9 Modified Radical Mastectomy
Indications
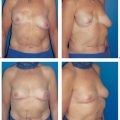
Breast-conserving surgery is now regarded throughout the world as being the standard procedure for the treatment of breast cancer. However, if it is contraindicated, or if the patient so wishes, a modified radical mastectomy has to be performed. A modified mastectomy rate of 30 % can be expected.
The indications for modified radical mastectomy include factors related to tumor biology, such as multicentric growth, extensive ductal carcinoma in situ, and signs of inflammation. A modified radical mastectomy may also be indicated if there is an unfavorable relation between breast and tumor size. Finally, the patient’s inability to undergo postmastectomy radiotherapy (e. g., due to a funnel-chest deformity) or difficult radiological follow-up may also be indications.
The term “modified radical mastectomy” is derives from Patey (1948), who found in comparative studies that preserving the pectoralis major—in contrast to the radical mastectomy operation using Halstedt’s technique (1882)—did not compromise local tumor control. Auchincloss (1963) modified the procedure to preserve the pectoralis minor and level III nodes. The procedure is still carried out in this fashion today.
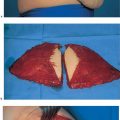
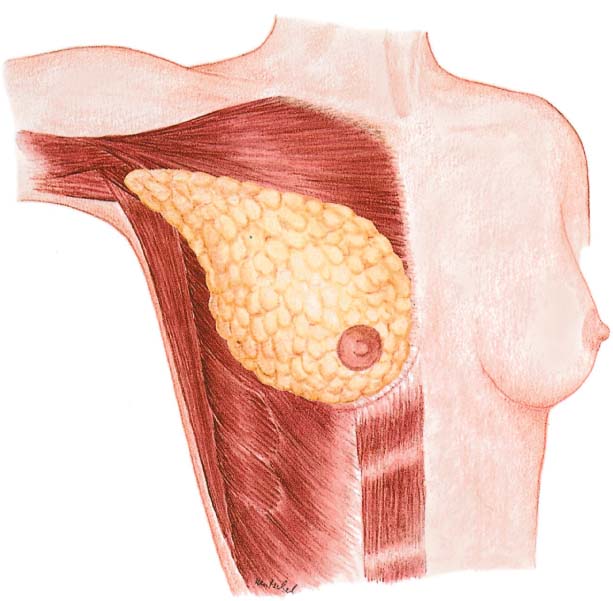
Modified radical mastectomy is defined as a total mastectomy—that is, complete removal of the mammary gland, including the nipple–areola complex. The skin envelope, including the pectoralis fascia, is preserved for primary wound closure. Level I and II axillary lymph nodes are dissected. It remains to be seen whether the nomenclature will change following the introduction of sentinel lymph-node dissection.
Surgical Technique
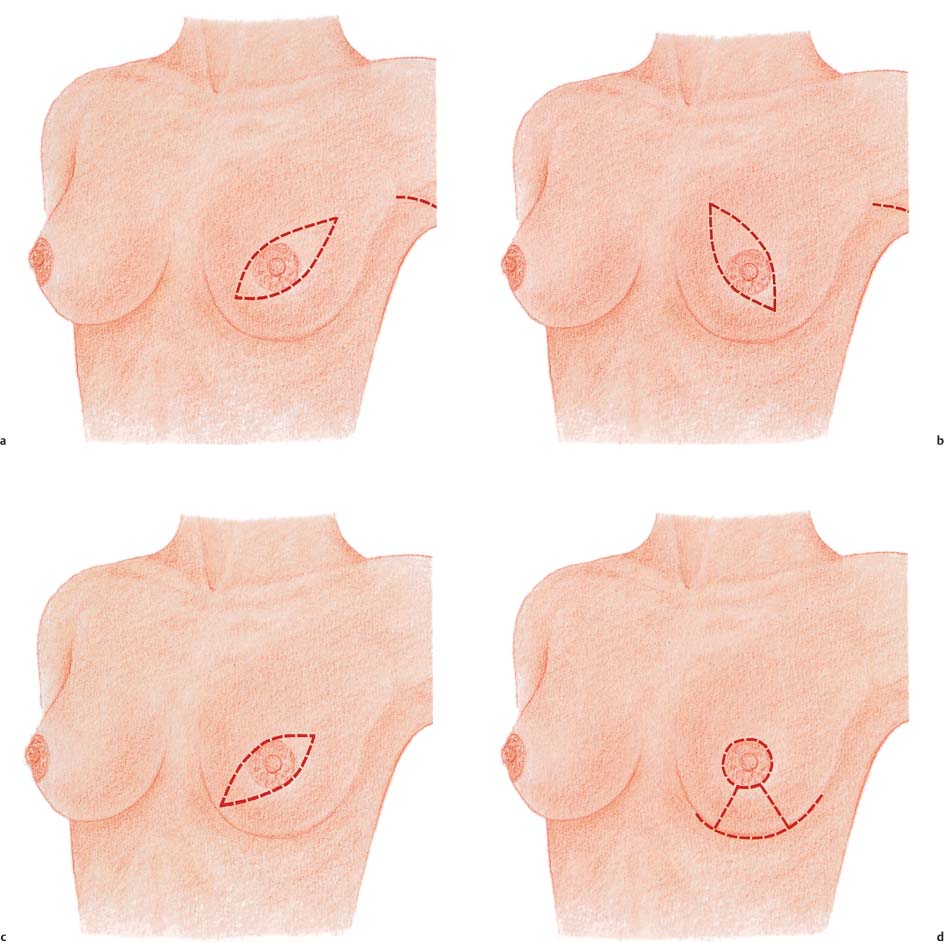
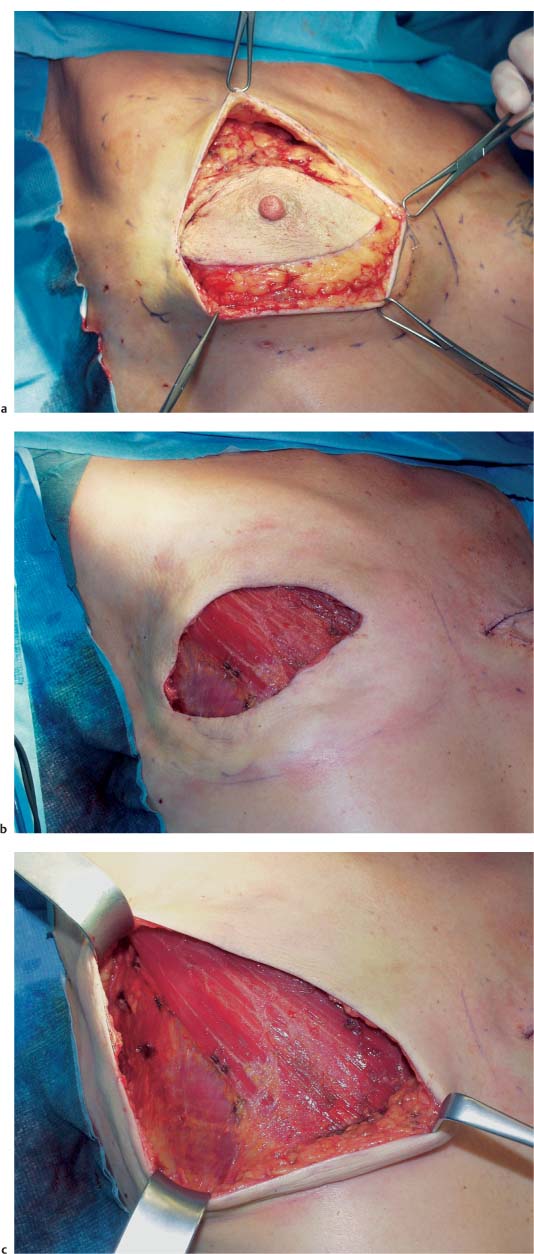
The skin incision in a modified radical mastectomy is oriented around the tumor site and nipple–areola complex. The advent of skin-sparing mastectomy, usually performed in conjunction with immediate breast reconstruction, has altered the concept of modified radical mastectomy (see Chapter 11). In rare circumstances, if the tumor has not infiltrated the skin, a periareolar incision can preserve the entire skin envelope. Resection with healthy margins must be ensured.
In mastectomies performed without planned reconstruction, it is necessary to resect a larger area of skin with the nipple in order to obtain good wound closure.
The preferred incision is transverse or oblique, and extends from superolateral to inferomedial. Resection of tumors high in the superomedial quadrant is challenging, in that the scar may extend into the cleavage area. Ultimately, the primary concern is local tumor control.
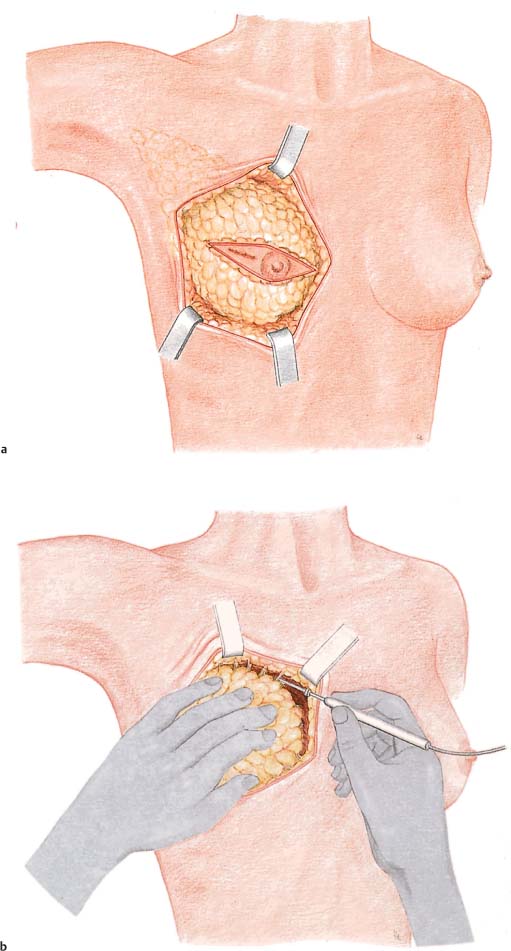
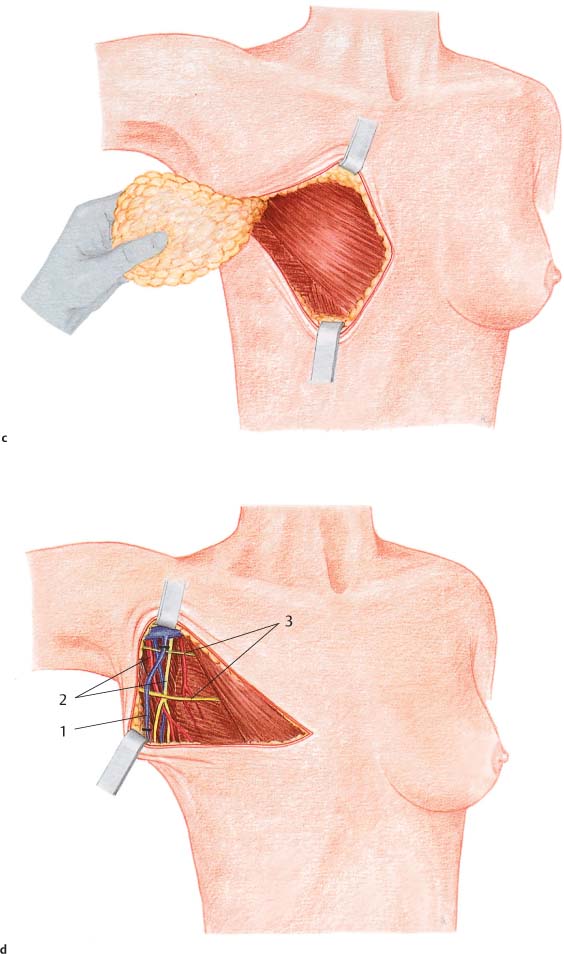
Grasping the superior and inferior skin flaps with ring forceps facilitates subcutaneous dissection of the gland by keeping the skin taut. Care should be taken to remove all of the glandular tissue from the subcutaneous tissue. The technically challenging steps are subcutaneous dissection and exposure of the borders of the gland. Dissection of the glandular tissue off the pectoralis muscle and resection of the pectoralis fascia (following the course of the lymphatic vessels) is technically easier, as it constitutes an anatomically well-defined layer.
Subcutaneous dissection of glandular tissue continues into the periphery—namely, from the second intercostal space into the inframammary fold and from alongside the sternum into the anterior axillary line. The glandular tissue is then dissected from medial to lateral off the chest wall or underlying muscles. Subcutaneous dissection can be performed well with scissors; for dissecting the breast tissue off the chest wall, a scalpel or electro-cautery knife is recommended. There is an increased risk of bleeding near the parasternal arterial perforators and from superficial veins traveling diagonally through the upper pole of the breast. Bleeding from larger vessels entering from lateral also has to be controlled.
In modified radical mastectomy, classic axillary dissection is performed en bloc. Dissection is carried from the peripheral projections of the breast tissue over the lateral border of the pectoralis directly into the axillary adipose tissue. Further exposure is carried out as described in the section on axillary dissection in Chapter 10 (p. 227).
Sentinel lymph-node dissection can be performed through the mastectomy skin incision. After opening of the preaxillary fatty tissue, the “hot” lymph node or nodes are removed with the aid of a gamma probe.
Use of tumescent infiltration can help minimize bleeding during dissection. However, infiltration of the subcutaneous tissue should not extend to the area immediately around the tumor. Also, in high-risk patients (smokers, diabetics, and those with thin subcutaneous tissue), the epinephrine dosage should be kept low enough to avoid skin necrosis.
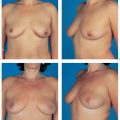
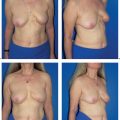
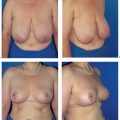
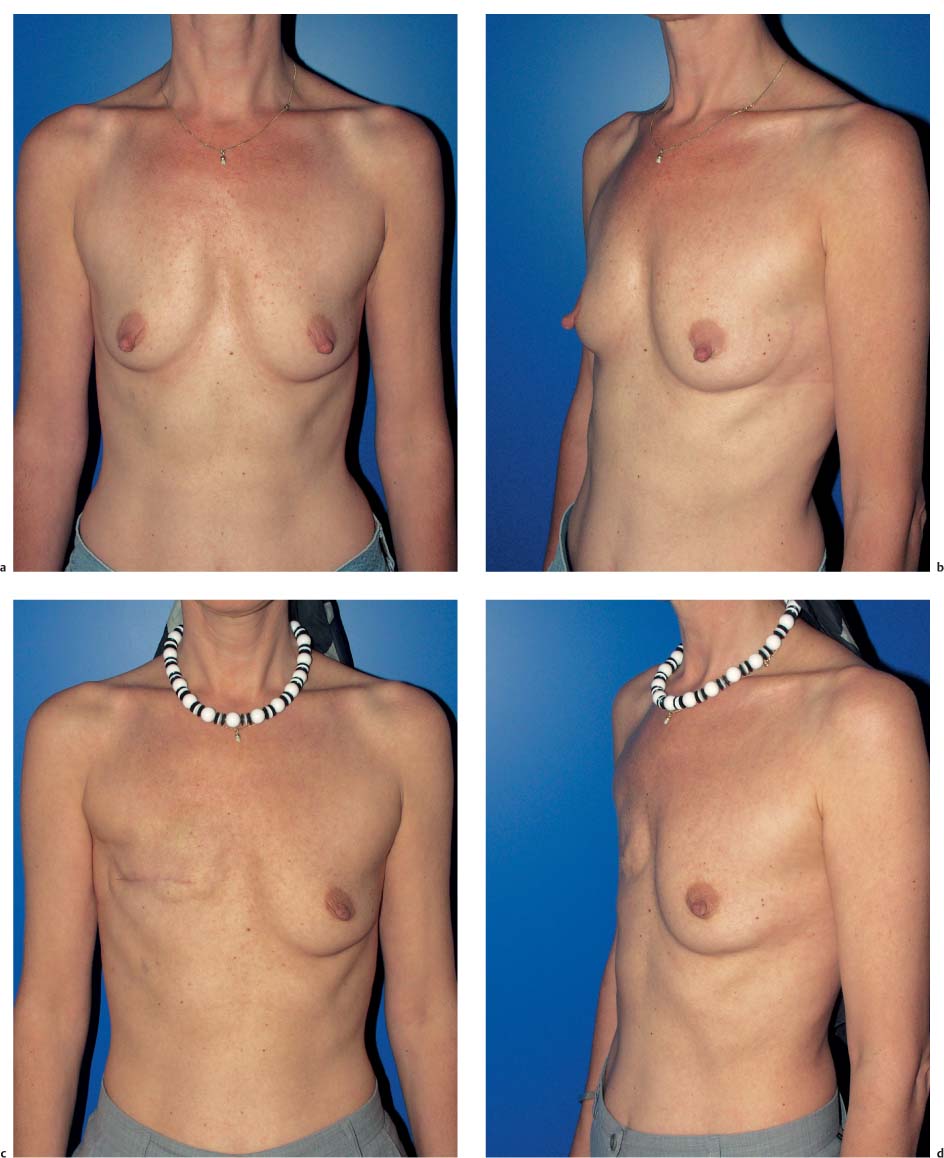
The functional complaints once associated with modified radical mastectomy (restricted arm motion, chronic pain due to a lack of soft-tissue coverage) are now very uncommon. However, the operation leaves a contour defect on the chest wall, which may be an even greater burden psychologically than functional impairment. Every patient should be informed about the option of immediate breast reconstruction (Chapter 11).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree