10 Breast-Conserving Therapy
Indications
Breast-conserving surgery has become the standard form of treatment for breast cancer. Nowadays, ablative measures are only indicated when factors related to tumor biology do not support preservation of the breast, or if there is a poor relationship between breast size and tumor size.
Surgical treatment has become less significant in proportion to the extent to which adjuvant systemic therapy has gained in importance—at least in relation to achieving a cure. Surgical techniques are thus also becoming increasingly less radical.
The first edition of this book, published over 10 years ago, described breast-conserving therapy as an optimal method of treating small tumors. This view is now completely outdated. Breast-conserving surgery can even be used for very large tumors, provided that a cosmetically satisfactory breast shape can still be obtained after tumor resection. Particularly in larger tumors, the prognosis depends not on local growth, but rather on systemic spread.
The clinical criteria set forth by Harris still apply:
Adequate resection of the tumor must be ensured without causing a major deformity of the breast.
Local tumor control must be comparable to the rate of local recurrence after mastectomy.
A key issue is therefore the extent of tumor excision needed to ensure local tumor control. On the one hand, the rate of local recurrence depends on resection with tumor-free margins, while on the other, it is the size of the tissue defect that determines the aesthetic result. This potential conflict of interest must never be allowed to compromise oncological safety.
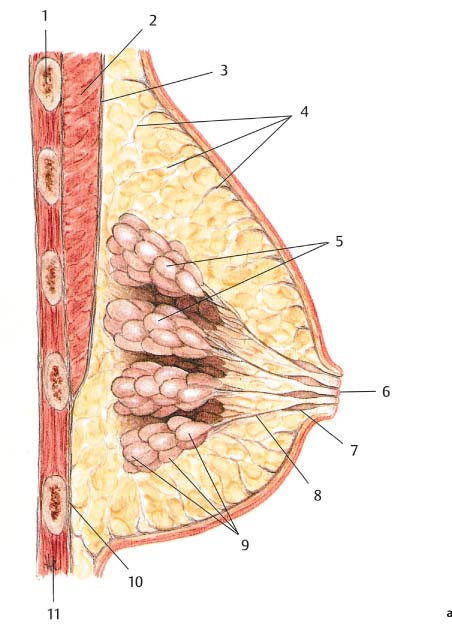
Segmental resection is the standard procedure, in view of the radial arrangement of the breast lobules. Resection with a tumor-free zone of about 1 cm has to be confirmed histologically. This is particularly important in poorly-differentiated tumors (grade 3), which have a higher risk of local recurrence.
Tumors with a large intraductal component are another problem. Intraductal lesions can occur as “skip lesions,” with more than 10 mm of normal tissue between them. Wide resection (e. g., quadrantectomy) should be performed to allow broad assessment of the breast tissue.
Contraindications to breast-conserving surgery include multi-centric tumors, an extensive intraductal component related to invasive carcinoma, a large tumor size relative to that of the breast, or a contraindication to postoperative radiotherapy. Younger patients (under 35) should also be informed about the increased risk of local recurrence. Factors such as tumor size exceeding 2 cm or lymph-node involvement are no longer regarded as contraindications to a breast-conserving procedure.
Postoperative radiotherapy is an essential part of breast-conserving therapy, and patients should be informed about this before surgery. As some radiation oncologists also administer boost radiotherapy to the previous tumor bed, and as glandular tissue is often displaced during plastic surgery, the (superior and inferior) resection margins should be marked using titanium clips to aid the radiation oncologist.
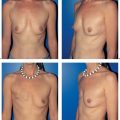
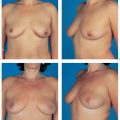
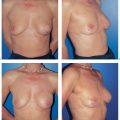
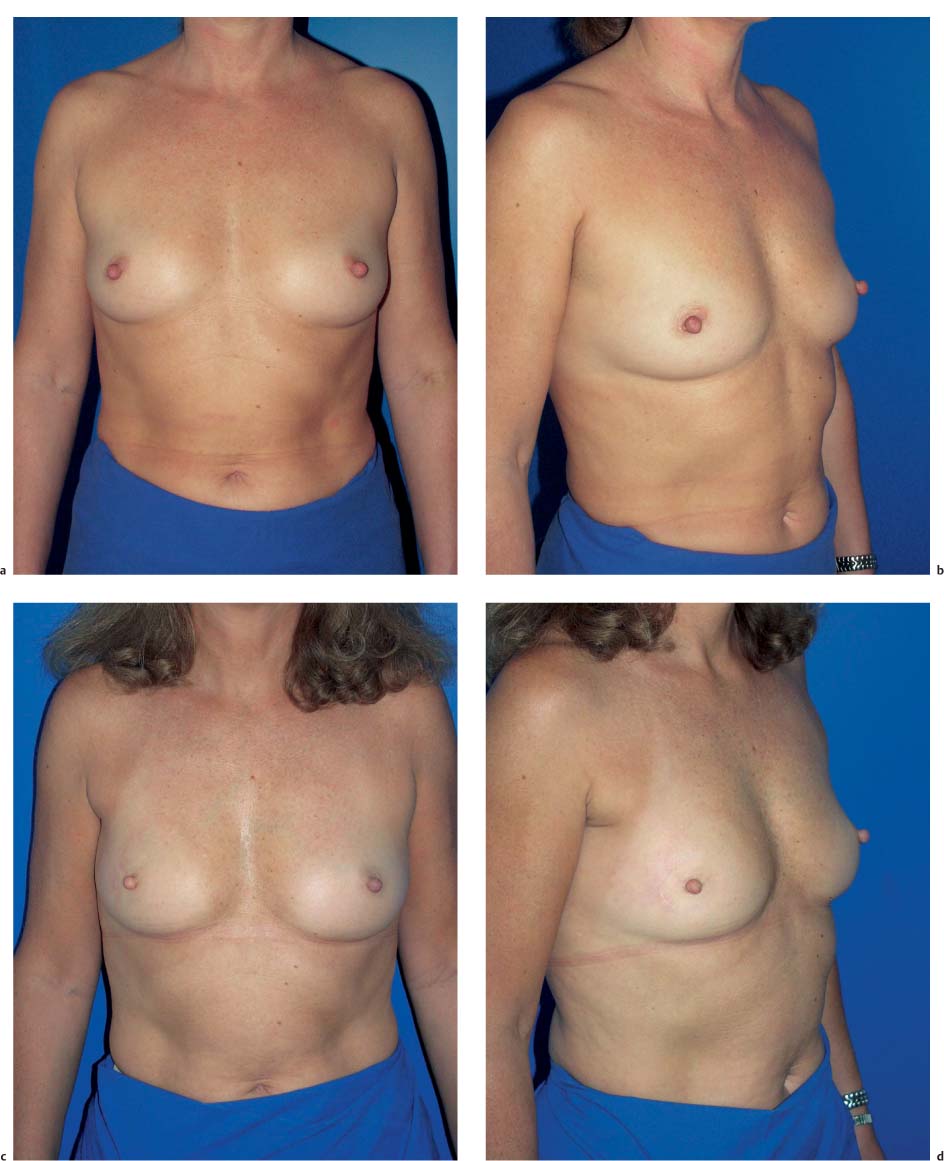
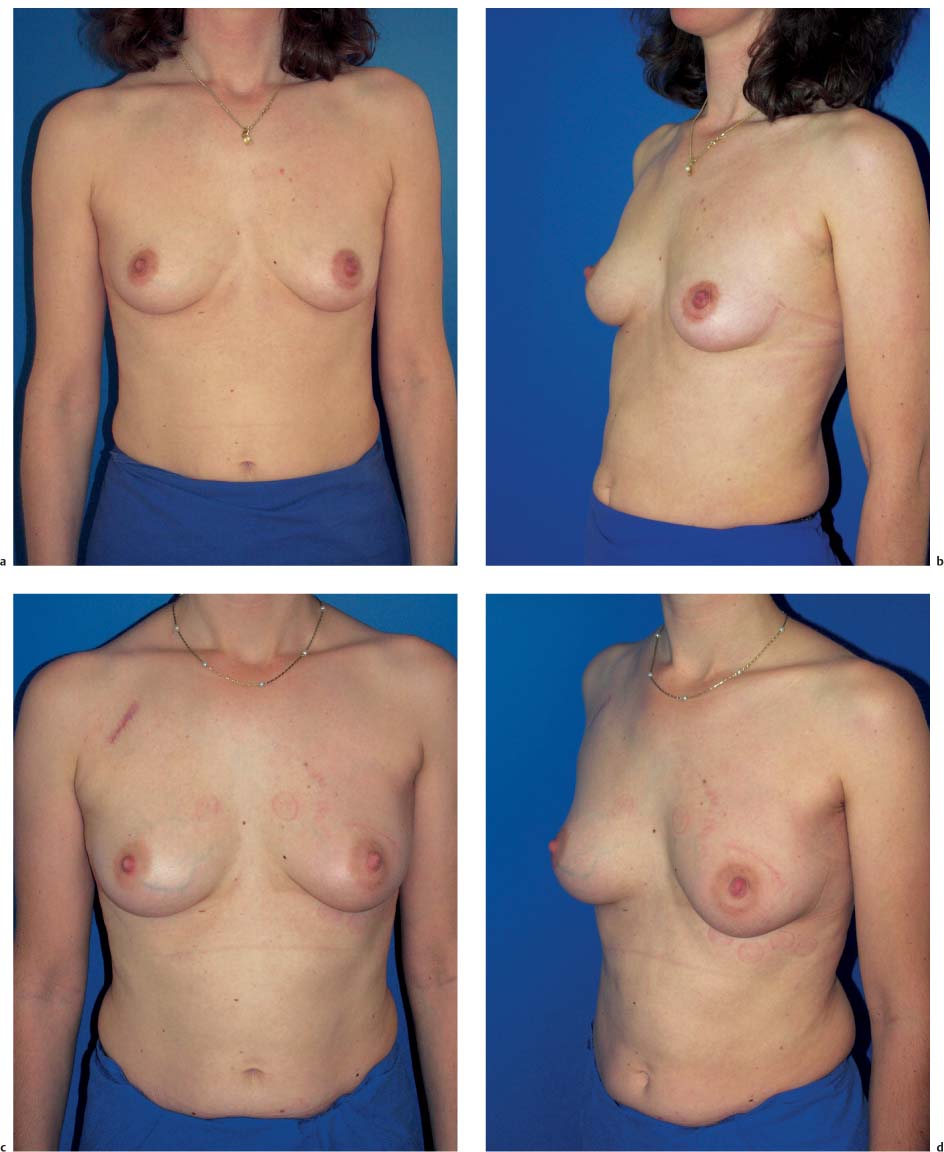
The cosmetic outcomes of breast-conserving therapy vary from one surgeon to the next. Generally, as one gains experience with the technique, the poor results initially seen become less common. The concept of allowing the defect to fill by hematoma formation has fallen out of favor. In some patients, this technique led to very poor results in conjunction with subsequent radio-therapy.
Nowadays, the defect is usually corrected by reapproximating the breast parenchyma. In the majority of patients, a subcutaneous parenchymal flap is a very good method of treatment.
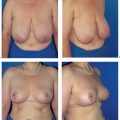
Another useful surgical strategy is to combine tumor resection and simultaneous breast reduction in patients with large breasts or with a disproportionate tumor/breast size. This not only provides excellent surgical results in terms of the aesthetic outcome, but also has positive secondary effects on the patient (see below and Chapter 4).
The choice of surgical procedure does not influence adjuvant systemic therapy, which is determined by individual factors and tumor biology.
Surgical Technique
Breast-conserving therapy consists of complete excision of the primary tumor, with or without resection of level I and II axillary lymph nodes (or sentinel lymph-node dissection), and follow-up radiotherapy of the remaining breast parenchyma.
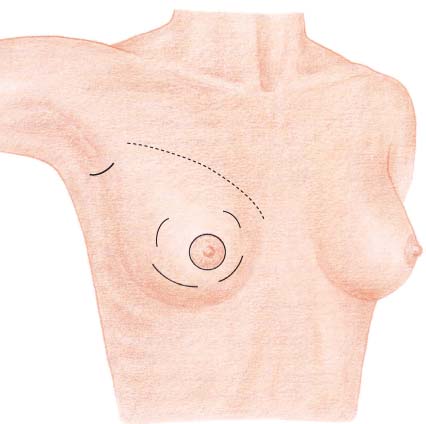
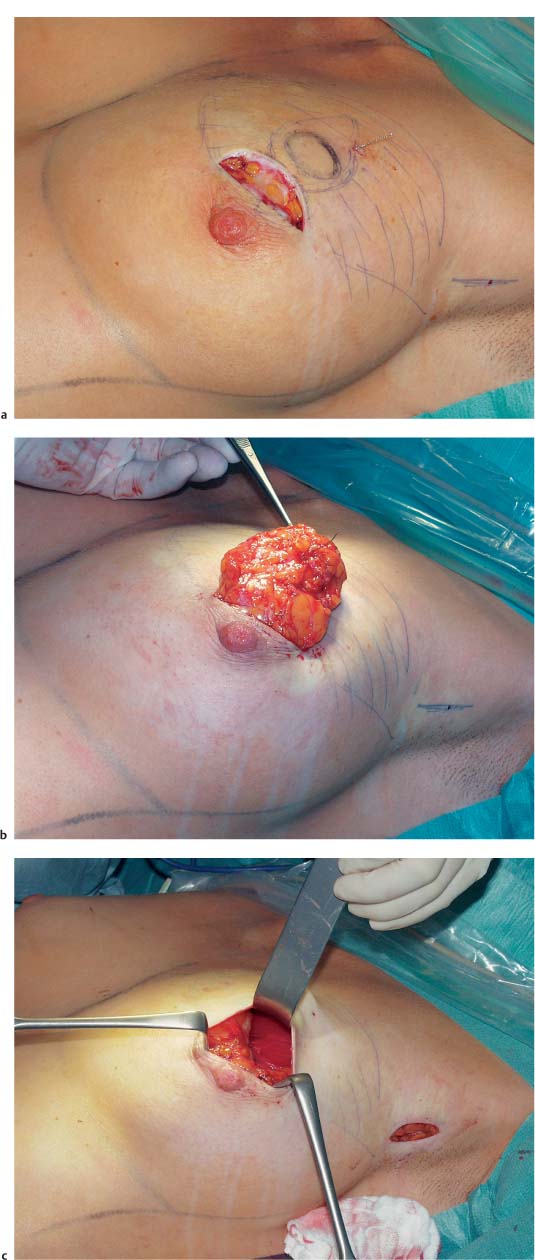
If the tumor is palpable, a curvilinear incision is made in the overlying skin (or nearby), followed by wide subcutaneous dissection of the parenchyma surrounding the tumor ( Fig.10.2 ). A periareolar incision is preferable when possible. The skin incision should not be carried into the cleavage area. A more direct surgical route is usually unnecessary, due to the subsequent radiotherapy; at most, it can be used to obtain better exposure of the surgical field. Resection of a skin ellipse is only indicated if there is skin infiltration or tethering.
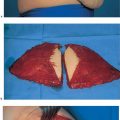
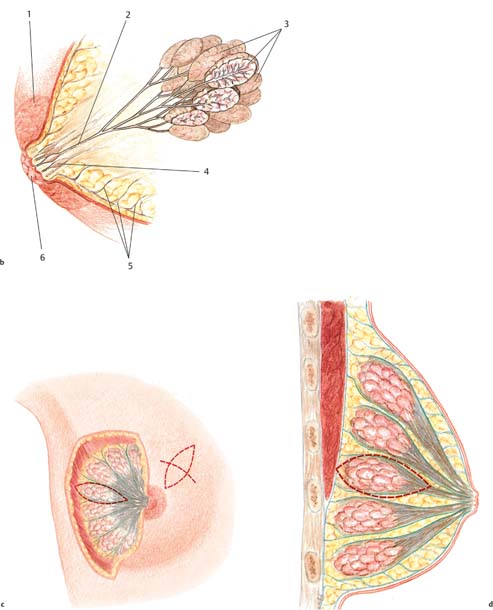
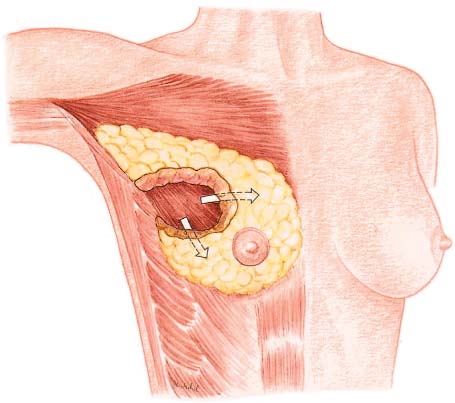
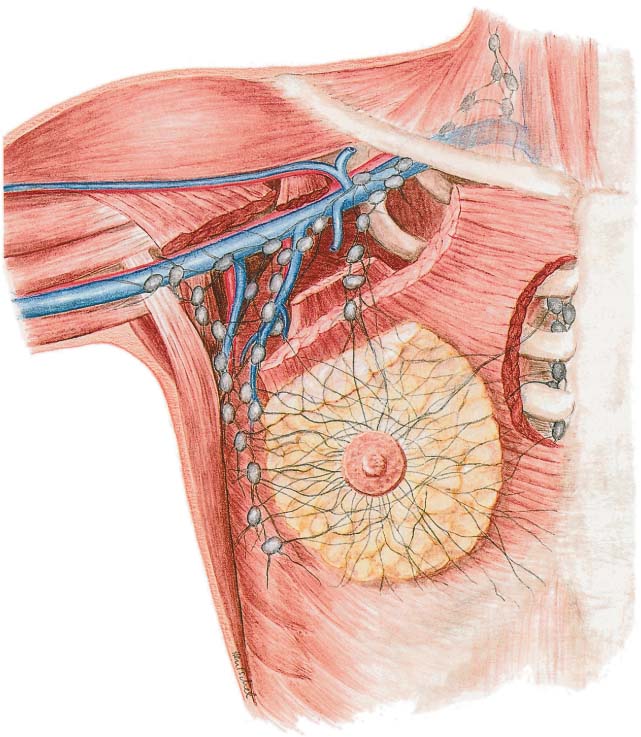
Tumor resection should be carried out with consideration of the radial arrangement of the lobules, ductal system, and lymph vessels of Cooper’s ligaments ( Fig.10.1 ). The anatomy of the breast thus makes segmental resection the most logical method of removing the tumor. All of the breast tissue, from the subcutaneous tissue to the pectoralis fascia, should be excised. Merely removing the lesion from the parenchyma is too imprecise and should be avoided.
Firstly, using digital palpation, the breast tissue is dissected along one side with a margin of safety around the tumor. Using a grasping forceps to remove the tumor is not advised, as it limits palpation of the lesion. Next, the segment containing the tumor can be dissected off the pectoralis muscle. This allows the breast tissue to be undermined with one finger, facilitating palpation of the tumor and subsequent resection with an adequate margin. The segment containing the tumor is resected in toto ( Figs.10.3 and 10.5 ).
After the specimen has been removed, it is reinspected by palpation to determine whether and where any additional resection is needed.
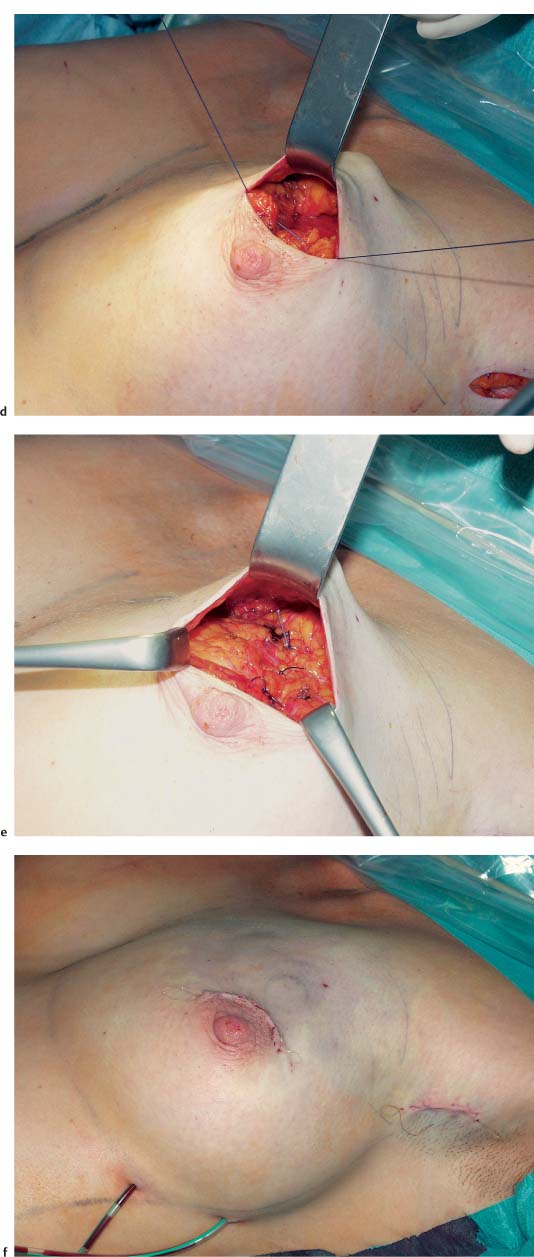
Next, the specimen is tagged with at least three threads—superior, medial, and lateral markings are helpful. It is advisable to discuss with the pathologist the preferred method of marking the mass. The specimen can also be sent for immediate frozen-section histological analysis if needed.
In addition to diagnosing the tumor type, the most important aspects for the surgeon are ensuring free margins and assessing the extent of a potential intraductal component. If there is any doubt, it is preferable to carry out a reexcision during the same procedure, rather than subjecting the patient to a later one.
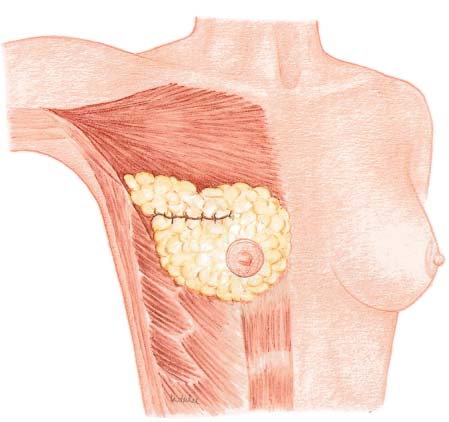
A subcutaneous glandular advancement flap is the method of choice for closure of smaller and moderately sized defects. Creating the flap involves wide subcutaneous dissection of the parenchyma directly adjacent to the defect. Tumescent infiltration—i. e., subcutaneous infiltration of glandular tissue with a solution of saline and epinephrine—has proved helpful. The defect is closed by reapproximating the subcutaneously mobilized lobules using absorbable sutures ( Fig.10.4 ). A certain amount of flattening and indentation of the breast contour is nevertheless unavoidable. It is important to reapproximate the mobilized fatty tissue to avoid the development of indented scars. Mobilizing the parenchymal tissue off the pectoralis muscle over a short distance can sometimes help correct indentation.
Dissection should not be too wide, as this can lead to parenchymal necrosis. Creation of a rotation flap—e. g., using the breast tissue projecting into the axillary region to fill in defects in the superolateral quadrant—has also not proved useful. Allowing the defect to fill with wound discharge and blood is an unsuitable method of correction.
The subcutaneous glandular advancement flap can be used in about two-thirds of the patients ( Figs.10.5–10.7 ). In the remaining third, other plastic surgery techniques have to be used—either as skin resection measures to lift the breast, or with a flap transfer to replace volume (see below).
Axillary Lymph-Node Dissection
Axillary lymph-node dissection still forms part of the standard treatment for breast cancer, as a way of obtaining information regarding the patient’s lymph-node status. The trend toward increasingly less invasive measures in breast surgery has led to debates regarding the need for axillary dissection. Although there are many factors involving tumor biology that would on their own represent indications for adjuvant systemic therapy, assessment of the axillary lymph-node status is still required as the most significant prognostic parameter.
In an effort to reduce the surgery-related morbidity involving the axilla, sentinel lymph-node dissection has now become the standard of care. For oncological or logistical reasons, however, this is not always feasible in all patients. Surgeons therefore also need to be proficient in “classic” axillary dissection.
According to the international guidelines, level I and II lymph nodes should be removed. Level I means the lymph nodes that lie lateral to the pectoralis minor, and level II refers to the nodes below the pectoralis minor. The nodes lying medial to the pectoralis minor and the infraclavicular lymph nodes are known as level III nodes ( Fig.10.9 ).
During resection, care needs to be taken to avoid dissecting the superior portion of the axillary vein, so as to avoid impaired lymph drainage along the cephalic vein route. Level III nodes are also spared in order to avoid lymphedema in the arm, and individual level III nodes are only dissected if there is suspected involvement. After axillary dissection has been completed, the interpectoral space is also checked digitally. Additional lymph nodes are only removed from this area if they appear abnormal.
In principle, the extent of axillary dissection should correspond to the extent of disease. If the tumor is small and lymph-node involvement is unlikely, dissection can be carried out conservatively. A more aggressive approach should be used for palpable nodes. Ten lymph nodes should be removed in a standard axillary dissection.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree