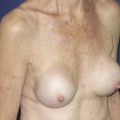
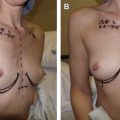
Breast augmentation remains one of the most common procedures performed in the United States. However, shape, feel, safety, and longevity of the implants remain important areas of research. The data provided by manufacturers show the safety and efficacy of these medical devices. Clinicians should strive to provide ongoing data and sound science to continue to improve clinical outcomes in the future. This article explores the evolution of breast implants with special emphasis on the advancement of silicone implants.
Key points
- •
Implant characteristics on the bench differ from implant performance within the body. Shape, feel, safety, and longevity of the implants remain important areas of research.
- •
The data provided by all 3 manufacturers show the safety and efficacy of these medical devices.
- •
Clinicians should strive to provide ongoing data and sound science to continue to improve clinical outcomes in the future.
Introduction
There has been a steady increase in breast augmentation surgery with the evolving importance of body image, changes in societal expectations, and the increasing acceptance of aesthetic surgery in the United States. Augmentation mammaplasty, performed 286,694 times in 2014, ranks as the most frequently performed cosmetic surgical procedure in women in the United States.
The first report of successful breast augmentation appeared in 1895, in which Czerny described transplanting a lipoma from the trunk to the breast in a patient deformed by a partial mastectomy. In 1954, Longacre described a local dermal-fat flap for augmentation of the breast. Eventually, both adipose tissue and omentum were also used to augment the breast.
During the 1950s and 1960s, breast augmentation with solid alloplastic materials was performed using polyurethane, polytetrafluoroethylene (Teflon), and expanded polyvinyl alcohol formaldehyde (Ivalon sponge). Ultimately, the use of these materials was discontinued after patients developed local tissue reactions, firmness, distortion of the breast, and significant discomfort. Various other solid and semisolid materials have been injected directly into the breast parenchyma for augmentation, including epoxy resin, shellac, beeswax, paraffin, petroleum jelly, and liquid silicone. In 1961, Uchida reported the injection of liquid silicone (polydimethylsiloxane) into the breast for breast augmentation. This technique resulted in frequent complications, including recurrent infections, chronic inflammation, drainage, granuloma formation, and even necrosis. Breast augmentation by injection of free liquid silicone and the various other solid and semisolid materials was abandoned in the United States in light of these complications.
The evolution of the modern breast implant began with a 2-component prosthetic device manufactured with a less permeable silicone elastomer shell filled with a stable filling material, consisting of either saline solution or silicone gel. This shell and gel filler implant was originally developed by Cronin and Gerow in 1962 using silicone gel as the filling material contained within a thin, smooth silicone elastomer shell. Since that time, both silicone gel and saline-filled implants have undergone several technical alterations and improvements.
Introduction
There has been a steady increase in breast augmentation surgery with the evolving importance of body image, changes in societal expectations, and the increasing acceptance of aesthetic surgery in the United States. Augmentation mammaplasty, performed 286,694 times in 2014, ranks as the most frequently performed cosmetic surgical procedure in women in the United States.
The first report of successful breast augmentation appeared in 1895, in which Czerny described transplanting a lipoma from the trunk to the breast in a patient deformed by a partial mastectomy. In 1954, Longacre described a local dermal-fat flap for augmentation of the breast. Eventually, both adipose tissue and omentum were also used to augment the breast.
During the 1950s and 1960s, breast augmentation with solid alloplastic materials was performed using polyurethane, polytetrafluoroethylene (Teflon), and expanded polyvinyl alcohol formaldehyde (Ivalon sponge). Ultimately, the use of these materials was discontinued after patients developed local tissue reactions, firmness, distortion of the breast, and significant discomfort. Various other solid and semisolid materials have been injected directly into the breast parenchyma for augmentation, including epoxy resin, shellac, beeswax, paraffin, petroleum jelly, and liquid silicone. In 1961, Uchida reported the injection of liquid silicone (polydimethylsiloxane) into the breast for breast augmentation. This technique resulted in frequent complications, including recurrent infections, chronic inflammation, drainage, granuloma formation, and even necrosis. Breast augmentation by injection of free liquid silicone and the various other solid and semisolid materials was abandoned in the United States in light of these complications.
The evolution of the modern breast implant began with a 2-component prosthetic device manufactured with a less permeable silicone elastomer shell filled with a stable filling material, consisting of either saline solution or silicone gel. This shell and gel filler implant was originally developed by Cronin and Gerow in 1962 using silicone gel as the filling material contained within a thin, smooth silicone elastomer shell. Since that time, both silicone gel and saline-filled implants have undergone several technical alterations and improvements.
Evolution of saline implants
The use of inflatable saline-filled breast implants was first reported in 1965 by Arion in France. The saline-filled implant was developed in order to allow the noninflated implant to be introduced through a small incision, and then the implant was inflated in situ.
Although these implants allow slight overfilling, aggressive overfilling may lead to a more spherical shape and scalloping along the implant edge, with knucklelike palpability and unnatural firmness. A disadvantage of saline-filled implants is that the consistency on palpation is similar to that of water instead of the more viscous feel of natural breast tissue.
Evolution of silicone implants
The first-generation silicone gel–filled implant introduced in 1962 by Cronin and Gerow was manufactured by the Dow Corning Corporation. The shell of the first-generation implant was constructed using a thick, smooth silicone elastomer as a 2-piece envelope with a seam along the periphery. The shell was filled with a moderately viscous silicone gel. The implant was anatomically shaped (teardrop) and had several Dacron fixation patches on the posterior aspect to help maintain the proper position of the implant. These early devices had a high contracture rate, caused by the quality of the shells and the lack of cohesivity of the gel, which then encouraged implant manufacturers to develop second-generation silicone gel–filled implants.
In the 1970s, the second-generation silicone implants were developed in an effort to reduce the incidence of capsular contracture with a thinner, seamless shell and without Dacron patches incorporated into the shell. These implants were round and filled with a less viscous silicone gel to provide a more natural feel. However, the second-generation breast implants had problems with diffusion or bleed of microscopic silicone molecules into the periprosthetic intracapsular space because of their thin, permeable shells and low-viscosity silicone gel filler. This diffused silicone produced an oily, sticky residue surrounding the implant within the periprosthetic capsule, which was noticeable during explantation of older silicone-filled implants.
The development of the third-generation silicone gel–filled implants in the 1980s focused on improving the strength and permeability of the shell in order to reduce silicone gel bleed from intact implants, and to reduce implant rupture and subsequent gel migration. The manufacturers designed new implant shells that consisted of multilayered silicone elastomer. These third-generation prostheses reduced gel bleed by introducing a barrier layer and a thicker shell, which significantly reduced the device shell failure rate.
After the US Food and Drug Administration (FDA) required the temporary restriction of third-generation silicone gel implants from the American market in 1992, the fourth-generation and fifth-generation gel devices evolved. These silicone gel breast implants were designed under more stringent American Society for Testing Methodology and FDA-influenced criteria for shell thickness and gel cohesiveness. Furthermore, they were manufactured with improved quality control, and with a wider variety of surface textures and implant shapes. They are currently available from all 3 breast implant manufactures in the United States (Sientra, Allergan, and Mentor) ( Figs. 1–3 ).

During the same time the concept of anatomically shaped implants was introduced with the fifth-generation silicone gel implants. In addition to having a textured surface, these anatomically shaped implants are filled with a more cohesive gel. The FDA approved fifth-generation implants from all of the US manufacturers in the following order: Sientra (2012), then Allergan and Mentor (both 2013). Each manufacturer was approved for a variety of shapes and styles, with Sientra offering 5 styles of the HSC+ line, 4 styles of the 410 implant from Allergan, and 1 CPG implant from Mentor.
To further understand the evolution of silicone-filled implants, implant characteristics are further reviewed, because the resultant breast form not only depends on the soft tissue envelope (in augmentation and reconstruction) and the breast parenchyma (in augmentation) but also on the following implant characteristics: surface, filler, shell, and implant shape.
Surface
Surface characteristics have undergone changes and have evolved with all 3 manufacturers working toward the common goal of using texture to possibly minimize or even disrupt capsule formation. The evolution of textured implants began with polyurethane-coated implants reportedly having lower capsular contracture rates. These foam-coated implants were eventually removed voluntarily from the US market because of concern caused by difficulty in complete removal and theoretic concern of carcinogenic conversion of the coating. Polyurethane foams are thought to undergo partial chemical degradation under physiologic conditions, releasing compounds that could become carcinogens in animals but are not known human carcinogens.
In the 1980s, manufacturers shifted their focus from foam-covered shells to textured silicone shells with different pore sizes. None of the textured surfaces are created in the same manner and each manufacturer has a proprietary process in place. One of the critical issues during the evolution of the texture is to find a way to stabilize the implant in the breast pocket. Studies have shown that the pore size is critical to allow for tissue adherence leading to the adhesive effect and implant stabilization. However, it was not clear whether the pore size correlated with a reduction in capsular contracture, but did correlate with implant stabilization. Danino and colleagues compared the BIOCELL texture with pore diameter of 600 to 800 μm with a depth of 150 to 200 μm with Siltex with pore diameter of 70 to 150 μm. Siltex pores led to no adhesive effect.
The manufacturing process of textured surface implants can be complex, whereas smooth surface implants are made by dipping a mandrel into liquid silicone, creating multiple layers, followed by allowing the surface to cure in a laminar flow oven. Additional steps beyond creating smooth surface implants are involved in the creation of textured implants. Sientra’s Silimed implant (Sientra, Inc, Santa Barbara, CA), named as TRUE Texture, avoids the use of sodium chloride, sugar, soak/scrub, or pressure stamping. Small, hollow pores are formed with minimal thin cell webbing that reduces particle formation. The BIOCELL (Allergan, Inc, Irvine, CA) texture is created using a loss-salt technique, which includes a layer of salt crystals with a thin overcoat of silicone followed by curing in a laminar flow oven. In contrast, the Siltex surface (Mentor Corp, Santa Barbara, CA) is created by imprint stamping, which dips the chuck into uncured silicone, pushing it into polyurethane foam and finalizing the imprint with pressure.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree