This article reviews the key elements of anesthesia care for patients presenting for craniofacial surgeries, including preoperative evaluation and preparation, intraoperative management, and pain management strategies.
Key points
- •
Preoperative preparation, patient optimization, and multidisciplinary involvement is critical in the anesthetic management of patients with craniofacial anomalies.
- •
Important factors in the airway plan include the patient’s age, development, underlying pathologies, and surgical procedure.
- •
Preoperative optimization and blood conservation strategies reduce transfusions in cranial vault reconstructions.
- •
Optimal multimodal analgesia enhances patient comfort, recovery, and facilitate discharge from the hospital.
- •
Animal data support the negative effect of general anesthesia on the developing brain. Human studies have shown mixed results.
Introduction
Craniofacial anomalies involve patients of all ages and include a wide range of disorders. Anesthetic plans are individualized to the patient, their underlying comorbidities, and the surgical procedure. Coordinated multidisciplinary care with clear communication among the patient’s providers is essential. This article is divided into 3 sections. The first section focuses on the preoperative assessment. The second addresses intraoperative management issues. The third section details pain management.
Preoperative considerations
An anesthesiologist’s preoperative evaluation not only depends on their own assessment but also depends on input from other subspecialists. Key elements of the preoperative evaluation include respiratory, cardiac, hematologic, and neurodevelopmental issues.
Respiratory and Airway Evaluation
Respiratory issues are common in patients with craniofacial anomalies. Underlying lung disease may be present in patients born prematurely. Even at older ages, patients with a history of prematurity are at risk for obstructive and restrictive lung disease. The 3 respiratory issues to consider for all pediatric surgical patients are upper respiratory tract infections (URIs), baseline obstructed breathing, and potential difficult airway.
Upper respiratory tract infections
Patients with URIs pose a challenge for the anesthesiologist and are the leading cause of surgery cancellations. Adverse events include laryngospasm, bronchospasm, oxygen desaturation, and bradycardia. The predictors of perioperative respiratory adverse events (PRAEs) include URIs, reactive airway disease, chronic lung disease related to prematurity, age, second-hand tobacco exposure, and airway surgery. Airway management with an endotracheal tube (ET) poses additional risk to airway irritability.
The COLDS score ( Table 1 ) is a validated risk assessment tool, which provides a framework to quantify the risk for PRAEs. The score ranges from 5 to 25, with higher scores being associated with greater risk. For children undergoing general anesthesia with an ET for craniofacial surgeries, the presence of an acute URI poses a higher risk for PRAEs. For elective surgeries, the discussion to proceed with or delay surgery should include anesthesiologist, surgeon, guardian, and, if appropriate, the patient. It is important to consider the child’s comorbidities, current illness, planned anesthetic, and surgery.
| 1 | 2 | 5 | |
|---|---|---|---|
| C Current signs/symptoms | None | Mild Examples: Parent confirms URI and / or congestion, rhinorrhea, sore throat, sneezing, low fever, or dry cough | Moderate/severe Examples: Purulence, wet cough, abnormal lung sounds, lethargy, “toxic” appearance, or high fever |
| O Onset | >4 wk ago | 2–4 wk ago | <2 wk ago |
| L Lung disease | None | Mild Examples: Hx of RSV, mild intermittent asthma, CLD if >1 y old, loud snoring, or passive smoker | Moderate/severe Examples: Moderate persistent asthma, infant with CLD, OSA, or pulmonary HTN |
| D Airway device | None or facemask | Laryngeal mask airway or supraglottic | Tracheal tube |
| S Surgery | Other (including gear tubes) | Minor airway Examples: T/A, NLD probe, flexible bronchoscopy, and dental extractions | Major airway Examples: cleft palate, rigid bronchoscopy, and maxillofacial |
Obstructed breathing
Patients with craniofacial anomalies may present with varying degrees of airway obstruction. For many children, the severity of airway obstruction is most pronounced during sleep, which manifests as snoring, sleep disordered breathing, and obstructive sleep apnea (OSA). For some children, such as those with Pierre Robin Sequence (PRS) or significant midface hypoplasia, airway obstruction may be present while awake.
Airway obstruction can occur at many levels, from the nose to the subglottic area. Preoperative involvement of the otolaryngologist to evaluate the level and severity of airway obstruction is critical. Prior to surgery, flexible fiberoptic nasolaryngoscopy can identify the site of airway obstruction, while polysomnography can quantify OSA severity and determine if there is a central component. Chronic obstructed breathing can cause chronic hypoxemia, pulmonary hypertension, and increased opioid sensitivity.
Airway examination
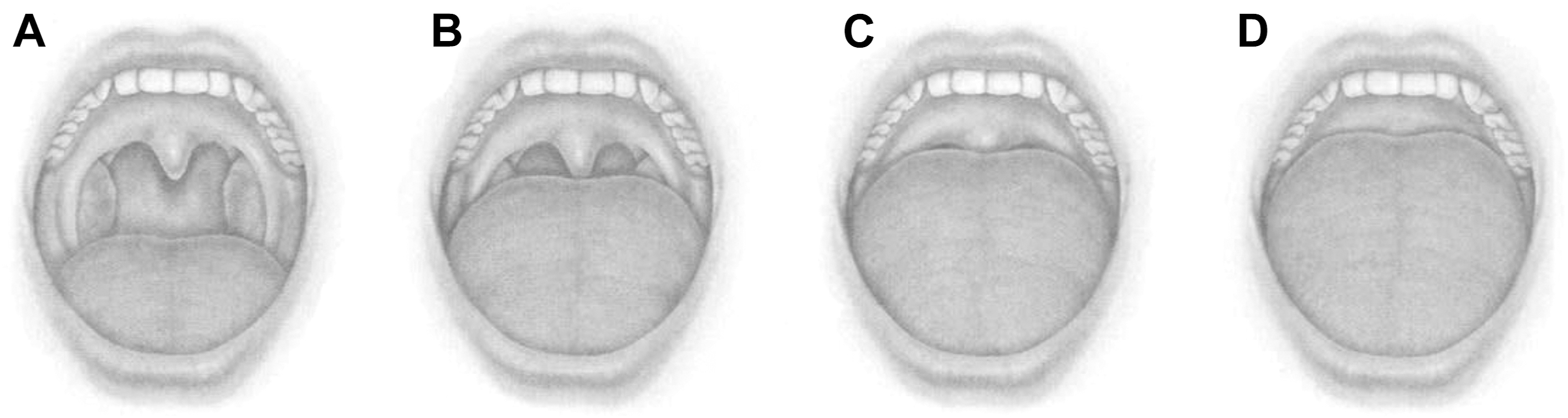
Pediatric patients presenting for craniofacial procedures may present the anesthesiologist with airway challenges, and thus, the airway evaluation is an essential part of the history and examination. The airway examination includes the evaluation of the patient’s craniofacial anomalies, cervical mobility, oropharynx, and work of breathing. Cervical extension is critical to opening the upper airway. In patients with Goldenhar syndrome as well as other skeletal dysplasias, there may be reduced cervical extension due to varying degrees of vertebral fusion. The size and shape of the head, midface, mandible, presence of cleft lip and/or palate, along with dentition and tongue size should be noted. When appropriate, anesthesiologists assign a Mallampati score, a noninvasive assessment of the tongue base relative to oral opening, which predicts visualization of the glottis ( Fig. 1 ). Work of breathing including the patient’s position, respiratory rate, and retractions provide evidence of airway obstruction and pulmonary reserve. Pediatric craniofacial syndromes and associated airway anomalies are described in Table 2 .
| Syndrome | Anesthesia Management Concerns |
|---|---|
| Apert syndrome (Acrocephalosyndactyly Type 1) |
|
| Carpenter syndrome (Acrocephalopolysyndactyly type 2) |
|
| Craniofrontonasal dysplasia |
|
| Crouzon syndrome (Craniofacial dysostosis) |
|
| Deletion 22q11.2 syndrome (Velo-cardio-facial syndrome, DiGeorge syndrome, and Shprintzen syndrome) |
|
| Goldenhar syndrome (Oculo-auriculo-vertebral spectrum, hemifacial microsomia, and first and second branchial arch syndrome) |
|
| Muenke syndrome (FGFR3-associated coronal synostosis syndrome) |
|
| Pierre Robin sequence |
|
| Pfeiffer syndrome (Pfeiffer-type acrocephalosyndactyly) |
|
| Saethre-Chotzen syndrome (Acrocephalosyndactyly type III) |
|
| Treacher Collins syndrome (Franceschetti–Klein syndrome) |
|
Cardiac Evaluation
Patients with craniofacial anomalies may have associated congenital heart disease (CHD), which carries a higher anesthetic risk (see Table 2 ). Seventy-five percent of patients with 22q deletion syndrome and up to 50% of patients with Goldenhar and Carpenter syndromes have CHD. If a complex CHD is present, management by a pediatric cardiac anesthesiologist is recommended. Prior to general anesthesia, a review of the child’s symptomatology, physical examination, and prior cardiac evaluation should be performed. Pre-existing cardiac conditions should be optimized prior to surgery with the child’s cardiologist and the need for infective endocarditis prophylaxis should be clarified.
Hematological Optimization and Preparation for Potential Blood Transfusion
Intracranial and fronto-facial procedures may involve substantial blood loss, leading to coagulopathy and the need for intraoperative blood transfusions. Preoperative strategies to minimize the need for transfusions should be discussed with patients and families.
Iron deficiency anemia is the most prevalent type of anemia in children globally. Treatment includes an iron-rich diet, and supplemental ferrous sulfate 3 mg/kg daily, with follow-up testing in 2 to 4 weeks.
The use of recombinant human erythropoietin (EPO) is well-studied in craniosynostosis surgeries. EPO administration in conjunction with iron 3 weeks prior to surgery significantly increases hemoglobin and lowers the rate of intraoperative transfusion. EPO is dosed at 600 units/kg weekly for 3 weeks prior to surgery and coadministered with iron (4 mg/kg elemental iron daily). Data from 369 pediatric patients undergoing cranial vault remodeling showed EPO was not associated with postoperative thrombotic complications. Despite this efficacy and safety data, as of 2017, EPO was utilized in only 0.2% of pediatric cranial vault reconstructions. Historically, EPO can only be administered intravenously or subcutaneously. In 2023, the US Food and Drug Administration (FDA) approved daprodustat, a hypoxia-inducible factor prolyl hydroxylase inhibitor, which stimulates EPO production; this medication is available as an oral formulation but has limited indication only for adults with chronic kidney disease.
For surgeries where bleeding risk is high, systematic processes should be in place to ensure availability of blood prior to surgical incision. Preparation of patients begins in the outpatient clinic, including communication with family, blood screening, and ordering appropriate blood products. For patients and families who object to blood transfusions, the need for preoperative conversations and preparation is especially critical.
Neurobehavioral and Developmental Evaluation
Patients with craniofacial anomalies present with a wide range of intellectual abilities and developmental behaviors, which can affect their response to anesthetic induction, emergence, and recovery (see Table 2 ). Discussing induction options with both the patient and family is essential. Regardless of the patient’s neurodevelopmental status, the craniofacial phenotype associated with many forms of syndromic craniosynostosis and other congenital anomalies can result in social stigma. Thus, sensitivity to these issues is important.
Some patients with craniosynostosis may have elevated intracranial pressure (ICP). The prevalence of increased ICP is 15% to 20% in patients with single suture nonsyndromic synostosis and 30% to 40% in patients with syndromic synostosis. These patients may have concomitant optic neuropathy, placing them at risk for vision loss. Children with craniosynostosis should be evaluated by an ophthalmologist, whose investigations include performing visual evoked potentials to assess the integrity of the visual pathway.
For patients with elevated ICP, clear communication between the anesthesiologist and neurosurgeon should take place prior to craniotomy. Anesthetic strategies may be employed to acutely decrease ICP, such as hyperventilation to reduce end tidal carbon dioxide levels and administration of mannitol or hypertonic saline.
Neurocognitive effects of early anesthetic exposure in children
Recently, long-term effects of anesthetic agents on neurodevelopmental outcomes have become a concern. In 2016, the FDA warned about the risks on the developing brain from administration of anesthetics for children aged under 3 years as well as pregnant women during their third trimester, especially with repeated exposures or procedures lasting more than 3 hours. Animal studies have demonstrated negative neural effects after early anesthetic exposure. Findings in humans have been less cohesive, due to the heterogeneity of the study outcomes, differences in patient comorbidities, home environments, and length of anesthetic exposures. In humans, there is a possible associated effect of repeated anesthetic exposure in early childhood with adverse child development outcomes, though existing studies have had varying conclusions.
In animals, from rodents to nonhuman primates, studies have demonstrated the effects of anesthesia on the developing brain. Studies in rodents described an increase of apoptosis in brains and impaired learning and memory formation. In nonhuman primates, isoflurane exposure was associated with resting-state functional connectivity alterations of the amygdala and the posterior cingulate cortex with other brain regions, increased astrogliosis, and a decrease in the animal’s close social behavior.
Comparison among human studies is difficult, due to heterogeneity of study primary outcomes. In 2016, a multi-institutional study found that children exposed to anesthesia had more behavioral issues compared to their siblings but no difference in intelligence quotient (IQ) scores. Multiple anesthetics, as opposed to a single anesthetic exposure, were associated with an increase in behavioral and learning difficulties.
In contrast to the aforementioned findings, other studies have shown that children exposed to general anesthesia in early childhood did not have worse child developmental outcomes, standardized testing, and/or IQ testing, compared to unexposed biological siblings or twin siblings.
In addition to concerns regarding intelligence and academic performance, issues of an association of early anesthetic exposure with attention deficit hyperactivity disorder (ADHD) and autism have also been raised. Using a Medicaid database from Texas and New York over an 11 year period, Ing and colleagues reported that children who were exposed to anesthesia and surgery before 5 years of age had a higher likelihood of receiving ADHD medications. In another study, children with exposure to 2 or more anesthetics before the age of 2 years were more likely to have a diagnosis of ADHD. There is no demonstrable association between anesthesia and the development of autism.
Intraoperative anesthetic management
Airway Management
Management of the airway in children starts with an age-appropriate plan. Proper management includes performing a preoperative assessment, formulating an airway plan, and discussing concerns among the involved providers.
In patients with an anticipated normal airway, an ET or laryngeal mask airway (LMA) is placed with ease. A child with a “difficult airway” may be challenging to mask and/or is unintubatable by direct laryngoscopy and videolaryngoscopy. In children, difficult facemask ventilation can occur in 6.6% and difficult intubation in 1% of patients.
In adults and older children with difficult airways, awake (sedated) fiberoptic intubation is preferred as this technique maintains spontaneous ventilation; in the event the trachea cannot be intubated, the patient can be easily awakened. In young pediatric patients, expecting cooperation for awake intubation is unrealistic.
There is no consensus on the management approach to the child with a difficult airway. Conventionally, deep sedation or general anesthesia with spontaneous ventilation is practiced. More recently, from the Pediatric Difficult Intubation (PeDI) Registry, Garcia-Marcinkiewicz noted that, during the intubation of a child with a difficult airway, maintenance of spontaneous ventilation was associated with more complications than controlled ventilation. In an attempt to maintain spontaneous ventilation, one may provide inadequate anesthetic depth, risking laryngospasm and bronchospasm. Regardless of technique, supplemental oxygenation with high-flow nasal cannula is beneficial for airway procedures, for apneic oxygenation as well as during spontaneous ventilation.
Mask ventilation and upper airway obstruction
Successful mask ventilation requires a good mask seal and a patent upper airway. Proper mask seal can be problematic in many craniofacial syndromes. In children with Goldenhar syndrome, for example, asymmetric facial bone and soft tissue development may compromise mask seal.
Upper airway obstruction can occur anywhere from the nose and mouth down to the supraglottic area. In patients with Apert syndrome, airway obstruction due to congenital bony nasal stenosis can be as high as 85%. With PRS, micrognathia and glossoptosis cause upper airway obstruction in 46% to 100% of patients.
Multiple techniques can relieve upper airway obstruction. Two hand mask ventilation improves mask seal. Effective jaw thrust relieves airway obstruction by lifting the tongue off the posterior pharynx. Similarly, oral and nasopharyngeal airway devices provide a conduit for oxygen by displacing the posterior aspect of the tongue off the larynx. Continuous positive airway pressure can stent the airway open and limit soft tissue collapse. Another technique is the use of a tongue stitch, which allows the tongue to be pulled anteriorly, lifting the tongue off the posterior airway and elevating the epiglottis.
Endotracheal intubation
Methods for securing the patient’s airway can involve single, combined, and invasive techniques. Single techniques for intubating the trachea include direct laryngoscopy, video laryngoscopy, flexible fiberoptic intubation, and rigid bronchoscopy. Combined techniques include flexible fiberscope via a supraglottic airway (LMA) or video-assisted fiberoptic bronchoscopy. Invasive approaches include cricothyrotomy, tracheostomy, retrograde intubation, and submental approaches; these approaches will not be discussed here.
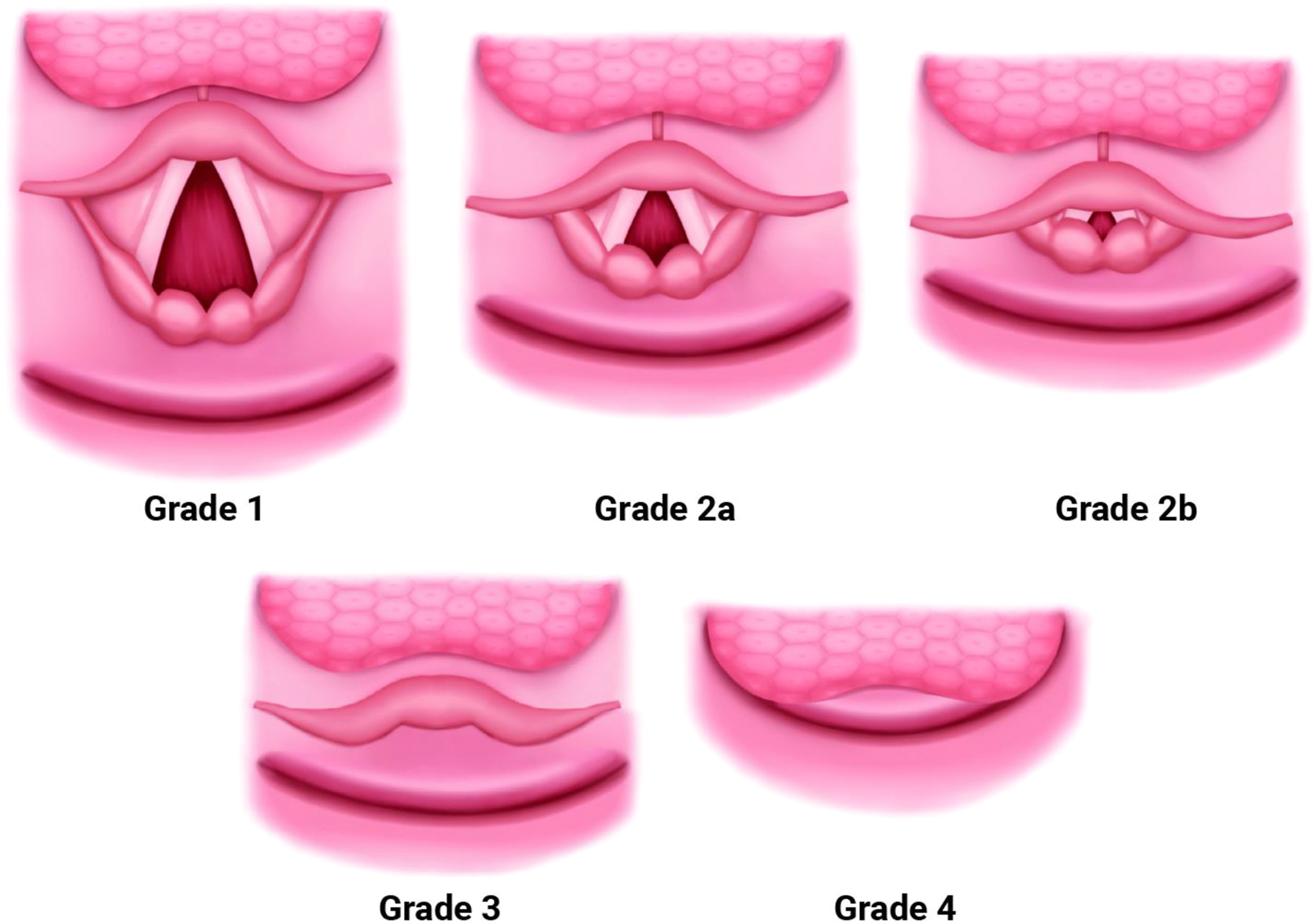
With conventional direct laryngoscopy, the patient is placed in a “sniffing” position to align the axes of the airway: oral, pharyngeal, laryngeal. With laryngoscopy, the Cormack–Lehane (CL) grade of the glottic opening predicts the difficulty of intubation ( Fig. 2 ). CL grades range from 1, a full view of the glottic opening, to 4, which is visualization of only the soft palate. In patients at risk for cervical fusion, such as those with Crouzon, Apert, and Goldenhar syndromes, limited cervical extension increases the difficulty of intubation.