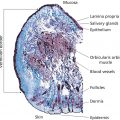
Jasmine C. Hollinger, Chesahna Kindred, and Rebat M. Halder Howard University College of Medicine, Washington, DC, USA The demographics of the United States reflect a dynamic mixture of people of various ethnic and racial groups. According to the 2010 census, just over one‐third of the US population reported their ethnicity and race as something other than non‐Hispanic white [1]. Projections of the size and composition of the US Population: 2014 to 2060 noted that by the year 2044, more than half of all Americans will belong to a minority group [2]. Persons of skin of color include Africans, African‐Americans, Afro‐Caribbeans, Asians, Latinos (Hispanics), Native Americans, Middle Easterners, Alaskan natives, pacific islanders, native Hawaiians, and Mediterraneans. The term “black” as in black skin refers to individuals with African ancestry, including Africans, African‐Americans, and Afro‐Caribbeans. Subgroups exist within each ethnoracial group. The differences in the structure, function, and physiology of the hair and skin in individuals of skin of color are important in understanding the structural and physiologic variations that exist and influence disease presentations. Pigmentation is especially important in patients of skin of color because pigmentary disorder is the most common reason for a visit to a dermatologist in this group [3]. Melanin, the major determinant of skin color, absorbs UV light and blocks free radical generation, protecting the skin from sun damage and aging. Melanocytes, the cells that produce melanin, synthesize melanin in special organelles, melanosomes. Melanin‐filled melanosomes are transferred from one melanocyte to 30–35 adjacent keratinocytes in the basal layer [4]. The number of melanocytes also decreases with age. There is more than one type of melanin: eumelanin, a dark brown–black pigment; and pheomelanin, a yellow–reddish pigment. Eumelanin is deposited in ellipsoidal melanosomes which contain a fibrillar internal structure. Synthesis of eumelanin increases after UV exposure (tanning). Pheomelanin has a higher sulfur content than eumelanin because of the sulfur‐containing amino acid cysteine. Pheomelanin is synthesized in spherical melanosomes and is associated with microvesicles [5]. Although not obvious to the naked eye, most melanin pigments of the hair, skin and, eyes are combinations of eumelanin and pheomelanin [6]. It is generally believed that genetics determine the constitutive levels of pheomelanin and eumelanin. Eumelanin is more important in determining the degree of pigmentation than pheomelanin. Eumelanin, and not pheomelanin, increases with visual pigmentation [6]. Lighter melanocytes have higher pheomelanin content than dark melanocytes. In one study [6], white persons had the least amount of eumelanin, Asian Indians had more, and African‐Americans had the highest. Of note, adult melanocytes contain significantly more pheomelanin than cultured neonatal melanocytes. Melanosomes also differ among different races. In black persons, they are mostly in the basal layer, but those of white persons are mostly in the stratum corneum. This is evident in the site of UV filtration: the basal and spinous layers in blacks and the stratum corneum in white persons. Of note, the epidermis of black skin rarely shows atrophied areas [7]. In black skin, melanocytes contain more than 200 melanosomes. The melanosomes are 0.5–0.8 mm in diameter, do not have a limiting membrane, are stuck closely together, and are individually distributed throughout the epidermis. In white skin, the melanocytes contain less than 20 melanosomes. The melanosomes are 0.3–0.5 mm in diameter, associated with a limiting membrane, and distributed in clusters with spaces between them. The melanosomes of lighter skin degrade faster than that of dark skin. As a result, there is less melanin content in the upper layers of the stratum corneum. Thus, the melanocytes in black skin are larger, more active in making melanin, and the melanosomes are packaged, distributed, and broken down differently than in white skin. Melanosome pH is also another factor that plays a role in regulating differences in skin color. Melanosomes derived from darker skin have a more neutral pH compared to those from lighter skin where the pH is more acidic. Tyrosinase activity is enhanced at neutral pH, thus those with lighter skin have lower tyrosinase activity [8]. There is also a difference in melanosomes between individuals within the same race with varying degrees of pigmentation. Despite greater melanin content in darker skins, there is no evidence of major differences in the number of melanocytes [9]. Also, dark Caucasian skin resembles the melanosome distribution observed in black skin [10]. Black persons with dark skin have large, nonaggregated melanosomes and those with lighter skin have a combination of large nonaggregated and smaller aggregated melanosomes [11]. White persons with darker skin have nonaggregated melanosomes when exposed to sunlight and white persons with lighter skin have aggregated melanosomes when not exposed to sunlight [9, 10, 12]. It has also been shown that the number of melanosomes transferred to keratinocytes is significantly higher in skin of African descent versus white skin [13]. The steps of melanogenesis are as follows. The enzyme tyrosinase hydroxylates tyrosine to dihydroxyphenylalanine (DOPA) and oxidizes DOPA to dopaquinone. Dopaquinone then undergoes one of two pathways. If dopaquinone binds to cysteine, the oxidation of cysteinyldopa produces pheomelanin. In the absence of cysteine, dopaquinone spontaneously converts to dopachrome. Dopachrome is then decarboxylated or tautomerized to eventually yield eumelanin. Melanosomal P‐protein is involved in the acidification of the melanosome in melanogenesis [14]. Finally, the tyrosinase activity (not simply the amount of the tyrosinase protein) and cysteine concentration determine the eumelanin–pheomelanin content [6]. Tyrosinase and tyrosinase‐related proteins 1 and 2 (TRP‐1 and TRP‐2) are upregulated when α‐melanocyte‐stimulating hormone (α‐MSH) or adrenocorticotropin binds to melanocortin‐1 receptor (MC1R), a transmembrane receptor located on melanocytes [14–17]. The MC1R loss‐of‐function mutation increases sensitivity to UV‐induced DNA damage. Gene expression of tyrosinase is similar between black and white persons despite tyrosinase activity being significantly higher in darker versus lighter skin, but other related genes are expressed differently. The expression of RAB27A, encoding for the melanosome transport molecule, plays an important role in melanocyte melanin content as evident in Griscelli syndrome. In a study by Yoshida‐Amano et al., darkly pigmented melanocytes were found to have a substantially higher RAB27A expression and thus able to transfer more to keratinocytes. It was concluded that RAB27A is essential in determining ethnic skin color differences [13]. The MSH cell surface receptor gene for melanosomal P‐protein is expressed differently between races. This gene may regulate tyrosinase, TRP‐1, and TRP‐2 [6]. In addition to the MC1R, protease‐activated receptor 2 (PAR‐2) is another important receptor that regulates epidermal cells and affects pigmentation [18]. PAR‐2 is expressed on many cells and several different organs. Accordingly, the receptor is involved in several physiologic processes, including growth and development, mitogenesis, injury responses, and cutaneous pigmentation. In the skin, PAR‐2 is expressed in the keratinocytes of the basal, spinous, and granular layers of the epidermis, endothelial cells, hair follicles, myoepithelial cells of sweat glands, and dermal dendritic‐like cells [19, 20]. PAR‐2 is a seven‐transmembrane domain G‐protein‐coupled receptor which undergoes activation via proteolytic cleavage of the NH2 terminus which acts as a tethered ligand which then activates the receptor (autoactivation). Protease‐activated receptor 2‐activating protease (PAR‐2‐AP), endothelial cell‐released trypsin, mast cell‐released trypsin and chymase, and SLIGKV (Ser‐Leu‐Ile‐Gly‐Lys‐Val) all irreversibly activate PAR‐2 while serine protease inhibitors interfere with the activation of the receptor [21–23]. SLIGKV and trypsin activate PAR‐2 to use a Rho‐dependent signaling pathway to induce melanosomal phagocytosis by keratinocytes. The result is an increase in pigmentation to the same degree as UV radiation [20–24]. Serine proteases are regulatory proteins involved in tumor growth, inflammation, tissue repair, and apoptosis in various tissues [19]. In the skin, serine protease inhibitors prevent the keratinocytes from phagocytosing melanosomes from the presenting dendritic tip of the melanocyte. This leads to a dose‐dependent depigmentation without irritation or adverse events. PAR‐2 also has a pro‐inflammatory effect in the skin [20]. The activation of PAR‐2 expressed on endothelial cells by tryptase, trypsin, or PAR‐2‐AP leads to an increase in proinflammatory cytokines interleukin 6 (IL‐6) and IL‐8 and also stimulates NF‐κB, an intracellular proinflammatory regulator [21]. Mast cells interact with endothelial cells to regulate inflammatory responses, angiogenesis, and wound healing, and PAR‐2 has a regulatory role in this cell–cell interaction [20, 21]. UV irradiation of keratinocytes induces pigmentation in several ways: upregulation of melanogenic enzymes, DNA damage that induces melanogenesis, increased melanosome transfer to keratinocytes, and increased melanocyte dendricity. UV radiation (UVR) increases the secretion of proteases by keratinocytes in a dose‐dependent manner. Specifically, UVR directly increases the expression of PAR‐2 de novo, upregulates proteases that activate PAR‐2, and activates dermal mast cell degranulation [24]. According to the literature, PAR‐2 expression is different in skin of color compared to white skin thus, suggesting the involvement of PAR‐2 in ethnic skin color phenotypes. One study demonstrated that PAR‐2 and its activator trypsin are expressed in higher levels in darker skin. PAR‐2 was also found to have higher cleavage ability in highly pigmented skin [25]. Another study did find differences in skin phototypes I, II, and III [24]. UVR increases the expression of PAR‐2 in the skin and activated PAR‐2 stimulates pigmentation. This study found that the response of PAR‐2 to UVR is an important determinant of one’s ability to tan. In the nonirradiated skin, PAR‐2 expression was confined to the basal layer and just above the basal layer. Irradiated skin showed de novo PAR‐2 expression in the entire epidermis or upper two‐thirds of the epidermis. Skin phototype I had a delayed upregulation of PAR‐2 expression compared to phototypes II and III. After cutaneous trauma or inflammation, melanocytes can react with normal, increased, or decreased melanin production; all of which are normal biologic responses. Increased and decreased production results in postinflammatory hyperpigmentation (PIH) or hypopigmentation. PIH is an increase in melanin production and/or an abnormal distribution of melanin resulting from inflammatory cutaneous disorders or irritation from topical medications [26, 27]. Examples include acne, allergic contact dermatitis, lichen planus, bullous pemphigoid, herpes zoster, and treatment with topical retinoids. Often, the PIH resulting from acne is more distressing to darker‐skinned individuals than the initial acute lesion. The color of the hyperpigmentation in PIH depends on the location of the melanin. Melanin in the epidermis appears brown, while melanin in the dermis appears blue–gray. Wood’s lamp examination distinguishes the location of the melanin: the epidermal component is enhanced and the dermal component becomes unapparent [27]. Postinflammatory hypopigmentation shares the same triggers as PIH but instead results from decreased melanin production with clinically apparent light areas [26]. The Wood’s lamp examination does not accentuate hypopigmentation in postinflammatory hypopigmentation; it is useful for depigmented disorders such as vitiligo and piebaldism. The pathogenesis of PIH and postinflammatory hypopigmentation are unknown. It is likely that an inflammatory process in the skin stimulates keratinocytes, melanocytes, and inflammatory cells to release cytokines and inflammatory mediators that lead to the hyperpigmentation or hypopigmentation. The cytokines and inflammatory mediators include leukotriene (LT), prostaglandins (PG), and thromboxane (TXB) [28]. Specifically for PIH, in vitro studies revealed that LT‐C4, LT‐D4, PG‐E2, and TXB‐2 stimulate human melanocyte enlargement and dendrocyte proliferation. LT‐C4 also increases tyrosinase activity and mitogenic activity of melanocytes. Transforming growth factor‐α and LT‐C4 stimulate movement of melanocytes. The basal layer can also be damaged due to inflammation which results in leakage of melanin from keratinocytes and thus accumulation of melanophages in the dermis exacerbating dermal hyperpigmentation [29]. In postinflammatory hypopigmentation, the pathogenesis likely involves inflammatory mediators inducing melanocyte cell‐surface expression of intercellular adhesion molecule 1 (ICAM‐1) which may lead to leukocyte–melanocyte attachments that inadvertently destroy melanocytes. These inflammatory mediators include interferon‐gamma, tumor necrosis factor α (TNF‐α), TNF‐β, IL‐6, and IL‐7. It is clear that those who fall within Fitzpatrick skin phototypes IV–VI are less susceptible to photoaging; this is most likely due to the photoprotective role of melanin [29, 30]. The epidermis of black skin has a protective factor (PF) for UVB of 13.4 and that of white skin is 3.4 [31]. The mean UVB transmission by black epidermis is 5.7% compared to 29.4% for white epidermis. The PF for UVA in black epidermis is 5.7 and in white epidermis is 1.8 [31]. The mean UVA transmission by black epidermis is 17.5% and 55.5% for white epidermis. Hence, 3–4 times more UVA reaches the upper dermis of white persons than that of black persons. The main site of UV filtration in white skin is the stratum corneum, whereas in black skin it is the basal layer [31]. The Malpighian layer of black skin removes twice as much UVB radiation as the stratum corneum [32]. It is possible that even greater removal of UVA occurs in black skin basal layers [32]. While the above characteristics of natural sun PF were studied in black skin, they can probably be extrapolated to most persons of skin phototypes IV–VI. The epidermal layer of skin is made up of five different layers: stratum basale, stratum spinosum, stratum granulosum, stratum lucidum, and stratum corneum. The stratum basale (also termed the basal layer) is the germinative layer of the epidermis. The time required for a cell to transition from the basal layer through the other epidermal layers to the stratum corneum is 24–40 days. The morphology and structure of the epidermis are very similar among different races, although a few differences do exist. The stratum corneum, the most superficial layer, is the layer responsible for preventing water loss and providing mechanical protection. The cells of the stratum corneum, the corneocytes, are flat cells measuring 50 μm across and 1 μm thick. The corneocytes are arranged in layers; the number of layers varies with anatomic site and race. There are no differences between races in corneocyte surface area, which has a mean size of 900 μm [3, 33]. The stratum corneum of black skin is more compact than that of white skin. While the mean thickness of the stratum corneum is the same in black and white skin, black skin contains 20 cell layers while white skin contains 16. The answer to whether or not there are racial differences in spontaneous desquamation is inconclusive [32–34]. It was observed in a study by Wesley and Maibach [35] that blacks have a 2.5 times greater spontaneous desquamation compared with whites and Asians [36]. Parameters for skin barrier function (stratum corneum hydration, sebum secretion, erythema, and laser Doppler flowmetry) are similar, even after an objective epicutaneous test with sodium lauryl sulfate [35]. Transepidermal water loss (TEWL) is the amount of water vapor loss from the skin, excluding sweat. TEWL increases with the temperature of the skin. Concrete evidence regarding the difference in TEWL between different races has yet to be established. In most studies, TEWL has been found to be greater in black skin compared to white skin but the opposite has also been reported. A study reported no difference in TEWL amongst blacks, whites, and Hispanics [36]. In a more recent study, basal TEWL was evaluated using an Aquaflux AF200 on 30 predefined regions of the face in sixteen South African females of different ethnicities (Indian, Black African, Chinese, and Caucasian). Baseline data were also measured on the volar forearms and dorsal hands. The authors found that TEWL was statistically significantly lower on the volar forearm for the Caucasian patients in comparison to Black African and Indian patients and, on the hands only, lower compared to the Indians. Facial TEWL was found to be similar between the Caucasian and Black African groups with both being lower than the Indian group [37]. Aside from TEWL, hydration is also a characteristic of skin. One of the ways to measure hydration, or water content, is conductance. Conductance, the opposite of resistance, is increased in hydrated skin because hydrated skin is more sensitive to the electrical field [38]. Skin conductance is higher in black persons and Hispanics than white persons [38]. Lipid content in black skin is higher than that of white skin [39]. However, black skin is more prone to dryness, suggesting that a difference in lipid content has a role. This includes the ratio of ceramide : cholesterol : fatty acids, the type of ceramides, and the type of sphingosine backbone. The total levels of ceramides were approximately 50% lower in the stratum corneum of blacks when compared to whites and Hispanics according to a study [40]. One study suggests that the degree of pigmentation influences lipid differences [41]. Pigmentation affects skin dryness. Skin dryness is greater on sun‐exposed (dorsal arm) sites for lighter skin, such as Caucasian and Chinese skin, than sites that are primarily out of the sun (ventral arm) [42]
CHAPTER 3
Pigmentation and Skin of Color
Introduction
Melanocytes
Dyspigmentation
Natural sun protective factor in skin of color
Skin of color
Epidermis
Stratum corneum
Transepidermal water loss
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree