Capsular contracture remains a common and preventable complication of implanted breast prostheses. As our understanding of the pathophysiology continues to develop, it is prudent to reexamine existing beliefs in a contemporary context. This article presents a current summary of clinical and laboratory evidence, expressed as an interaction between potentiating and suppressing factors, and how this understanding can be applied to practice.
Key points
- •
Chronic inflammation attributable to the presence of bacterial biofilm is currently the predominant theorem of capsular contracture formation. Conversely, maintenance of implant sterility will prevent capsular contracture.
- •
The surgical field in breast augmentation is not rendered sterile by standard techniques and perioperative antibiotics.
- •
Thus, a comprehensive strategy for maintaining implant sterility is critical in preventing bacterial contamination, and is supported by mounting clinical and laboratory evidence.
Introduction
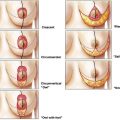
In an ideal world a medical device, once inserted into the human body, will undergo biological integration with complete tolerance and without any remote-to-implant effects, leading to long-term successful functional and aesthetic outcomes. In the real world, however, medical devices continue to be plagued with a wide spectrum of adverse outcomes, ranging from acute infection to long-term device failure necessitating removal. For breast implants, capsular contracture (CC) represents the most common and most problematic adverse outcome. Following primary augmentation, CC occurs in up to 59% of patients, and is the commonest indication for reoperation. Patients undergoing revision surgery for contracture can expect to have recurrence in 18.1% to 39.7% of cases. Furthermore, revision surgery is technically more difficult, and satisfaction rates are lower ( Fig. 1 ).
Several hypotheses have been forwarded to explain why capsules form and why they progress pathologically around some breast implants, including infection, hematoma, and granulomatous response to free silicone. The most recent, and closest unifying theory is that of septic biofilm formation, elegantly expressed by Adams as an interaction between potentiators and suppressors that come into play when an implant is first inserted into a perimammary space. It is this interplay of factors that leads to either implant tolerance or progressive peri-implant inflammation and subsequent fibrosis, generating CC ( Fig. 2 ).
This review examines the science behind each of these factors, determining their relative importance as either potentiators or suppressors of CC formation. The article also outlines the likely strategies that surgeons can deploy in the operating room to minimize the risk of adverse outcomes in breast augmentation based on the cumulative scientific evidence. In this way, evidence can be used to guide clinicians toward translating the findings of this growing body of research into clinical practice, ultimately for the benefit of patients.
Introduction
In an ideal world a medical device, once inserted into the human body, will undergo biological integration with complete tolerance and without any remote-to-implant effects, leading to long-term successful functional and aesthetic outcomes. In the real world, however, medical devices continue to be plagued with a wide spectrum of adverse outcomes, ranging from acute infection to long-term device failure necessitating removal. For breast implants, capsular contracture (CC) represents the most common and most problematic adverse outcome. Following primary augmentation, CC occurs in up to 59% of patients, and is the commonest indication for reoperation. Patients undergoing revision surgery for contracture can expect to have recurrence in 18.1% to 39.7% of cases. Furthermore, revision surgery is technically more difficult, and satisfaction rates are lower ( Fig. 1 ).
Several hypotheses have been forwarded to explain why capsules form and why they progress pathologically around some breast implants, including infection, hematoma, and granulomatous response to free silicone. The most recent, and closest unifying theory is that of septic biofilm formation, elegantly expressed by Adams as an interaction between potentiators and suppressors that come into play when an implant is first inserted into a perimammary space. It is this interplay of factors that leads to either implant tolerance or progressive peri-implant inflammation and subsequent fibrosis, generating CC ( Fig. 2 ).
This review examines the science behind each of these factors, determining their relative importance as either potentiators or suppressors of CC formation. The article also outlines the likely strategies that surgeons can deploy in the operating room to minimize the risk of adverse outcomes in breast augmentation based on the cumulative scientific evidence. In this way, evidence can be used to guide clinicians toward translating the findings of this growing body of research into clinical practice, ultimately for the benefit of patients.
Factors influencing capsular contracture formation
Bacteria and Subclinical Infection (Strong Potentiator)
The deep breast parenchyma is not intrinsically a sterile structure: it contains a penetrating ductal anatomy, populated by flora predominantly consisting of Staphylococcus epidermidis . The contribution of periprosthetic infection to CC has been recognized for some time, being first proposed by Burkhardt and colleagues in 1981. However, many CCs form without gross clinical infection or positive culture, implying that infection may not be the universal mechanism.
An important step forward was the recognition of periprosthetic bacterial biofilms. Biofilm consists bacteria enclosed within a matrix of their own excreted polysaccharides. This structure allows bacteria to densely adhere to prosthetic and biological surfaces. In this state, bacterial contamination may elude standard microbiological sampling, owing to a paucity of surface bacteria and low metabolic rate. Specialized techniques are required to detect biofilms and identify their contributing microorganisms. The biofilm structure also confers significant resistance against antibiotics, host defenses, and antiseptics. There may be other more subtle advantages to the bacteria, such as improved nutrition through promotion of host cell lysis, cooperative metabolism, and horizontal gene transfer ( Fig. 3 ).
Through treatment resistance and eliciting a chronic inflammatory response, biofilms may ultimately lead to implant failure in the clinical situation. A landmark experiment by Tamboto and colleagues inoculated silicone implants with S epidermidis in a porcine model; this was strongly associated with biofilm formation, which itself correlated with a 4-fold increase in CC. It may be that periprosthetic infection has, all along, been a major causative factor in CC formation, and that the ability of biofilms to evade detection by standard sampling methods has confounded our understanding to date. The chronic inflammatory response to S epidermidis biofilms is now recognized as a major pathogen in CC.
Implant Texturing (Both Suppressor and Strong Potentiator)
Barr and colleagues investigated the cytoskeletal reaction of fibroblasts to silicone surfaces. Smooth surfaces predisposed to the planar arrangement of fibroblasts around implants, whereas textured implant surfaces caused fibroblasts to anchor in deep and random patterns. This situation implied an advantage for texturing with respect to CC. The body of conflicting clinical evidence was unified in 2 meta-analyses in 2006, which concluded a 5-fold superiority of textured over smooth implants, maintained out to 3 years.
However, this advantage can be a double-edged sword. When inoculated in vitro, a significantly higher biofilm load is found in textured implants than in smooth implants. This finding suggests that the benefits of texturization may become a liability in infection because of a higher risk of bacterial colonization. With the recent implication of textured implants and biofilms in the causation of anaplastic large cell lymphoma, their use may bear additional scrutiny born of valid concern for oncological safety.
Polyurethane (PU)-coated implants are a notable subset of textured implants. The surface texture is a fibrillar, mesh-like coating, as opposed to having being molded into the silicone or generated through salt removal. This process generates a capsular microarchitecture distinct from textured silicone surfaces, possibly caused by a controlled inflammatory response guided by polyurethane fragment phagocytosis, and lower expression of periprosthetic parallel myofibrils. Previous concerns regarding carcinogenic degradation into 2,4-toluenediamine products have been disproved, although the coating does indeed degrade over time, leaving the equivalent of a standard textured silicone surface. PU implants have been shown in numerous studies to have a lower CC rate than other implants.
Silicone Foreign Body Reaction (Potentiator)
Despite being chemically stable, silicone is not biologically inert: a universal foreign body reaction is elicited. An issue with older generations of silicone implants was gel bleed through the shell, or mechanical shell rupture, producing free silicone that initiated a granulomatous response. Silicone fillers were hence seen as a potentiating factor in CC formation. However, in modern implants, this is addressed through modified shell characteristics and the use of form-stable cohesive gels. Hence, previous studies implicating implant fillers as a significant potentiator of CC cannot be readily discussed in a contemporary context.
Surprisingly, a 2010 literature review by Schaub and colleagues was unable to reliably determine whether a difference existed between silicone and saline with respect to CC rate in cosmetic augmentation. Confounding their meta-analysis was the sheer heterogeneity of implant and technical variables present throughout the literature, in addition to differences in the basic definition of CC.
Incision Choice
Augmentation mammoplasty wounds were previously considered clean, contrary to the now accepted knowledge that the deeply penetrating ductal system is colonized with bacterial flora. Periareolar bacterial counts have been demonstrated to be 5 times that of the inframammary fold and 4 times that of the axilla. Thus, a periareolar incision may contaminate the sterile field by transecting ductal tissue, and places an implant in contact with bacteria during insertion. In 2008, Wiener demonstrated that a periareolar incision was associated with a 9.5% CC rate, compared with 0.59% for an inframammary incision. The relatively high CC rate in the periareolar group was despite the universal use of povidone-iodine pocket irrigation and perioperative antibiotics. Thus, the evidence clearly supports periareolar incisions as a strong potentiating factor in CC formation; conversely, inframammary incisions are protective. Because of their preference for cohesive gel implants, the authors predominantly use the inframammary incision, as the form-stable implants are difficult to squeeze through a periareolar approach. Should a surgeon elect to use a periareolar incision, the authors would recommend using a plastic insertion sleeve to isolate the implant from the wound tract.
The authors have little experience with transaxillary incisions in their practice. Previous criticisms of hematoma from blind, blunt transaxillary pocket dissection have been addressed through the use of endoscopic direct vision. This technical difference was the basis of a comparison series by Tebbetts, who reported decrease in CC from 4.2% to 1.3% when adopting an endoscopic technique, similar to that reported by Giordano and colleagues. Conversely, a series by Huang and colleagues was largely performed without endoscopic assistance, with a CC rate of 1.9%. Stutman and colleagues did not find any significant difference in infection rates between transaxillary, inframammary, and periareolar incisions, although their retrospective study design was limited by a large number of other variables. Contrary to this, Namnoum and colleagues found a significantly higher rate of CC associated with axillary incisions, but not periareolar, when compared with the inframammary approach.
Submuscular Pocket (Suppressor) Versus Subglandular Pocket (Potentiator)
It has been a consistent finding that the submuscular pocket is protective against CC in comparison with subglandular placement, likely because the implant is not exposed to potential colonization by contact with glandular-bearing breast tissue. The subfascial approach may offer a similar advantage. However, the authors are not aware of a trial demonstrating that irrigation of a subglandular pocket with antiseptics reduces the risk of CC relative to that of a submuscular pocket.
Implant Exposure Time (Potentiator)
Through controlled laminar airflow and particular filtration, modern operating theaters are designed to minimize the introduction of particulate contaminants into the sterile surgical field. Despite this, airborne infection is predisposed to by turbulent disruption of settled particles, increased traffic of operating room personnel, talking, and uncovered skin areas. It follows that increased open-air exposure time predisposes to infection; increased operative time is associated with increased rates of infection in orthopedic joint replacement procedures. Although there are no data specifically linking increased implant exposure time to CC, the authors recommend that the infection risk be curtailed by only opening implants immediately before insertion.
Induction Intravenous Antibiotics (Suppressor)
Routine use of a single, preincision dose of intravenous antibiotic has been shown to decrease the microbiological load on the intraoperative wound, and the rate of infection in breast implants. Extending the duration of antibiotic cover postoperatively does not result in reduced superficial or periprosthetic infections. Once a biofilm is established, a 1000-fold increase in minimum antibiotic concentration may be required to provide bactericidal activity. It would therefore seem reasonable, given that infection can lead to CC let alone gross implant sepsis, to routinely administer prophylactic antibiotics before incision. Yet a recent meta-analysis by Hardwicke and colleagues reported little high-quality contemporary evidence available for analysis: no trials of antibiotics versus controls were found in the preceding 17 years. Only studies comparing antibiotic regimens were available, and no significant difference was found with respect to infection rates. Specifically with respect to CC, the role of systemic antibiotic prophylaxis was concluded as unknown, and overall recommendations could not be made.
Despite these data and in view of the strong evidence favoring subclinical infection as a potentiator of CC, it is the authors’ practice to routinely administer a single dose of intravenous antibiotics at anesthetic induction. However, because of known resistance of some S epidermidis isolates to methicillin (and thus cephalosporins), antibiotic use does not afford total protection. Wixtrom and colleagues were able to culture S epidermidis from 34.9% of nipples and nipple shields at the conclusion of augmentation procedures, despite povidone-iodine surface preparation and preoperative intravenous antibiotics. Other methods of reducing microbial contamination of implants thus remain important adjuncts to reducing the risk of CC.
Pocket Irrigation (Suppressor)
Burkhardt first proposed the subclinical infection model in 1981, and has advocated povidone-iodine pocket irrigation. Wiener was able to achieve CC rates much lower than those in contemporary publications through routine use of povidone-iodine pocket irrigation. Irrigation with povidone-iodine, cefuroxime, and gentamicin seems to be advantageous over systemic antibiotics alone in significantly reducing CC, even if no difference is observed in infection rates. Both Adams and colleagues and Araco and colleagues routinely used pocket irrigation and perioperative antibiotics, and achieved commendably low rates of CC in their respective series. However, infection per se rather than CC formed the focus of their discussions, and there was no comparison between regimens. Pfeiffer and colleagues were forced to withdrawal cephalothin from their routine pocket irrigation because of supply issues, thus creating a trial of antibiotic versus nonantibiotic irrigation. Despite a significant increase in seroma and infection, there was no significant difference in CC rate.
Nipple Coverage (Suppressor)
Collis and colleagues demonstrated that bacterial contamination of the operative field may occur despite adequate skin preparation. These investigators also demonstrated that this may be prevented by placement of an occlusive dressing over the nipple-areola complex, which was subsequently confirmed by Wixtrom and colleagues. Although neither study design was suited to examining the role of nipple shields in preventing CC, Wiener achieved particularly low rates of CC when placing povidone-iodine–soaked gauze over the nipple-areola complex, in addition to pocket irrigation. These studies support the routine use of nipple-areolar dressings to prevent bacterial contamination of the surgical field, a practice that is both inexpensive and easy.
Introduction Sleeves (Suppressor)
As already mentioned, the breast surface and parenchyma may not necessarily be free of bacterial contamination, even in the context of standard sterile technique. Thus, implant contact with the surgical field may cause a breach of sterility. Mladick described the use of introduction sleeves as part of a “no-touch” technique for saline implant insertion. Although a specialized medical device exists expressly for this purpose, the authors have used a cut-off surgical glove as an effective improvisation. Aside from a protective role, sleeves may also ease the inset (and thus decrease handling) of polyurethane implants. Although no comparisons exist to specifically support sleeves as protective against CC, the technique is a simple and inexpensive modification to surgical practice ( Fig. 4 ).