Fig. 24.1
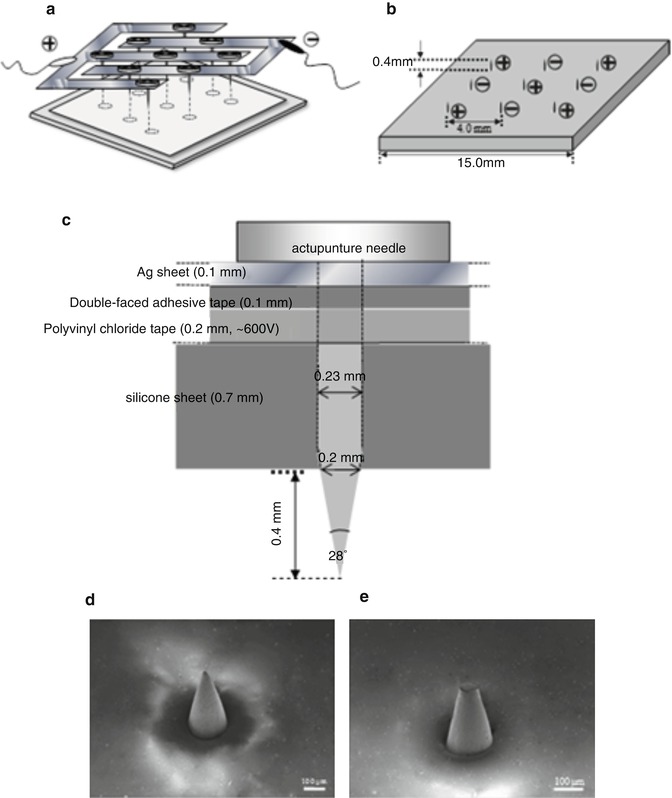
Schematic illustration of microneedle array made by acupuncture needles. (a) Top view, (b) back view, (c) one needle. Each needle tapered over a 400-μm length to a sharp tip with 28° angle, and the base diameter was approximately 200 μm. The microneedle array was manually pressed onto the rat skin, with the stratum corneum facing uppermost, at an approximate pressure of 1.6 kg/cm2 for 10 s
Since microneedles can provide a minimally invasive means of delivery of therapeutic molecules into the skin (Matriano et al. 2002), the combination of barrier impairment using microneedles and following iontophoresis or electroporation can be expected as a means to broaden the range of drugs suitable for transdermal delivery; however, few studies have reported on the combination of iontophoresis or electroporation with microneedle technologies. Thus, in this chapter, the synergistic action of iontophoresis or electroporation after combination with microneedle was considered to further increase the skin permeation and bioavailability of high molecular weight compounds, due to the achievement of a longer duration of pore opening which leads to higher skin permeation of macromolecules.
24.2 Physical Methods (Electroporation and Iontophoresis) to Increase Skin Permeation of High Molecular Weight Compounds
In order to overcome the high barrier function in the stratum corneum for delivering these macromolecular drugs through the skin, several penetration-enhancing physical means have already been evaluated in addition to chemical enhancers to promote their skin permeation, such as iontophoresis (Chien and Banga 1989), electroporation (Prausnitz et al. 1993), phonophoresis (sonophoresis) (Mitragotri et al. 1995), and microneedles (McAllister et al. 2003), as aforementioned. These physical means usually show higher penetration-enhancing ability than chemical penetration enhancers, like low molecular alcohols and aliphatic esters (Bhatia and Singh 1999). Iontophoresis has been shown to increase the skin permeation of many molecules (Wu et al. 2007). Leduc had shown nearly 100 years ago that this technique could be used to deliver drugs across mammalian skin in vivo, but this did not gain scientific prominence until 20 years ago (Chien and Banga 1989). Iontophoretic flux is obtained not only due to electrorepulsion but also electroosmotic solvent flow, which is generated from anode to cathode by anodal iontophoresis. Thus, iontophoresis can be used to enhance transdermal delivery of ionic drugs as well as nonionic compounds. In the present chapter, iontophoresis-induced electroosmotic flow, electroosmosis, was combined with microneedle drug delivery as a “push force.” The question now is whether the electrical properties of the skin, which are the origin of electroosmotic flow (Pikal 2001), are affected by microneedle pretreatment. Physical abrasion of the stratum corneum and the application of a depilatory cream on the skin surface can facilitate the iontophoretic delivery of insulin (Kari 1986; Kanikkannan et al. 1999). However, removal of the stratum corneum by tape stripping decreased or even abolished the skin electroosmotic flow (Abla et al. 2005). Hirsch et al. also reported that iontophoresis applied on stripped skin did not result in a higher drug delivery than iontophoresis alone (Hirsch et al. 2005). The question whether iontophoresis in conjunction with physical impairment of the skin barrier can further enhance the transdermal delivery of high molecular weight compounds must therefore be related to the electrical properties of the skin, i.e., electroosmotic flow, after the pretreatment. On the other hand, another physical means, electroporation technology, widely used for introducing deoxyribonucleic acid (DNA) and ribonucleic acid (RNA) into cells, biological tissues, and bacteria (Zimmermann et al. 1973; Neumann and Rosenheck 1972), is also an attractive skin penetration-enhancing method (Prausnitz et al. 1993, 1995). Electroporation involves the creation of tiny and transient aqueous pathways (pores) in the transcellular lipid region in the stratum corneum barrier by applying a high-voltage pulse for a very short period (millisecond order) (Chen et al. 1999). Electroporation application with a high-voltage electric pulse for a short period results in the enhanced permeation of high molecular weight compounds (molecular weight of several hundreds to kilodaltons) through the skin (Wong et al. 2006; Tokumoto et al. 2006). For both microneedles and electroporation, in spite of their different mechanisms, enhancement of the skin permeability of drugs is a result of the creation of new permeation pathways in the stratum corneum; however, the pathways do not persist, so that the skin barrier function is recovered immediately after the pathways close up. Transepidermal water loss (TEWL), an index of skin permeability for drugs (Zhou et al. 2010), increases after the application of microneedles or electroporation and then decreases with time, which explains the recovery of the skin barrier function. In this chapter, the new combination of microneedles and electroporation, the so-called in-skin electroporation (IN-SKIN EP), was developed to combine the advantages of microneedles and electroporation.
To date, iontophoresis and electroporation have shown great success in increasing skin permeation of many low molecular weight molecules. The transport of large molecules, however, remains a challenge (Abla et al. 2006). Since the microelectronics industry could fabricate uniform arrays of micron-scale needles, microneedle systems gained high interest as transdermal drug delivery systems. However, only few studies have reported on the combination of iontophoresis or electroporation with microneedles. Therefore, in the present chapter, the enhancement of skin permeation of high molecular weight compounds by a combination of microneedles and iontophoresis or electroporation is discussed (Wu et al. 2007).
24.3 The Synergistic Effect of Iontophoresis and Microneedles on the Skin Permeation of High Molecular Weight Compounds
Two model compounds with low and high molecular weight, deuterium oxide (D2O) and fluorescein isothiocyanate (FITC)-dextrans (FDs) (FD-4, FD-10, FD-40, FD-70, and FD-2000; average molecular weight of 4.3, 9.6, 42.0, 71.2, and 200.0 kDa, respectively), were used, and the effect of microneedle pretreatment and iontophoresis on their in vitro permeation was evaluated (Wu et.al. 2007).
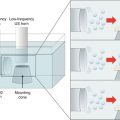
The following figure shows the modified diffusion cell according to Pikal and Shah (1990) and Kobatake et al. (1968) in order to directly measure the electroosmotic volume flow across the skin.
In vitro skin permeation experiment was performed in order to evaluate the synergistic effect of iontophoresis and microneedle system on the skin permeation profiles for of FDs and D2O. Hairless rat skin was excised and then mounted between two chambers of side-by-side diffusion cells (each 5.0 mL in volume and 0.95 cm2 in effective diffusion area) (Fig. 24.2) for iontophoresis, to conduct the permeation experiment. The stratum corneum side of the skin faced the drug donor chamber. FD-4, FD-10, FD-40, FD-70, or FD-2000 (1.0 mg/mL) in 1/30 M phosphate-buffered saline (PBS, pH 7.4) was added to the donor side, and PBS alone was added to the receiver side. In D2O permeation experiments, D2O was added to the donor cell instead of FD solution. After adding FDs or D2O on the stratum corneum side, the passive transport of FDs and D2O was first monitored for 5 h as a pre-iontophoretic period. Next a constant current of 0.29 mA (0.3 mA/cm2) was then delivered to the electrodes for the next 5 h, with the anode and cathode in the donor and receiver compartments, respectively, during the iontophoretic period. After termination of the current, post-iontophoretic passive transport was measured for a further 5 h.
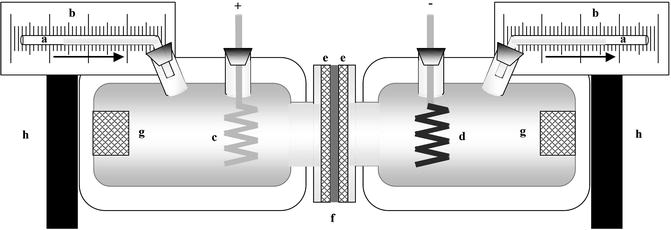

Fig. 24.2
Schematic illustration of diffusion cell used to measure solvent volume flow. (a) Capillary tube having an inner diameter of 1.0 mm, (b) calibrated paper, (c, d) Ag/AgCl electrodes (anode and cathode), (e) Teflon mesh of 0.5-mm thickness and several holes of 1.0-mm diameter, (f) excised hairless rat skin, (g) magnetic stirrer bar, (h) magnetic stirrer
The results of skin permeation of high and low molecular weight compounds obtained by the combination of microneedles and iontophoresis are shown in Figs. 24.3 and 24.4.
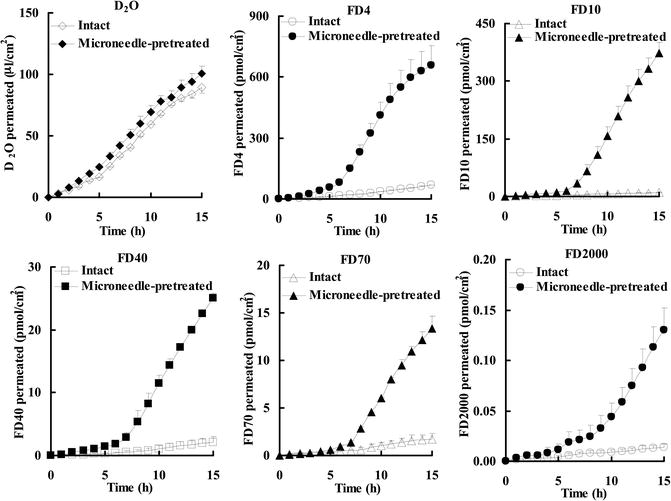
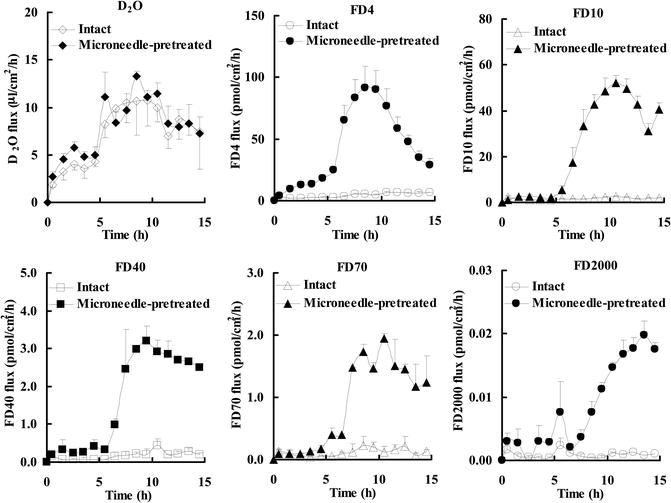

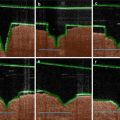
Fig. 24.3
Permeation of D2O and FDs through intact and microneedle-pretreated hairless rat skin before (0–5 h), during (5–10 h), and after (10–15 h) iontophoresis at 0.3 mA/cm2

Fig. 24.4
Flux of D2O and FDs through intact and microneedle-pretreated hairless rat skin before (0–5 h), during (5–10 h), and after (10–15 h) iontophoresis at 0.3 mA/cm2
The cumulative amount of FDs that permeated through microneedle-pretreated skin showed a special behavior pattern, i.e., long lag time, and the passive permeability of FDs was increased during post-iontophoresis duration. The lag time of FD-4, FD-10, FD-40, FD-70, and FD-2000 was 0.36 h, 1.66 h, 1.17 h, 1.18 h, and 2.82 h, respectively, which showed a tendency to being dependent on their molecular weight. Moreover, the combination of microneedle pretreatment and subsequent iontophoresis significantly enhanced FD flux compared with microneedle pretreatment alone or iontophoresis alone. In contrast, microneedle pretreatment of the stratum corneum did not further increase the cumulative amount and the flux of D2O. These results suggest that the combination of iontophoresis with microneedle pretreatment may be a useful means to further increase skin permeation of high molecular weight compounds.
24.4 The Synergistic Effect of Electroporation and Microneedles on the Skin Permeation of High Molecular Weight Compounds
A minimally invasive method was developed for the delivery of macromolecular drugs to the deep skin layers, the so-called in-skin electroporation (IN-SKIN EP). Each microneedle could serve as a microelectrode for electroporation, which forms an electric field inside the skin barrier. After the IN-SKIN EP application, drugs are administered to the skin surface to be delivered into the deeper skin layers. This method is different from conventional electroporation (ON-SKIN EP), where electrodes are applied just on the skin surface. IN-SKIN EP may facilitate the delivery of high molecular weight and hydrophilic compounds to the deep skin. Furthermore, it affects only a shallow region of the skin, not deep skin layers by the microneedle-electrode array; thus, IN-SKIN EP probably increases skin permeability but with low skin irritation. The microneedle-electrode array for IN-SKIN EP application is shown in Fig. 24.5. Positive and negative electrodes were alternately arranged on the silicon sheet with 4.0-mm spacing, as shown in Fig. 24.5b. Each needle was 400-μm long with a 28° angle beveled tip, and the base diameter of the needle was approximately 200 μm, as shown in Fig. 24.5c. Figure 24.5d shows a scanning electron microscope (SEM) image of one microneedle tip. In order to compare the effect of IN-SKIN EP with conventional ON-SKIN EP, the needle tips of the microneedle array were filed and struck with a file and hammer to make them obtuse, as shown in Fig. 24.5e. This obtuse tip needle array was used to apply electroporation at various points on the skin surface for ON-SKIN EP (where the microneedle electrodes were applied on skin), which was the control experiment to evaluate the IN-SKIN EP.