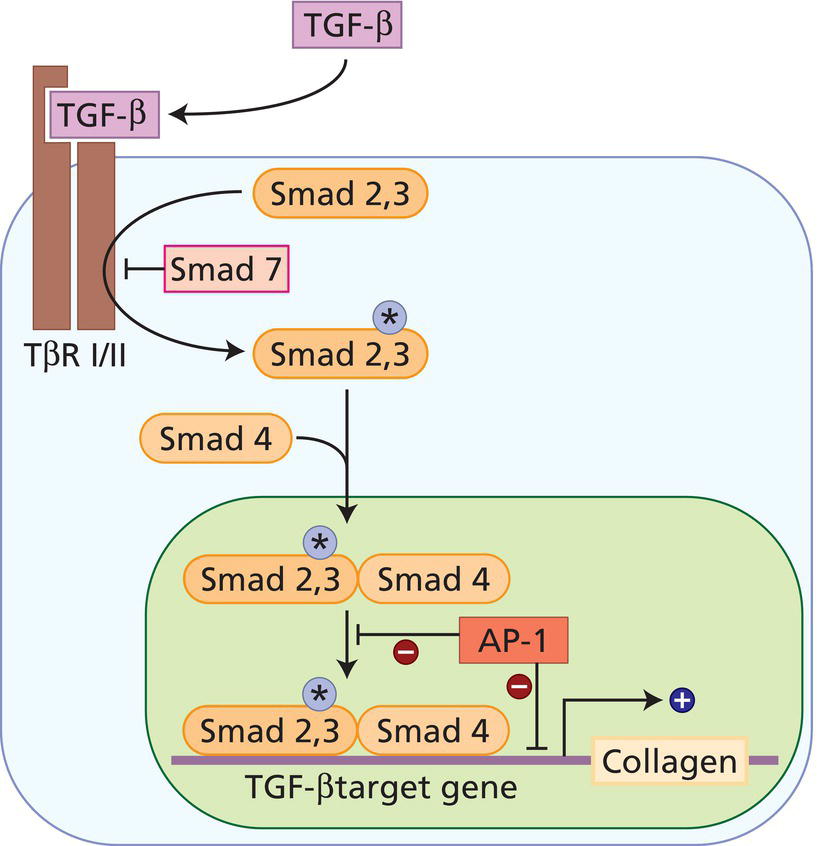
Kalee Shah1, Kira Minkis1, Jillian Havey Swary2, and Murad Alam2 1 Weill Cornell Medical College, Cornell University, New York, NY, USA 2 Feinberg School of Medicine, Northwestern University, Chicago, IL, USA Skin, the largest human organ, is chronically exposed to UV radiation from the sun. The skin is at the frontline of defense for the human body against the harmful effects of UV exposure. Chronic absorption of UV radiation leads to photoaging, sunburn, immunosuppression, and carcinogenesis. Photoaging is the most common form of skin damage caused by UV exposure, affecting connective tissue, melanocytes, and the microvasculature [1]. Recent advances in understanding photoaging in human skin have identified the physical manifestations, histologic characteristics, and molecular mechanisms of UV‐induced skin damage. Photoaging describes the clinical, histologic, and functional changes that occur in the skin as a result of long‐term exposure to UV radiation. Chronic UV exposure results in premature or accelerated skin aging, which is marked clinically by fine and coarse wrinkling of the skin, dyspigmentation, sallow color, textural changes, loss of elasticity, and premalignant actinic keratoses. Most of these clinical signs are caused by dermal alterations. Pigmentary disorders such as seborrheic keratoses, lentigines, and diffuse hyperpigmentation are characteristic of epidermal changes [2]. The clinical features of photoaged skin are superimposed on changes seen with intrinsic chronological aging of the skin. These clinical characteristics are confirmed histologically by epidermal thinning and disorganization of the dermal connective tissue. Solar elastosis, caused by the accumulation of disorganized connective tissue elastin, is a characteristic histologic finding of photoaged skin [3]. Similar alterations seen in the cellular component and the extracellular matrix of connective tissue may affect superficial capillaries, causing surface telangiectasias clinically [4]. The significance of photoaging lies in both the cosmetic and medical repercussions – in the demand for agents that can prevent or reverse the cutaneous signs associated with photoaging and its strong association with cutaneous malignancies. In 2017, the global anti‐aging product market was estimated to be US$324.6 billion and is expected to reach US$429 billion by 2022 [5]. Furthermore, UV‐induced cutaneous malignancies, namely basal cell carcinoma and squamous cell carcinoma, are the most common malignancies diagnosed in the United States, with approximately 5.4 million cases diagnosed annually [6]. Skin ages over time like all other organs. Skin aging can be subdivided into intrinsic and extrinsic aging. Intrinsic aging is a hallmark of human chronological aging and occurs in both sun‐exposed and sun‐protected skin. Extrinsic aging, on the contrary, is affected by exposure to environmental factors such as UV radiation. While sun‐protected chronologically aged skin and photoaged chronologically aged skin share common characteristics, many of the physical qualities of skin that decline with age show an accelerated decline with photoaging [7]. Chronologically aged skin is characterized by dryness, fine wrinkles, skin atrophy, homogeneous pigmentation, and seborrheic keratoses [8]. Extrinsically aged skin, on the contrary, is characterized by roughness, dryness, fine as well as coarse wrinkles, atrophy, uneven pigmentation, and superficial vascular abnormalities (telangiectasias) [8]. It is important to note that these attributes are not absolute and can vary according to Fitzpatrick skin type classification and history of sun exposure. While the pathophysiology of photoaged and chronologically aged skin overlap, the histologic features of these two entities are distinct. In chronologically aged skin, the epidermis is thin with an intact stratum corneum, the dermoepidermal junction and the dermis are flattened, and dermal fibroblasts produce less collagen. In photoaged skin, the thickness of the epidermis can either increase or decrease, corresponding to areas of keratinocyte atypia. The dermoepidermal junction is atrophied in appearance and the basement membrane thickness is increased, reflecting basal keratinocyte damage. Changes in the dermis of photoaged skin can vary based on the amount of acquired UV damage. Solar elastosis is the most prominent histologic feature of photoaged skin. While the quantity of elastin in the dermis decreases in chronologically aged skin, it increases in proportion to the amount of UV exposure in photoaged skin [9, 10]. Accumulated elastic fibers occupy areas in the dermal compartment previously inhabited by collagen fibers [11]. This altered elastin deposition, or solar elastosis, is seen clinically as wrinkles and yellow discoloration of the skin. Another feature of photoaged skin is collagen fibril disorganization. Mature collagen fibers, which constitute the bulk of the skin’s connective tissue, are replaced by collagen with a basophilic appearance, termed basophilic degeneration. Additional histologic characteristics of photoaged skin include an increase in the deposition of glycosaminoglycans and dermal extracellular matrix proteins [12, 13]. In fact, the overall cell population in photodamaged skin increases, leading to hyperplastic fibroblast proliferation and infiltration of inflammatory substrates that cause chronic inflammation, or heliodermatitis [14]. Changes in the microvasculature also occur, as is clinically manifested in surface telangiectasias and other vascular abnormalities. Cutaneous vasculature in chronologically aged skin and photoaged skin share similar characteristics, such as decreased cutaneous temperature, pallor, decreased cutaneous vessel size, reduced erythema, reduced cutaneous nutritional supply, and reduced cutaneous vascular responsiveness [15–17]. However, there are also significant differences in the microvasculature. Studies have reported that the blood vessels in photoaged skin are obliterated and the overall horizontal architecture of the vascular plexuses is disrupted [18]. In contrast to photodamaged skin, chronologically aged skin does not display a greatly disturbed pattern of horizontal vasculature. In addition, while cutaneous vessel size has been reported to decrease with age in both, only photoaged skin exhibits a large reduction in the number of dermal vessels. This reduction is especially highlighted in the upper dermal connective tissue, where it is hypothesized that the chronic UV‐induced degradation of elastic and collagen fibers limits the ability to provide the physical support required for normal cutaneous vessel maintenance [15]. The effects to skin vasculature may differ between acute and chronic UV exposure. Recent studies have implied that a single exposure to UVB radiation induces skin angiogenesis in human skin in vivo [19, 20]. The epidermis‐derived vascular endothelial growth factor (VEGF) is an angiogenic factor that is significantly upregulated with UV exposure in keratinocytes in vitro and in human skin in vivo. Chung and Eun [15] demonstrated that epidermal VEGF expression increased significantly on days 2 and 3 post‐UV‐irradiation compared to non‐UV‐irradiated control skin, consequently inducing cutaneous angiogenesis. Therefore, acute UV exposure induces angiogenesis. In contrast, chronic UV‐exposed photodamaged skin is known to have a significant reduction in the number of cutaneous blood vessels. The reason for this discrepancy is still under investigation. All races are susceptible to photoaging. However, people with Fitzpatrick skin phototypes IV–VI are less susceptible to the deleterious effects of UV irradiation than people with a lower Fitzpatrick skin type classification. This phenomenon is most likely a result of the protective role of melanin [21]. Studies reporting characteristics of photoaging in ethnic skin are limited and findings are briefly highlighted. Hopefully, as new scales for assessing photoaging in ethnic skin are established and validated, research in this area will increase [22]. Kaidbey et al. [23] compared UV absorption of African‐American skin with Caucasian skin. It is known that only 10% of the total UVB rays penetrates the dermis. However, the mean UVB transmission into the dermis of African‐American skin was found to be significantly less than in Caucasian dermis (5.7% vs. 29.4%, respectively). Similar experiments were performed with UVA irradiation. UVA transmission into African‐American dermis was 17.5% compared to 55% for Caucasian epidermis [23]. The physiologic reason behind this difference in black and white skin lies at the site of UV filtration. The malpighian layer (basal cell layer) of African‐American skin is the main site of UV filtration, while the stratum corneum absorbs most UV rays in Caucasian skin. The malpighian layer of African‐American skin removes twice as much UVB radiation as the overlying stratum corneum, thus mitigating the deleterious effects of UV rays in the underlying dermis [24]. Langton et al. [25] characterized the biomechanical and histologic characteristics of photoaging in African‐American skin. Skin exposed to chronic UV demonstrated reduced biomechanical properties, such as elasticity, over and above that observed in chronologically aged skin. Histology of skin samples showed that photoaging resulted in complete flattening of the dermoepidermal junction, disruption of elastin fiber organization, and remodeling of the collagen fibrillar matrix. Notably, solar elastosis, a characteristic histologic finding of photoaging in fair skin‐types, was not detected [25]. In African‐Americans, photoaging may not be clinically apparent until the fifth or sixth decade of life and is more common in individuals with a lighter complexion [26]. The features of photoaging in this ethnic skin group manifest as signs of laxity in the malar fat pads sagging toward the nasolabial folds, as well as dermatosis papulosa nigra [27, 28]. In patients of Hispanic and European descent, photoaging occurs in the same frequency as Caucasians and clinical signs are primarily wrinkling rather than pigmentary alterations. On the contrary, skin of East and South‐East Asian patients mainly exhibits pigmentary alterations (seborrheic keratoses, hyperpigmentation, actinic lentigines, sun‐induced melasma) as a result of photoaging. Wrinkling is minimal and occurs later in life [28–30]. Finally, very few studies have reported on the signs of photoaging in South Asian (Pakistani, Indian) skin. UV‐induced hyperpigmentation, dermatosis papulosa nigra, and seborrheic keratosis are noted [31]. It is important to note that the number of melanocytes per unit area of skin does not vary across ethnicities. Instead, it is the relative amount of melanin packaged into melanocytes that accounts for the pigmentation differences between Caucasian skin and ethnic skin [32]. Increasing age leads to senescence of melanocytes; senescent melanocytes, in turn, can cause greater melanin production that has been observed in some darker‐skinned individuals [33]. This may be responsible for the general “bronzing,” and darkening, appearing as a “permanent tan” observed in some photoaged individuals of darker skin tones. A recent study, however, observed that pigmentation decreased with age in sun‐exposed sites compared to sun‐protected sites in African Americans. This pattern was opposite in Caucasian study participants, where sun‐exposed sites were darker than sun‐protected sites [34]. Genetics may also play a role in photoaging. A recent meta‐analysis of five genome‐wide association studies from three different cohorts identified genetic alleles that may affect the severity of skin aging. The authors found that single‐nucleotide polymorphisms (SNP) near the SLC45A2, IRF4, and MC1R genes were significantly associated with wrinkling and photoaging. Interestingly, the lower‐pigmentation alleles of each gene were associated with more severe photoaging mirroring the established association between low‐pigmentation alleles and the increased risk of melanoma and keratinocyte carcinomas [35]. Substantial progress has been made to ascertain the molecular mechanisms accountable for photoaging in human skin. UV irradiation damages human skin by at least three interdependent mechanisms: Gene expression profiles from Caucasian females ranging in age from 20 to nearly 75 years old revealed age‐induced and photoinduced changes in pathways related to oxidative stress, senescence, metabolism, and barrier function. Molecular patterns of gene expression in women that were younger appearing were similar to women that were actually younger [36]. The prominent molecular processes of photoaging are described in detail below. Before these processes are highlighted, however, it is important to consider the biology of UV radiation as well as the structure and function of collagen, which plays a key role in the strength and integrity of the skin. Photobiology The UV spectrum is further categorized into three subtypes: UVC (270–290 nanometers [nm]), UVB (290–320 nm), and UVA (320–400 nm). UVC radiation is filtered by the ozone layer and atmospheric moisture, and consequently never reaches the Earth. In contrast, UVA and UVB rays do reach the terrestrial surface; the ratio of UVA to UVB rays is 20:1 [37] and UVB is greatest during the summer months. Both forms of radiation have acute and chronic effects on human skin. In order to exert biologic effects on human skin, both categories of UV rays must be absorbed by chromophores in the skin. UV light interacts with different skin cells at different depths depending on the wavelength absorbed (Figure 2.1). More specifically, energy from UVB rays is mostly absorbed by the epidermis and affects epidermal cells such as the keratinocytes, whereas energy from UVA penetrates deeper into the skin, affecting both epidermal keratinocytes and the deeper dermal fibroblasts. Approximately 50% of UVA penetrates the skin in a fair‐skinned individual (versus <10% of UVB photons). The absorbed energy is converted into varying chemical reactions that cause histologic and clinical changes in the skin. UVA absorption by chromophores mostly acts indirectly by transferring energy to oxygen to generate ROS, leading to transcription factor activation, lipid peroxidation, and DNA‐strand breaks. On the contrary, UVB has a more direct effect on the absorbing chromophores by damaging DNA via cross‐linking of adjacent DNA pyrimidines, among other mechanisms [39]. Up to 50% of UV‐induced photodamage is from the formation of free radicals, while mechanisms such as direct cellular injury account for the remainder of UV effects [40]. Thus UVB induced photodamage is implicated as the predominant cause of photoaging. The important role of UVA in photoaging, however, stems from the fact that in distinction to UVB, UVA is also transmitted through glass. This enables exposure while indoors, near windows, as well as while driving leading to significant long‐term exposure. Evidence for this includes dramatic unilateral dermatoheliosis in some long‐term occupational drivers [41]. Figure 2.1 Ultraviolet light interacts with different skin cells at different depths. More specifically, energy from UVB rays is mostly absorbed by the epidermis and affects epidermal cells such as the keratinocytes. Energy from UVA rays affects both epidermal keratinocytes and the deeper dermal fibroblasts. AP‐1, activator protein 1; NF‐κB, nuclear factor κB; MMP, matrix metalloproteinase; mtDNA, mitochondrial DNA; ROS, reactive oxygen species. (Source: Berneburg et al., 2000 [38]. Reproduced with permission of John Wiley & Sons.) The unique physical characteristics of collagen fibers are essential for providing strength, structural integrity, and resilience to the skin. Type I collagen accounts for greater than 90% of the protein in the human skin, with type III collagen accounting for a smaller fraction (10%). Dermal fibroblasts synthesize individual collagen polypeptide chains as precursor molecules called procollagen. These procollagen building blocks are assembled into larger collagen fibers through enzymatic cross‐linking and form the three‐dimensional dermal network. This intermolecular covalent cross‐linking step is essential for maintenance and structural integrity of large collagen fibers, especially type I collagen. Collagen gene expression in human skin fibroblasts is regulated by the cytokine transforming growth factor β (TGF‐β) and the transcription factor activator protein (AP‐1). When TGF‐βs bind to its cell surface receptors (TβRI and TβRII), transcription factors Smad2 and Smad3 are activated, combine with Smad4, and enter the nucleus, where they regulate type I procollagen production. AP‐1 has an opposing effect and inhibits collagen gene transcription by either direct suppression of gene transcription or obstructing the Smad complex from binding to the TGF‐β target gene (Figure 2.2) [42]. Therefore, in the absence of any inhibiting factors, the TGF‐β/Smad signaling pathway results in a net increase in procollagen production. Figure 2.2 The regulation of procollagen production: the TGF‐β/Smad signaling pathway. AP‐1, activator protein 1; TβR, TGF‐β receptor; TGF‐β, transforming growth factor β. (Source: Kang et al., 2001 [3]. Reproduced with permission of Elsevier.) The natural breakdown of type I collagen is a slow process and occurs through enzymatic degradation [43]. Dermal collagen has a half‐life of greater than 1 year [43]. This slow rate of type I collagen turnover allows for disorganization and fragmentation of collagen that impair its functions. Fragmentation and dispersion of collagen fibers are features of photodamaged skin. UV irradiation stimulates photoaging through several molecular mechanisms, discussed in detail below. DNA damage and defective DNA repair mechanisms have been implicated in carcinogenesis as well as the intrinsic aging process [44]
CHAPTER 2
Photoaging
Introduction
Definition
Clinical features
Physical characteristics of photoaged versus chronologically aged skin
Histology of photoaged versus chronologically aged skin
Photoaging in ethnic skin
Genetics of photoaging
Molecular mechanisms of photoaging
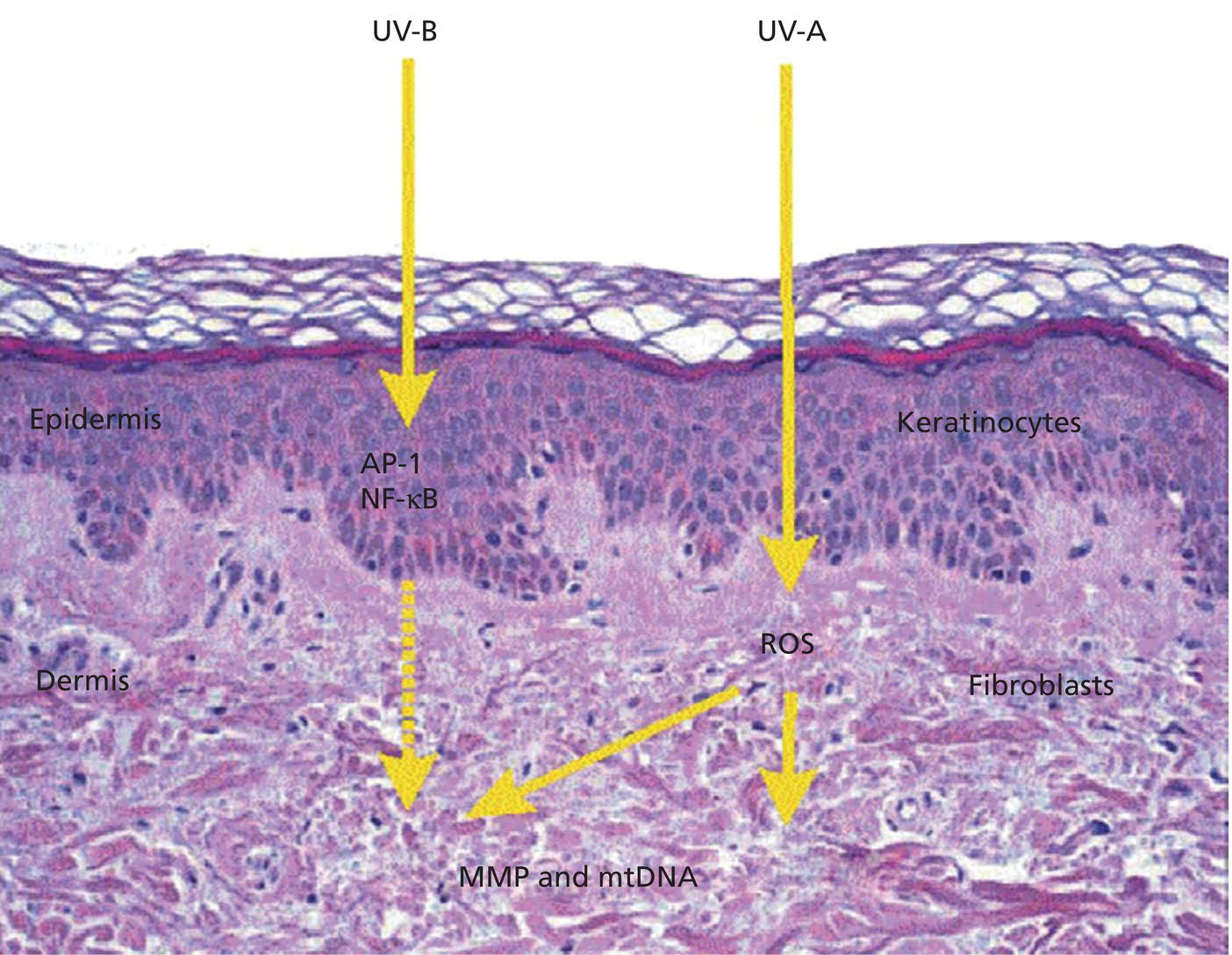
Collagen
How does UV irradiation stimulate photoaging?
UVB leads to direct DNA damage
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree