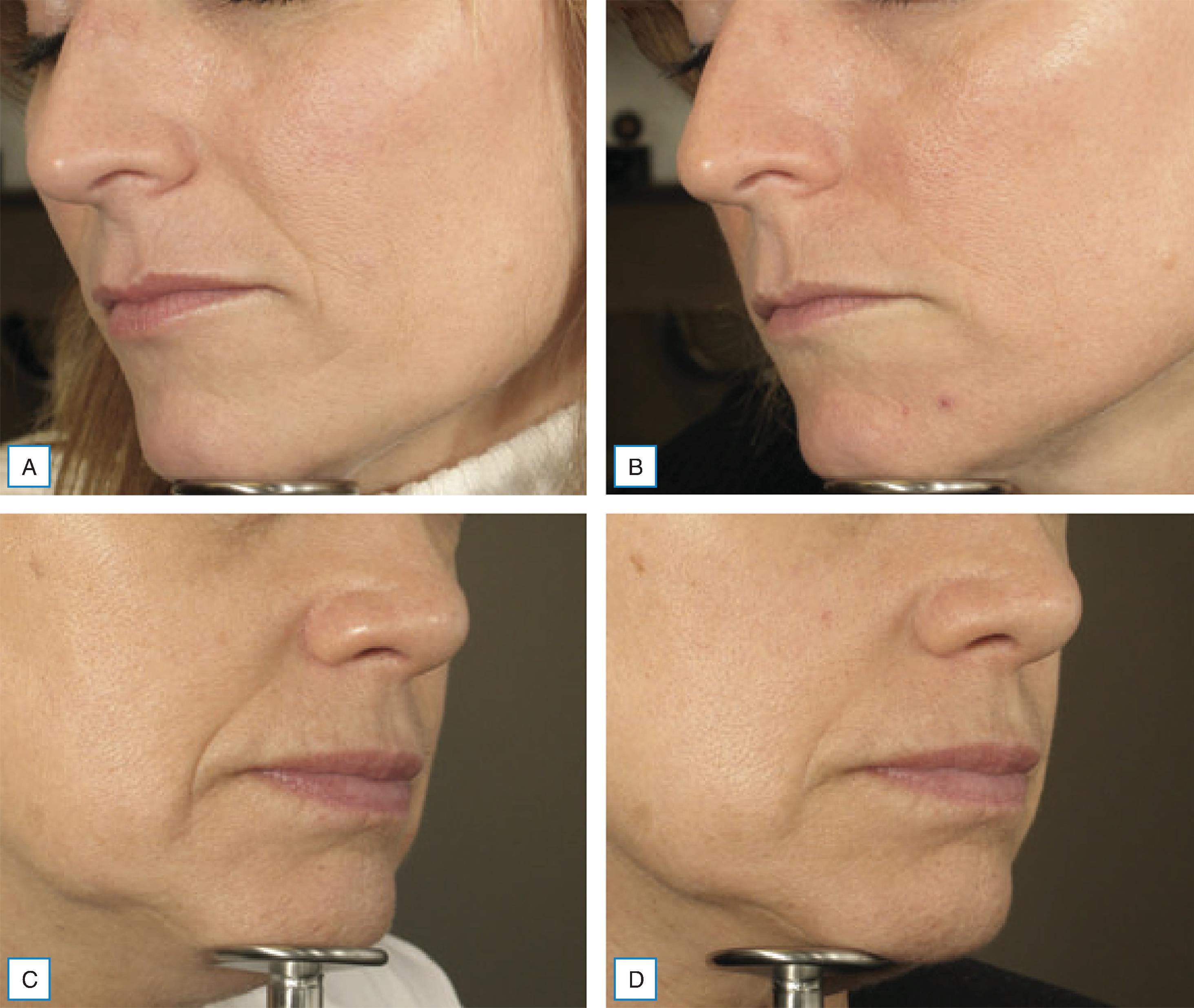
A study by Smith et al. (2012) demonstrated that autologous fibroblast cell therapy was effective at mitigating NLFs ( Fig. 15.2 ). Improvement in NLFs was noted 2 months following the first injection series. Continuous improvement was noted up to 6 months after a series of three injections. The autologous cell filler did not degrade over 6 months. Prior studies showed improvement in NLFs for up to 1 year or more.
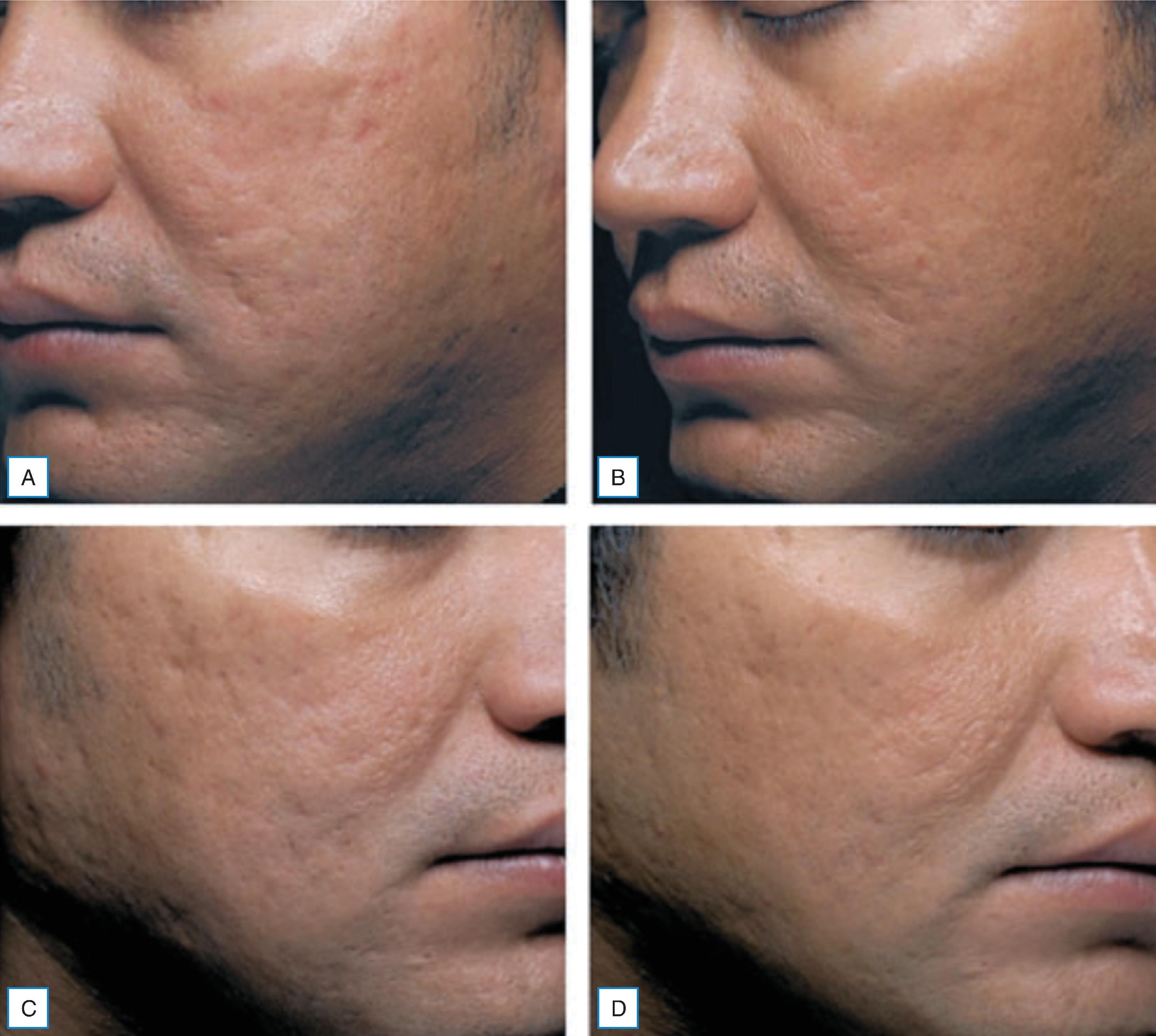
Given the diversity of acne scarring morphology and severity, certain treatment modalities are more effective than others. demonstrated the efficacy of autologous fibroblasts on dispensable acne scars ( Fig. 15.3 ). The fibroblast treatment was statistically significant in identifiable improvement versus the vehicle control. The authors report the autologous injections were well tolerated without any permanent adverse events.
In summary, autologous fibroblasts continue to provide a safe alternative for multiple aesthetic conditions. Major downsides for more routine use include having to send the tissues to a CLIA-approved laboratory for fibroblast collection and the time required to prepare the fibroblast culture. In addition, the need to freeze the tissues for storage remains time consuming.
Regulations for Use of Autologous Fibroblast Cells and Adipose Tissues
The FDA oversees autologous fibroblast cells and autologous adipose tissue as human cells, tissues, and cellular and tissue-based products (HCT/Ps). HCT/Ps constitute human cells or tissues that are intended for implantation, transplantation, infusion, or transfer into a human recipient. The FDA takes a risk-based approach determining whether the HCT/P needs an Investigational New Drug (IND) application or a Biologics License Application (BLA) by determining whether the HCT/P is minimally manipulated. Minimal manipulation means that the structural tissue is not altered from its original relevant characteristics or the cells are not altered to change the biologic characteristics. HCT/P is for homologous use only and does not involve the combination of the cells or tissues with another article, except water, crystalloids, or a sterilizing, preserving, or storage agent that does not raise new clinical concerns. Also, the HCT/P needs to be for autologous, allogeneic use in a first- or second-degree blood relative or for reproductive use.
In most cases, the provider would need to follow the preceding rules to stay within the definition of the HCT/P so as not to be creating a drug or biologic that should be overseen by the FDA. Fibroblast cells need to be autologous and not altered to be used for point of care. If the fibroblast cells are not autologous or are altered, they fall under the purview of the FDA. Adipose tissue is a more complex situation, as in some cases it can be derived from liposuction and then reinjected into the patient in a different anatomical area. In such a case, the adipose tissue is being altered by isolating stem cells from the stromal vascular fraction. The FDA does view this as more than minimal manipulation. It is also important, if processing the autologous adipose tissue, that the provider uses an FDA-cleared/approved centrifuge or processor so that they do not have to worry about whether they are altering the tissue in a way that falls under FDA purview.
In general, most uses of autologous cells or tissues fall under the purview of the practice of medicine. When the FDA issues letters to providers, in most cases the provider has made product claims on their website for that specific cell or tissue use where the product claims fall under the purview of the FDA due to them manipulating the cell or tissue or by suggesting a clinical indication that has not been reviewed by the FDA. The use of these HCT/Ps is a hot topic within the industry, and it is expected that regulations will change in the future as the FDA learns more.
Platelet-Rich Plasma
History
The origins for the clinical use of PRP go back to the 1970s when hematologists were looking for treatment for patients with various platelet deficiency disorders (e.g., thrombocytopenia). Fast-forward 25 years, where orthopedic medicine and dental medicine have successfully used PRP for various degenerative joint disorders and bone degradation within the mouth. Orthopedic studies with PRP have demonstrated stimulation of de novo collagen and elastin in areas of poor vascularity. Other medical subspecialties, including thoracic surgery, cardiology, oral reconstructive surgery, periodontal dentistry, and wound healing, have embraced PRP as a safe and efficacious treatment modality.
Preparation
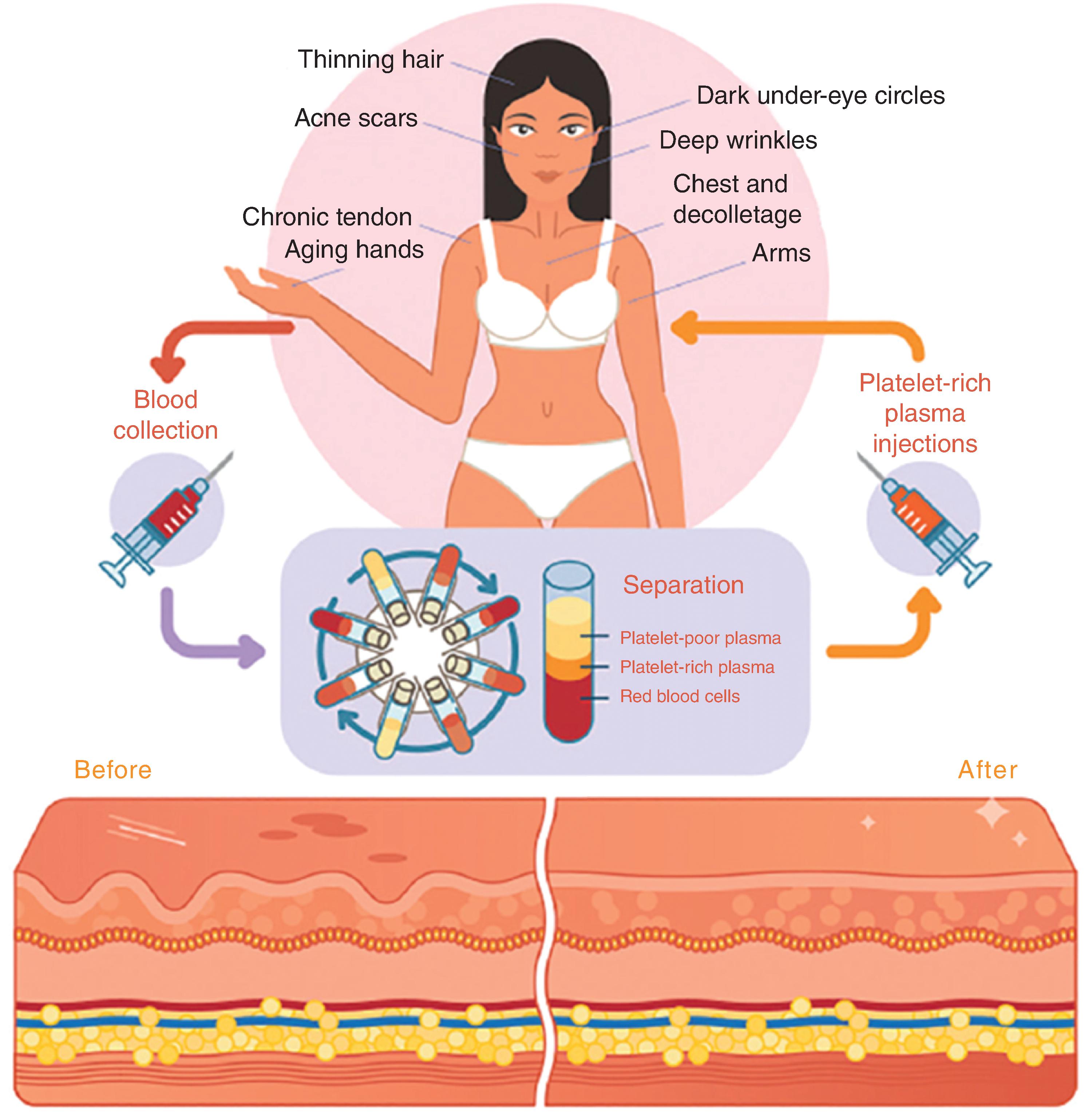
As evident with preparation of other autologous cosmeceutical products, extraction and preparation of the tissue is critical. Additionally, centrifugation is critical for reliable and reproducible PRP yields. There are multiple commercial centrifuges and PRP collection kits requiring FDA 510K clearance on the global market, and regulated devices designed to increase PRP yield and simplify the blood collection process are continually being developed. Clinical results have demonstrated that successful centrifugation separation steps to achieve functional PRP, with little blood cell contamination, are needed to observe identifiable, reproducible skin and hair changes. Fig. 15.4 shows an overview of PRP.