Ventral Hernia: Component Separation Technique
Mark W. Clemens
Charles E. Butler
DEFINITION
The component separation technique is a type of rectus abdominis muscle advancement flap that reconstructs ventral hernia and large abdominal wall defects.
Component separation is a fascial release of the external oblique fascia with creation of musculofascial advancement flaps.
The general indications for performing a component separation of the abdominal wall include a deficiency of the abdominal wall fascia, which would require a bridged repair without fascial release.
Ramirez and colleagues’ description of the surgical technique of component separation moves the rectus musculofascia medially, to close midline defect by releasing the external oblique aponeuroses and posterior rectus sheath bilaterally.1
Myofascial advancement techniques, or component separation, takes advantage of the laminar nature of the abdominal wall and the ability to release one muscular or fascial layer to enable medial advancement of another.
ANATOMY
The lateral abdominal wall musculature is composed of three layers, with the fascicles of each muscle directed obliquely at different angles to create a strong envelope for the abdominal contents.
Each of the muscles forms an aponeurosis that inserts into the linea alba, a midline structure joining the two sides of the abdominal wall.
The external oblique muscle is the most superficial muscle of the lateral abdominal wall.
The deepest muscular layer of the abdominal wall is the transversus abdominis muscle with its fibers coursing horizontally. These three lateral muscles give rise to aponeurotic layers lateral to the rectus abdominis muscle, which contribute to the anterior and posterior layers of the rectus sheath.
The transversus abdominis muscle inserts into the transversus abdominis aponeurosis to form the posterior rectus sheath and continues as a muscular component laterally in the upper abdomen. This feature is important when considering the technical aspects of a posterior component separation.
At the midline, the two anterior rectus sheaths form the tendinous linea alba. On either side of the linea alba is the rectus abdominis muscle, the fibers of which course vertically and run the length of the anterior abdominal wall.
The arcuate line, located 3 to 6 cm below the umbilicus, delineates the point below which the posterior rectus sheath is absent and is composed of the transversalis fascia and peritoneum.
The abdominal wall receives the majority of its innervation from the 7th to 12th intercostal nerves and the first and second lumbar nerves.
The lateral abdominal muscles receive their blood supply from the lower three to four intercostal arteries, the deep circumflex iliac artery, and the lumbar arteries. The rectus abdominis muscles are perfused by the superior epigastric artery (a terminal branch of the internal mammary artery), the inferior epigastric artery (a branch of the external iliac artery), and the lower intercostal arteries.
PATHOGENESIS
Indications for abdominal wall reconstruction include tumor ablation, congenital anomalies, and trauma.
Abdominal defects and hernias may result from genetically impaired collagen formation, deposition, organization with sequelae from injury, failed laparotomy closures, or failed hernia repairs.
Risk factors for the development of hernias included tobacco use and a strong family history of hernia, which suggests a genetic predisposition.
PATIENT HISTORY AND PHYSICAL FINDINGS
The most common presentation to reconstructive surgeons is an incisional hernia as sequelae to a previous laparotomy.
There are three general indications to repair an incisional hernia: the hernia is symptomatic, causing pain or alterations in the bowel habits; the hernia results in a significant protrusion that affects the patient’s quality of life; and the hernia poses a significant risk of bowel obstruction (such as a large hernia with a narrow neck).
Acquired hernias typically occur after surgical incisions and thus are commonly referred to as incisional hernias.
Epigastric hernias occur from the xiphoid process to the umbilicus, umbilical hernias occur at the umbilicus, and hypogastric hernias occur below the umbilicus in the midline.
Although not a true hernia, diastasis recti can present clinically as a bulge in the midline. In this condition, the linea alba is stretched, resulting in bulging at the medial margins of the rectus abdominis muscles. There is no fascial ring or true hernia sac. Unless significantly symptomatic, diastasis recti does not need to be corrected surgically.
Previous rectus abdominis muscle flap harvest does not preclude the future use of component separation techniques.2
IMAGING
Preoperative computed tomography to examine the defect characteristics and abdominal wall anatomy and vascularity is helpful for surgical planning.
Computed tomography (CT) allows for visualization of intraabdominal organs, and the abdominal wall, three-dimensional data sets, and multiplanar reformation capabilities.
CT scans may assist in detecting fluid collections, bowel obstruction, incarceration, strangulation, and traumatic wall hernias.
Magnetic resonance (MR) imaging also permits the detection of soft tissue defects and abdominal wall hernias though this modality does not usually offer further sensitivity and therefore may be cost-prohibitive.
SURGICAL MANAGEMENT
Preoperative Planning
Physical examination should be performed to assess the patient’s general condition, the abdominal wall integrity, and the extent and location of any abdominal wall abnormalities.
The presence of scars that compromise blood supply could become an obstacle to raising reliable tissue flaps.
Routine laboratory tests and a nutritional workup are advised.
Patients should be marked in the preoperative holding area, and it is beneficial to evaluate patients in a recumbent and supine position for complete evaluation of abdominal wall defects.
Markings may delineate anatomical boundaries such as the pelvis, midline, and costal margin as well as the fascial extent of any intra-abdominal defects.
Bowel preps may be beneficial in patients with anticipated violation of the gastrointestinal tract.
Intravenous antibiotics are initiated in the holding area.
Positioning
Patients are placed supine on the operative table, sedated, and intubated.
The abdomen is widely draped and prepped to expose the patient’s flanks and from the pelvis to the midsternal area.
Patients should receive sequential compression devices and or compression hoses for deep vein thrombosis prophylaxis.
Patients requiring greater exposure should have room temperatures maintained above 75°F to minimize postoperative infections.
Approach
All approaches to abdominal wall closure should be aimed at re-establishment of the abdominal domain integrity with complete fascial coaptation.
All attempts should be made to avoid a bridged mesh repair because there is a clear trend toward higher recurrence rates compared with when the fascia can be reapproximated over a mesh repair.
Component separation release can be done by open or minimally invasive component separation. A minimally invasive component separation can be performed in various ways, but all of the techniques (to a certain degree) maintain the blood supply to the skin from the underlying rectus abdominis muscles.
TECHNIQUES
▪ Open Component Separation
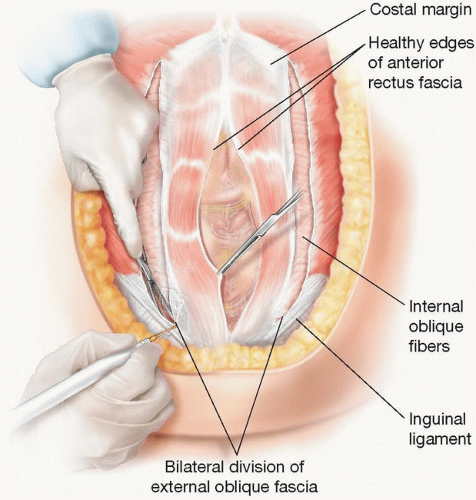
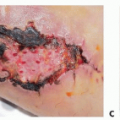
An open component separation is performed by raising large subcutaneous flaps to expose the external oblique fascia3 (TECH FIG 1).
The cutaneous perforators emerging from the anterior rectus sheath are ligated and divided to facilitate exposure of the linea semilunaris in its entirety. These flaps are carried laterally past the linea semilunaris.
An anatomically precise external oblique aponeurotomy is made 1 to 2 cm lateral to the linea semilunaris on the lateral aspect of the external oblique aponeurosis from several centimeters above the costal margin to the pubis. It is important to confirm that the incision is not carried through the linea semilunaris, as this would result in a full-thickness defect of the lateral abdominal wall, which is challenging to repair.
The external oblique aponeurosis is then bluntly separated in the avascular plane away from the internal oblique aponeurosis to the midaxillary line, allowing the internal oblique and transversus abdominis muscles with the rectus abdominis muscle or fascia to advance medially as a unit.
These techniques, when performed bilaterally, can yield up to 20 cm of mobilization in the midabdomen.
Once the mesh inset and fascial closure are performed, the subcutaneous skin flaps are advanced and closed at the midline.
To reduce subcutaneous dead space, interrupted quilting sutures should be placed between the Scarpa fascia and musculofascial repair.
This technique also decreases shear stress, which is thought to contribute to postoperative seroma formation, and decreases the total drain output, allowing the surgeon to place fewer drains and leave them in for a shorter period.
After paramedian skin perfusion is critically assessed, a vertical panniculectomy may be performed so that the skin is reapproximated in the midline without redundancy.
▪ Laparoscopic Component Separation
Laparoscopically, component separation is performed through a 1-cm incision below the tip of the 11th rib overlying the external oblique muscle.4,5
The external oblique muscle is split in the direction of its fibers, and a standard bilateral inguinal hernia balloon dissector is placed between the external and internal oblique muscles and directed toward the pubis (TECH FIG 2).6
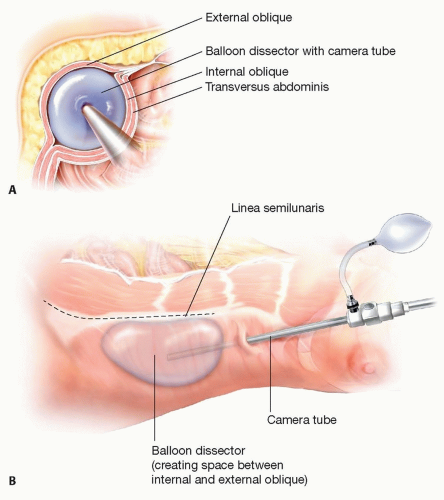
TECH FIG 2 • Laparoscopic component separation. A. Access to the external oblique aponeurosis is achieved through a small incision at the costal margin through which a balloon dissector is placed. The external oblique aponeurosis is then divided from the pubis to above the costal margin. B. This minimally invasive approach preserves the attachments of the subcutaneous tissue (including myocutaneous perforators) to the anterior rectus sheath throughout its course.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree

 Get Clinical Tree app for offline access
Get Clinical Tree app for offline access