FIGURE 4.1 Absolute detection thresholds for sinusoidal stimuli, where it can be seen that as the vibration frequency increases, the detection thresholds decrease. (Reproduced from Bolanowski, S. J., Gescheider, G. A., Verrillo, R. T., and Checkosky, C. M., J Acoustic Soc Am, 84, 1680–1694, 1988, with the permission of the Acoustical Society of America.)
Psychophysical procedures have been traditionally employed to study the sense of touch, and as in hearing research where the sensory receptor is another type of specialized mechanoreceptor, different frequencies of vibration are used to quantify the response properties of this sensory system. Von Bekesy (7) was the first to use vibratory stimuli as an extension of his research interests in audition. In a typical experiment, participants are asked to respond with a simple button press when they can just detect the presence of a vibration presented to a digit within one of the two time periods. This two-alternative forced choice paradigm provides a threshold-tuning curve, the slopes of which provide information about a particular class of the response properties of LTMs. As can be seen from Figure 4.1, a U-shaped function is generated, with increasingly lower detection thresholds being measured as vibrotactile frequency increases to a peak at around 300 Hz, at which point the curve begins to increase again as sensitivity decreases.
By carefully controlling the spatial configuration of the vibrating probe (i.e., its diameter and the gap between it and a static surround), the vibratory frequency, the amplitude, the stimulus duration, the skin surface temperature, and the use of various masking techniques, Verrillo et al. (8,9–12) proposed that there are four distinct psychophysical channels mediating tactile perception in the glabrous skin of the hand. This model proposes that each psychophysically determined channel is represented by one of the four anatomical end organs and nerve fiber subtypes with frequencies in the 40–500 Hz range providing a sense of vibration, transmitted by Pacinian corpuscles (Pacinian corpuscle channel or fast-adapting I [FAI]), Meissner’s corpuscles being responsible for the sense of flutter in the 2–40 Hz range (non-Pacinian I [NPI] channel or fast-adapting II [FAII]), the sense of pressure being mediated by Merkel’s discs in the 0.4–2.0 Hz range (non-Pacinian III [NPIII] or slowly adapting I [SAI]), and Ruffini end organs producing a buzzing sensation in the 100–500 Hz range (non-Pacinian III [NPII] or slowly adapting II [SAII]). Neurophysiological studies have by and large supported this model, but there is still some way to go to link the anatomy with perception (see Table 4.1 for a summary of the properties of these LTMs).
There have been relatively few studies of tactile sensitivity on hairy skin, the cat being the animal of choice for most of these studies. Mechanoreceptive afferents (Aβ fibers) have been described that are analogous to those found in human glabrous skin (FAI, FAII, SAI, and SAII), and Essick and Edin (13) have described sensory fibers with these properties in human facial skin. The relationship between these sensory fibers and tactile perception is still uncertain, and this is exemplified by the response properties of SAI afferents. Harrington and Merzenich (14) found that these afferents are responsive to levels of stimulation that are below perceptual thresholds, and Jarvilehto et al. (15) describe high levels of activity in human hairy skin SAIs that are not perceivable, in contrast to the responses of this class of afferent in glabrous skin where SAI nerve activity is directly correlated with a sense of pressure.
Sensory axons are classified according to their degree of myelination, the fatty sheath that surrounds the nerve fiber. The degree of myelination determines the speed with which the axon can conduct nerve impulses and, hence, the conduction velocity (CV) of the nerves. The largest and fastest axons are called A-α and include some of the proprioceptive neurons, such as the muscle stretch receptors. The second largest group, called A-β, includes all the discriminative touch receptors being described here. Pain and temperature include the third and fourth groups, A-δ and C fibers, and will be dealt with in the section “Temperature” (Table 4.1).
Electrophysiological studies by Vallbo and Johansson (16), on single peripheral nerve fibers innervating the human hand, have provided a generally accepted model of touch that relates the four anatomically defined types of cutaneous or subcutaneous sense organs to their neural response patterns.
TABLE 4.1
Main Characteristics of Primary Sensory Afferents Innervating Human Skin
Class | Modality | Axonal Diameter (μm) | CV (m/s) |
Myelinated | |||
A-α | Proprioceptors from muscles and tendons | 20 | 120 |
A-β | LTMs | 10 | 80 |
A-δ | Cold, noxious, thermal | 2.5 | 12 |
Unmyelinated | |||
C-pain | Noxious, heat, thermal | 1 | <1 |
C-tactile | Light stroking, gentle touch | 1 | <1 |
C-autonomic | Autonomic, sweat glands, vasculature | 1 | <1 |
The technique that they employed and developed is called microneurography and involves inserting a fine tungsten microelectrode, with tip diameter of <5 μm, through the skin of the wrist and into the underlying median nerve which innervates the thumb and the first two digits. A sensitive biological amplifier records and amplifies the spike discharges conveyed by the axons and feeds these to a loudspeaker to enable the experimenter to hear the spike activity and home-in on a single unit. The skilled manual micromanipulation of the electrode, coupled with stroking across the hand to stimulate LTMs, first results in a population response being recorded, that is, neural activity in a nerve fascicle containing hundreds of peripheral axons until finally, sometimes after many hours, a single axon is isolated. At this stage, the RF of the single unit is mapped with a Von Frey hair and the unit subtype (i.e., FA or SA) identified. Once this stage is completed, a small pulsed current of a few microamperes (typically <10 μA) is delivered to the nerve that provides a final, perceptual confirmation of the unit subtype.
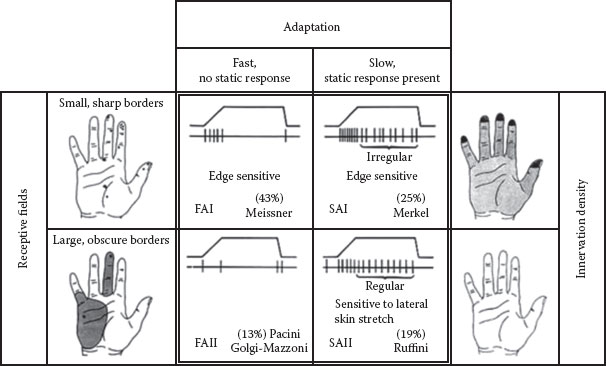
FIGURE 4.2 The four types of LTMs in human glabrous skin are depicted. The four panels in the center show the nerve-firing responses to a ramp-and-hold indentation, and in percentage, the frequency of occurrence and putative morphological correlate. The black dots in the left panel show the RFs of type 1 (top) and type 2 (bottom) afferents. The right panel shows the average density of type 1 (top) and type 2 (bottom) afferents with darker area depicting higher densities. Abbreviations: LTM: low-threshold mechanoreceptor; RF: receptive field. (After Westling, G. K., Sensori-Motor Mechanisms during Precision Grip in Man, Medical dissertation, Umea University, Sweden, 1986.)
If, for example, an RA unit has been isolated, microstimulation is perceived as a flutter or vibration, depending on the frequency of the electrical pulses, and is perceptually localized to the previously mapped RF. Figure 4.2 depicts the relationships between RF, adaptation rate, and unit type from studies carried out on the human hand (17).
Temperature
The cutaneous somatosensory system detects changes in ambient temperature over an impressive range, initiated when thermal stimuli that differ from a homeostatic set point excite temperature-specific sensory nerves in the skin, and relays this information to the spinal cord and brain. It is important to recognize that these nerves code for temperature change, not absolute temperature, as a thermometer does. The system does not have specialized receptor end organs, such as those found in LTMs, but uses free nerve endings throughout the skin to sense changes in temperature. Within the innocuous thermalsensing range, there are two populations of thermosensory fibers, one that responds to warmth (warm receptors) and one that responds to cold (cold receptors), and include fibers from the A-δ and C ranges. Specific cutaneous cold and warm receptors have been defined as slowly conducting units that exhibit a steady-state discharge at constant skin temperature and a dynamic response to temperature changes (18,19). Cold- and warm-specific receptors can be distinguished from nociceptors that respond to noxious low and high temperatures (<20°C and >45°C) (20,21) and also from thermosensitive mechanoreceptors (18,22). Konietzny (22) recorded from 13 cold-specific units in humans employing the microneurography technique and measured CVs which were in the C fiber range (0.43–2.04 m/s).
Serra et al. (23) reported a number of spontaneously active fibers employing microneurography, which were sensitive to small temperature changes and that were described as cold-specific units, but all had CVs in the C fiber range (0.43–1.27 m/s). Standard medical textbooks describe the cutaneous cold sense in humans as being mediated by myelinated A fibers with CVs in the range of 12–30 m/s (24), but a work from Campero et al. (25) concludes that either human cold-specific afferent fibers are incompletely myelinated BC fibers, described by Duclaux et al. (26) as having electrophysiological and morphological properties of C fibers in their distal part and B fibers in their proximal part, or there are C as well as A cold fibers, with the C fiber group contributing little to sensation. For example, the resting discharge at room temperature (21°C) is characterized by a low-frequency discharge (~1 Hz), and this steady-state activity is suppressed by sudden warming of the RF and increased by cooling the RF.
The free nerve endings for cold- or warm-sensitive nerve fibers are located just beneath the skin surface. The terminals of an individual temperature-sensitive fiber do not branch profusely or widely. Rather, the endings of each fiber form a small, discretely sensitive point, which is separate from the sensitive points of neighboring fibers. The total area of the skin occupied by the receptor endings of a single temperature-sensitive nerve fiber is relatively small (1 mm in diameter) with the density of these thermosensitive points varying in different body regions. For example, there are up to 15–25 cold points per square centimeter in the lips, 3–5 cold points per square centimeter in the finger, and less than 1 cold point per square centimeter in some broad areas of the trunk. There are 3–10 times as many cold-sensitive points as warm-sensitive points in most areas of the body.
It is well established from physiological and psychological testing that warm- and cold-sensitive nerve fibers are distinctively different from one another in both structure and function.
Pain
Here, we consider a system of peripheral sensory nerves that innervate all cutaneous structures and whose sole purpose is to protect the skin against potential or actual damage. These primary afferents include A-δ and C fibers which respond selectively and linearly to the levels of thermal, mechanical, and chemical intensities/strengths that are tissue threatening, that is, having the potential to damage the skin. This initial encoding mechanism is termed nociception and describes the sensory process detecting any overt, or impending, tissue damage. The term pain, on the other hand, describes the perception of irritation, stinging, burning, soreness, or painful sensations arising from the skin.
It is important to recognize, especially when we are investigating an area such as sensitive skin, that the perception of pain depends not only on nociceptor inputs, but also on other processes and pathways giving information about, for example, emotional or contextual components. Pain is, therefore, described in terms of an experience rather than just a simple sensation. There are again submodalities within the nociceptive system which, at the peripheral anatomical level, are evident with respect to the degree of myelination of the nerve fibers (A-δ and C) subserving nociception (Table 4.2). A-δ fibers are thin (1–5 μm), poorly myelinated axons of mechanical nociceptors, thermal receptors, and mechanoreceptors with axon potential CVs averaging 12 m/s, and C fibers are very thin (<1 mm) slowly conducting axons (<1 m/s). Mechanical nociceptors are in the A-δ range and possess RFs distributed as 5–20 small sensitive spots over an area approximately 2–3 mm in diameter. In many cases, the activation of these spots depends upon stimuli intense enough to produce tissue damage, such as a pinprick. A-δ units with a short latency response to intense thermal stimulation in the range of 40–50°C have been described as well as other units excited by heat after a long latency—usually with thresholds in excess of 50°C. Over 50% of the unmyelinated axons (C fibers) of a peripheral nerve respond not only to intense mechanical stimulation, but also to heat and noxious chemicals, and are therefore classified as polymodal nociceptors (27) or C-mechanoheat (CMH) nociceptors (28). A subgroup of polymodal nociceptors has been reported to respond to extreme cold; however, many of these units develop an excitatory response to cooling after prior exposure to noxious heat. A small number of C fibers have mechanical thresholds in the nociceptor range with no response to heat, whereas others have been found to respond preferentially to noxious heating. RFs consist of single zones with distinct borders, and in this respect, they differ from A-δ nociceptors that have multipoint fields. Innervation densities are high, and responses have been reported to a number of irritant chemicals such as dilute acids, histamine, bradykinin, and capsaicin. By employing microneurography, Schmidt et al. (29) described not only CMH-responsive units, but also a novel class of C nociceptors responding only to mechanical stimuli (CM), units responding only to heating (CH), and units that were insensitive to mechanical and heating stimuli and also to sympathetic provocation tests (CMiCHi). Of relevance here is that some CM, CH, and CMiCHi units were sensitized to thermal and/or mechanical stimuli after topical application of skin irritants such as mustard oil or capsaicin; these units then acquired responsiveness to stimuli to which they were previously unresponsive. The recruitment of these silent nociceptors implies spatial summation to the nociceptive afferent barrage at central levels and may, therefore, contribute to primary hyperalgesia after chemical irritation and to secondary hyperalgesia as a consequence of central sensitization (detailed subsequently).
TABLE 4.2
Major Findings by Bolanowski et al. (8) and Previous Work Done by These Researchers at the Institute for Sensory Research at Syracuse University
Channel | Pacinian | NPI | NPII | NPIII |
Frequency response (Hz) | 40–80 | 3–100 | 15–400 | <0.3 to >100 |
Threshold (at 1 μm) | <−20 dB at 300 Hz | 28 dB at 3 Hz | 10 dB at 300 Hz | 28 dB at 3 Hz |
Sensation | Vibration | Flutter | Not known | Pressure |
Temporal summation | Yes | No | Yes | No |
Spatial summation | Yes | No | Not known | No |
Receptor type | FAI Pacinian corpuscle | FAII Meissner’s corpuscle | SAII Ruffini end organ | SAI Merkel’s disk |
Source: Bolanowski, S. J., Gescheider, G. A., Verrillo, R. T., and Checkosky, C. M., J Acoustic Soc Am, 84, 1680–1694, 1988; Gescheider, G. A., O’Malley, M. J., and Verrillo, R. T., J Acoustic Soc Am, 74, 474–485, 1983; Gescheider, G. A., Sklar, B. F., Van Doren, C. L., and Verrillo, R. T., J Acoust Soc Am, 78, 534–543, 1985; Gescheider, G. A., Verrillo, R. T., and Van Doren, C. L., J Acoustic Soc Am, 72, 1421–1426, 1982; Verrillo, R. T., J Acoustic Soc Am, 35, 1962–1966, 1963.
Nociceptors do not show the kinds of adaptation response found with rapidly adapting LTMs (i.e., they fire continuously to tissue damage), but pain sensation may come and go, and pain may be felt in the absence of any nociceptor discharge. They rely on chemical mediators around the nerve ending, which are released from nerve terminals and skin cells in response to tissue damage. Koltenzenburg et al. (30
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree