Parameters for assessing stability of vitiligo
Consensus recommendation of IADVLa taskforce on stability [5]
History of progression
The absence of new lesions
No new lesions
Extension of old lesions
No extension of old lesions
No progression of existing lesions
Koebner phenomenon
The absence of Koebner phenomenon either based on history or by checking for experimentally induced vitiligo
The absence of Koebner phenomenon during the past 1 year
Spontaneous repigmentation
Should be considered as a favorable sign for the transplantation procedure
Minigrafting test or test grafting
The original test was proposed by Falabella et al. to select patients with stable vitiligo who may respond to melanocyte transplantation [4]. The test is considered positive if unequivocal repigmentation took place beyond 1 mm from the border of the implanted graft over a period of 3 months
May be considered whenever there is a doubt about the stability, or the patient is unable to give a clear history on stability
28.2.2 Patient Selection Criteria (Table 28.2)
Table 28.2
Patient selection criteria for vitiligo surgery
1. | Patients not responding to medical treatment |
2. | Patients with stable lesions of vitiligo vulgaris, localized or segmental vitiligo lesions, though it may be done for all types of vitiligo (including segmental, generalized, and acro-facial types) Test graft should be always used to confirm the stability of vitiligo in case of a doubt or inconclusive history |
3. | Patients with realistic expectations and who are psychologically stable |
28.2.2.1 Patient Age
No current guidelines exist regarding the age group selection for vitiligo surgery. Children may be difficult candidates. The procedure generally performed under local anesthesia may be not tolerated by this younger age group. On the other hand, surgery done under general anesthesia poses unacceptable risk. Moreover, progress of the vitiligo is hard to predict in children. Hence, it has been suggested that surgical procedures should not be performed in children. However, in various studies, it has been found that results of the transplantation procedures yielded better results in younger individuals than in older ones [6, 7]. Thus no consensus exists in this aspect, and physicians should exercise their judgment after taking all aspects of the individual patient into consideration.
28.2.3 Pre-procedure Preparation
28.2.3.1 Preoperative Counseling and Informed Consent [5]
The nature of the disease, procedure, expected outcome, and possible complications after the procedure must be explained to the patient. The need for concomitant medical therapy should be emphasized. Patients should understand that proper results may take time to appear (few months to more than a year). They should be provided with adequate opportunity to seek information through brochures, computer presentations, and one-on-one discussions.
The consent form should specifically state the limitations of the procedure, possible future disease progression, and whether more procedures will be needed for optimal outcome. A detailed consent form describing the procedure and possible complications should be signed by the patient.
28.2.3.2 Preoperative Investigations (Based on the Procedure)
Complete blood counts, coagulation profile platelet count, blood sugar, screening for hepatitis B, VDRL, HIV (ELISA), routine urinalysis, serum creatinine, blood urea nitrogen, SGOT, SGPT (if large areas are going to be phenolized), and ECG may be needed in elderly patients.
28.2.3.3 Anesthesia [5]
The recipient site is locally anesthetized by infiltration of 2 % lidocaine HCl (Xylocaine), the pain of which can be reduced by prior application of topical combination anesthetic lidocaine 2.5 %/prilocaine 2.5 % (EMLA® cream) under occlusion for 1–2 h. Adrenaline should not be used on the recipient site as it makes the judgment difficult regarding the adequacy of the denudation to the required depth. Tumescent anesthesia and nerve blocks may be used in larger areas. If grafting is planned for extensive areas, general anesthesia may be needed in a hospital setting Recent studies suggest that adequate anaesthetsia can be achieved in a significant proportion of patients by using topical anaesthesia alone, provided adequate amount of EMLA cream is applied under effective occlusion for sufficient duration.
28.3 Methods of Surgical Modalities
Surgical modalities for vitiligo have been broadly classified into two types: tissue grafts and cellular grafts (Table 28.3). Special methods of treatment used in selected situation are tattooing and therapeutic wounding.
Table 28.3
Grafts used for vitiligo surgery
Tissue grafts | Cellular grafts |
|---|---|
Punch grafting/minigraft | Autologous, non-cultured epidermal cell suspension |
Suction blister grafting/epidermal grafting | Autologous, cultured melanocyte transplantation |
Split-thickness skin grafting | Autologous, cultured epithelial grafts |
Modified grafting techniques Mesh grafting Flip-top grafting Hair follicle grafting |
28.3.1 Tissue Grafts
28.3.1.1 Autologous Minipunch Grafting (PG)
Among the surgical modalities for vitiligo, minipunch grafting is the easiest, fastest, least aggressive, and minimally expensive.
- A.
Evolution
Norman Orentreich in 1972 first reported autograft repigmentation in humans and treated a black woman with long-standing leukoderma. Orentriech deployed nine, 1 and 2 mm diameter, normal skin autografts and observed the “pigment spread phenomenon” and reported a maximum of 1 mm pigment spread from both the 1 and 2 mm grafts [8]. Falabella treated vitiligo and secondary leukoderma by minigraft technique. He used minigrafts of size 1–1.2 mm in diameter from donor site and grafted them onto chambers of the same size at the recipient site spacing them 3 to 4 mm apart and further secured them using Monsel’s solution and pressure dressing [9, 10]. Because of similar size, the grafts did not snugly fit into the recipient chambers which led to circular perigraft scarring. Hence, Loewenthal’s method of using donor grafts larger than the recipient chambers was used by Falabella leading to better fixation of the grafts and lesser textural alterations. The procedure proved to be a highly effective modality in various studies [11–15].
- B.
Test grafting
Test grafting is done to confirm the stability of the disease before attempting repigmentation of the entire area. Few grafts (1–1.2 mm) are placed in the center of the depigmented lesion to be scrutinized. Dressing is done by adhesive tape (Micropore®) and kept for a couple of weeks. After removal of the tape, the area is exposed to sunlight for 15 min daily, for a period of 3 months. No treatment is given during this test period. All test sites are visualized under Wood’s light. The test is considered positive if unequivocal repigmentation takes place beyond 1 mm from the border of the implanted grafts. Apart from stability, it also gives a fair idea about the perigraft pigment spread which will guide to maintain adequate distance to be kept between the recipient chambers in future attempts. Over the years, this test has been acknowledged as a powerful tool for detecting stability of vitiligo and thereby anticipating success of the surgery in terms of repigmentation.
- C.
Instruments
The instruments required are cylindrical skin biopsy punches (1.2 to 1.5mm), “S”-shaped scissors, ring forceps, iris scissors, Adson’s toothed forceps, and a sterile petri dish/bowl.
- D.
Procedure
Recipient area
The recipient area is prepared first. Lignocaine 2 %, with or without adrenaline, is infiltrated as a local anesthetic. To minimize the chance of developing any perigraft halo, the initial recipient chambers are made on or very close to the border of the lesion. The punched out chambers are spaced according to the result of test grafting or at a gap of 5–10 mm from each other.
Donor area
The donor area is either the upper lateral portion of the thigh or the gluteal area. Punch impressions are made very close to each other so that a maximum number of grafts can be taken from a small area. In one sitting, up to 200 grafts can be harvested and grafted (average, 25–60). The remaining (extra) grafts can be stored in a refrigerator (on a shelf, not in the freezer) for reuse (to replace rejected grafts up to 24–48 h later).
Same-sized punches are used for both the donor and recipient areas.
The grafts are placed directly from the donor area to the recipient area which speeds up the procedure and lessens the chance of infection. The needle of the syringe or tip of the scissors can be used for the proper placement of grafts so that the graft edges are not folded and tissue is not placed upside down.
Hemostasis is achieved by applying firm pressure with a saline-soaked gauze piece over the area.
The recipient area is dressed with the three layers of paraffin-embedded, nonadherent sterile gauze (Jelonet®), sterile Surgipad®, and bio-occlusive Micropore® from inside to out.
For the donor area, only Surgipad® and Micropore® are used.
- E.
Post-procedure instructions
The recipient area may be immobilized, if necessary.
For special areas like the lips, the patients are advised to be on a liquid diet for the first 24 h, preferably with a straw, and are allowed a normal diet after this period.
To find out any dislodgment of grafts, dressings may be opened after 24 h, and if any are found, they need to be replaced.
Antibiotics and anti-inflammatory drugs are administered for 8–10 days.
- F.
Mechanism of pigmentation after grafting
The graft of the donor area functions independently of the recipient area. It produces active melanin, and the melanocytes from the infundibulum of hair follicles present in the graft spread centrifugally to the basal cell layer and thereby recolonize the epidermis of recipient area with active and functional melanocytes leading to perigraft pigmentation [8, 10, 20].
- G.
Complications [21] (Table 28.4)
Table 28.4
PG complications: recipient and donor sites
Recipient site
Donor site
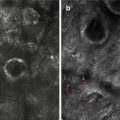
Fig. 28.1 After minigrafting, good repigmentation evident but cobblestoning seen
Cobblestoning (Fig. 28.1)
Polka dot
Variegated appearance and color mismatch
Static graft (no pigment spread)
Depigmentation of graft
Perigraft halo
Graft dislodgment/rejection
Hypertrophic scar and keloid formation
Target-like pigmentation [22]
Keloid
Hypertrophic scar
Superficial scar
Depigmentation/spread of disease
Contact dermatitis to adhesive tapes
- H.
Advantages and disadvantages (Table 28.5)
Table 28.5
PG: advantages and disadvantages
Advantages
Disadvantages
Simple, safe, and inexpensive office procedure
Needs no special training for a dermatologist.
High success rate and excellent cosmetic results
Large lesions and any site except the angle of the mouth can be treated
Areas of residual vitiligo between grafts or rejected can be regrafted
Specifically suitable for the areola
Perhaps the only suitable method for the palm
Commonly associated with side effects
Perfect color match does not occur
Phototherapy is required after grafting to achieve pigmentation
28.3.1.2 Suction Blister Grafting/Epidermal Grafting (SBEG)
Suction blister epidermal grafting is an established technique for the treatment of recalcitrant and stable vitiligo. The pigmented epidermis is harvested from the donor site by using suction to raise a blister which is then transferred to the vitiliginous area.
- A.
Evolution
Falabella, in 1971, first used epidermal sheets to treat vitiligo by in vivo separation of the viable epidermis [23]. However, various modified techniques have been used for producing the blister, including the use of syringes, three-way connectors, surgical glue, and modified BFY dermis– epidermis separator, making the procedure simple with improved results [24–27].
- B.
Instruments
Required are the following instruments: disposable syringes (10, 20, and 50 cc), three-way cannulas, dermabrader (manual or electrical), iris scissors, non-toothed forceps, artery forceps, sterile glass slides, and surgical glue (N-butyl-2-cyanoacrylate). If available, lasers (CO2 or Er:YAG) may be used.
- C.
Procedure
Donor Area
Preferred sites include the medial aspect of the forearm, the medial/lateral aspect of the upper arm, and the medial or posterior aspect of the upper thigh. After surgical cleansing, a topical local anesthetic is applied to the area as the procedure is painful; alternatively, 1 % lidocaine (Xylocaine) can be injected as a field block.
Raising of the Blister
Blisters may be raised using syringes, suction pump, suction cups, or a negative pressure cutaneous suction chamber system [24, 28, 29]. The most commonly used is the syringe suction. The bases of syringes sized 10 and 20 ml, coated with petroleum jelly (Vaseline), are applied on the donor site. Approximately 20–30 ml of air is aspirated using a 50 ml syringe and a three-way cannula. The three-way cannula is locked, and the 50 cc syringe is disconnected. This maintains constant negative pressure within the 10 or 20 cc syringe. It usually takes 1.5–3 h for the development of blisters. The best result is a single unilocular nonhemorrhagic blister. In case of smaller blisters, one can either increase the negative pressure in the syringe by another 5 ml or intradermally inject saline into the blister to expand it [18].
Blister deroofing
The roofs of the blisters are gently cut using iris scissors. The graft is then placed on a glass slide with the dermal side facing upward. The graft is then cleaned and spread to its maximum size and kept moist with normal saline. The donor site is then cleaned and bandaged using nonadherent dressing such as chlorhexidine gauze.
Recipient site
The recipient area is surgically cleaned using methylated spirit and povidone iodine and then anesthetized using plain lignocaine 1 %. The area is then dermabraded using a manual or motorized dermabrader, a microdermabrader, or a CO2 laser till minute pinpoint bleeding spots are visible. Gupta et al. used hypodermic needle as a dermabrading device for the smaller recipient area preparation before suction blister grafting [30]. Dermabrasion should extend for 1–2 mm beyond the border of the vitiliginous area. Hemostasis is achieved by pressure, and the area is covered with saline-soaked gauze pieces. Cryoblistering or suction blistering has also been used for the preparation of the recipient site [31].
Transfer of grafts
The grafts are then placed such that the dermal side of the graft is now in contact with the dermabraded area. A gap of 0.5 cm can be left between two grafts because there is a pigment spread.
Graft fixation
With sterile moist gauze, firm pressure is given over the graft to remove any serous collection underneath. This helps in graft adherence. The recipient area is dressed with double layer framycetin tulle moist gauze, followed by sterile gauze and Elastocrepe bandage. Alternately, surgical glue (N-butyl-2-cyanoacrylate) can be applied along the edges of the grafts for fixation. Donor area is dressed with dry sterile pads.
- D.
Post-procedure instructions
The dressing over the donor and recipient sites is removed after 24 h and 7 days, respectively. The patient is advised to keep the area immobile. Usually, the grafts fall off in 1–2 weeks.
Oral or topical Psoralen-UVA or PUVASOL can be started from the day of removal of dressing to facilitate repigmentation.
- E.
Principle of suction blister epidermal grafting
In suction blister grafting, the cleavage occurs between the basal cells and the basal lamina of the basement membrane zone. Hence, only the epidermal portion of the donor area is grafted. So the graft generally acquires the characteristics of the recipient site rather than the donor site, resulting in a better color match and cosmetic outcome [32]. The melanocyte transfer takes place within 48–72 h from the graft to the recipient site.
- F.
Complications (Table 28.6)
Table 28.6
SBEG complications: donor and recipient sites
Donor site
Recipient site
Ecchymosis
Hematoma
Pigmentary changes
Secondary infection
Hyperpigmentation
Incomplete pigmentation
Perigraft halo
Graft rejection
Allergic contact dermatitis
- G.
Advantages and disadvantages (Table 28.7)
Table 28.7
SBEG: advantages and disadvantages
Advantages
Disadvantages
Safe, easy, and inexpensive method
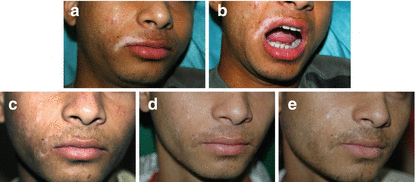
Fig 28.2 (a, b) Segmental Vitiligo – before suction blister epidermal grafting; (c–e) Post-procedure – good repigmentation of both vitiligo skin and leukotrichia
Very good success rates (Fig. 28.2 a–e)
Repigmentation is faster
Very good color match
Time-consuming
Painful procedure
Larger areas require multiple sittings
Improper handling may lead to tearing of the graft
Wrong placement of graft leads to failure of repigmentation
Not suitable for palms and soles
Color mismatch (Fig. 28.3 a, b)
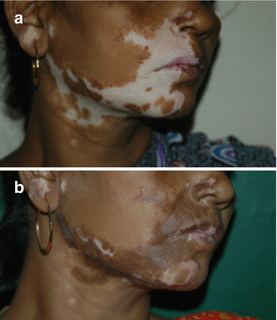
Fig 28.3 (a) Segmental Vitiligo – before suction blister epidermal grafting (b) after the procedure, note color mismatch
28.3.1.3 Split-Thickness Skin Grafting (STSG)
Split-thickness skin grafting involves transfer of epidermis and often the uppermost part of superficial dermis into the dermabraded patch of vitiligo, thereby achieving the transfer of melanocytes and keratinocytes from donor graft to the recipient area [8]. The thickness of the graft ranges from 0.1 to 0.7 mm [35].
- A.
Evolution
In 1964, Behl was the first to report the use of thin Thiersch’s skin grafts to treat vitiligo [36]. Since then, various modifications of the same procedure like ultrathin epidermal sheets or ultrathin split-thickness grafting (less than 0.08–0.15 mm) have been tried for the treatment of vitiligo [37–40].
- B.
Instruments
These include dermabrader (manual or electrical), skin grafting equipment, graft spreading rods/spatula, non-traumatizing ring forceps, iris scissors, surgical glue-Cyanoacrylate adhesive (N-butyl-2-cyanoacrylate), and a sterile petri dish/bowl.
- C.
Procedure
Donor area
Preferred sites include the gluteal area and anterolateral aspect of the thigh, abdomen, and arms [37, 41]. After shaving off the hairs and proper cleansing with povidone iodine and 70 % ethanol, the area is anesthetized with 1 % lidocaine (Xylocaine) without adrenaline. The area is then stretched, and a thin, even split-thickness graft is harvested, using either a sterile razor blade mounted on a Kocher’s forceps or a blade-holding instrument. Alternatively, a hand dermatome, Humby’s knife, Silver’s knife, or an air-driven power dermatome may be used for harvesting the grafts [42–44]. The grafts thus obtained are transferred to a petri dish containing normal saline. The donor area is dressed with nonadherent dressing.
Recipient area
After proper cleansing with povidone iodine and 70 % ethanol, the area is abraded using a dermabrader, until pinpoint hemorrhages are seen uniformly all over the lesion. The area is then cleaned with normal sterile saline and covered with a gauze piece soaked in normal saline. The grafts obtained are then placed upside down on the sterile glass slides. The slide is placed on the recipient area and pressed against the skin. Any blood or exudates between the graft and recipient area are evacuated by firm pressure over the graft with the help of wet gauze, without displacing the graft. The graft is immobilized by using surgical adhesive, octyl-2-cyanoacrylate and pressure dressing. The adhesive gives excellent results to secure the graft and also has antimicrobial properties against Staphylococci, Pseudomonas, and E. coli [45]. The recipient site can also be prepared using a pulsed Erbium-YAG laser or ultrapulse CO2 laser [46].
- D.
Post-procedure instructions
Patients are advised to avoid excessive movement of the grafted area and to follow up after 1 week for a change of dressing. Donor site dressing is also changed after 1 week.
Prophylactic oral antibiotics are given for 1 week to prevent postoperative infection.
If there is perigraft depigmentation or achromic fissures, NB-UVB is required postoperatively for complete repigmentation.
- E.
Principle of split-thickness skin grafting
Three biological changes occur following skin grafting [41].
Graft take adherence
This stage is comprised of two phases. The first phase occurs within 72 h of the placement of the graft. Fibrin bonding occurs between the graft and recipient area leading to graft adherence. The second phase begins with the onset of vascular anastomosis and fibrovascular growth.
Graft revascularization
The graft and host vessels get connected with the formation of new vascular channels.
Contracture
The contraction of the elastin fibers leads to the contracture of the graft after harvesting, and a similar phenomenon also occurs at the recipient site. The above two factors contribute to achromic fissures and perigraft halo which follows the grafting. Overlapping of the graft edges at the recipient site can prevent these complications.
- F.
Complications (Tables 28.8 and 28.9)
Table 28.8
STSG complications: recipient and donor sites
Recipient site
Donor site
Graft rejection
Textural abnormalities like “curling” or “branching,” resulting in peripheral beading at the graft edge or a “stuck on tire patch” appearance
Hyperpigmentation
Perigraft halo
Hypertrophy
Milia
Secondary infection
Reactivation of vitiligo
Post inflammatory hyperpigmentation
Scarring
Recurrence or Koebnerization
- G.
Advantages and disadvantages
Table 28.9
STSG: advantages and disadvantages
Advantages
Disadvantages
Pigmentation can instantly cover larger areas over a short period of time
Difficult areas such as eyelids, inner canthus of eyes, areola, nipples, and genitals can be treated readily
Pigmentation achieved is uniform [47]
Less time-consuming as compared to other procedures
Prolonged hyperpigmentation particularly on the exposed areas in dark-skinned patients
Superficial scarring and chances of vitiligo at the donor site
Larger area requires multiple sitting
Requires surgical skill for graft harvesting
Tips, palms, soles, and mucous membranes are difficult to treat by this method
Scarring and contracture at the recipient area [50]
Risk of malignant transformation in keratinized grafts applied in the oral mucosa [51]
28.3.1.4 Modifications of Techniques at Special Sites [32]
- A.
Eyelids
The thinnest graft should be selected for the upper eyelid. For small area, suction blister graft is ideal. Thin or ultrathin split-thickness skin grafts give good results if the entire eyelid is involved. Strict immobilization for the first 72 h is essential for graft uptake.
- B.
Lips
Suction blister grafts for small areas and thin split-thickness skin grafts for larger areas give a good cosmetic outcome on the lips.
- C.
Areola
The entire areola should be grafted in order to maintain a uniform color.
- D.
Acral areas
Minipunch grafting is most suitable for the fingers, toes, palms, and soles. Suction blister grafting or thin split-thickness skin grafting is suitable for the dorsal sites.
- E.
Genitals
The history of genital herpes should be always ruled out before attempting surgical treatment. Long-term prophylaxis with acyclovir should be given in proven and doubtful cases and the prognosis clearly explained. Suction blister grafts, thin split-thickness skin grafts, minipunch grafts, and non-cultured melanocyte suspensions can be tried, depending on the size of the vitiligo patch and expertise of the surgeon [52].
- F.
Hairy areas
The hair should be plucked out instead of shaving before grafting to delay hair regrowth and thereby preventing lifting up of the graft. Alternatively, a chemical depilatory may be used.
28.3.1.5 Mesh Grafting (MG)
Mesh grafting is a technique where the graft is expanded by making slits in it such that it appears like a mesh [53].
- A.
Instruments
These include a Padgett or Duvals dermatome, an ampligreffe or Discard-A-pad, and a dermabrader.
- B.
Procedure
Donor site
Under general anesthesia, a 0.01 mm thickness graft is obtained from the anterolateral aspect of the thigh, using a Padgett dermatome. The grafts, with the dermis facing upward, are transferred to an ampligreffe, specifically placed on its slightly inclined metal plate. With one end of the graft inserted between the spiral barrels of the ampligreffe, the latter’s handle is then rotated. There occurs the formation of a diamond-shaped mesh when the graft passes between the two barrels of the ampligreffe. The resultant mesh is of four times the size of the original graft.
Recipient site
After the recipient site is cleaned and dermabraded, the graft is transferred and bandaged with saline-soaked dressing or framycetin (Sofratulle®). The donor site is also dressed using aseptic precautions.
- C.
Post-procedure instructions
The dressing at the recipient site is removed after a week.
Phototherapy is started immediately or after a week.
- D.
Advantages and disadvantages (Table 28.10)
Table 28.10
MG: advantages and disadvantages
Advantages
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree

 Get Clinical Tree app for offline access
Get Clinical Tree app for offline access