Repigmentation in vitiligo is the process that replaces, in the epidermal basal layer of vitiligo skin, the mature melanocytes that have been killed by cytotoxic T cells specific for melanocyte antigens. It consists of mobilization of melanocyte precursors in the hair follicle bulge and infundibulum to proliferate, migrate, and differentiate into mature melanocytes, moving from the hair follicle bulge to the interfollicular epidermis. The most common clinical presentation of repigmentation in vitiligo is the perifollicular pattern. The most potent stimulus for repigmentation is the UV light.
Key points
- •
Repigmentation is an active process during epidermal crisis reversing the loss of epidermal melanocytes.
- •
It usually develops in hair-bearing areas.
- •
The most common clinical presentation is the perifollicular pattern.
- •
The initiating event is the activation of melanocyte precursors from the hair follicle and of immature melanocytes from the basal epidermis.
- •
It is induced by different stimuli: UV light, drugs (steroids, calcineurin inhibitors).
Introduction
The loss of epidermal mature melanocytes in vitiligo depends on melanocyte-specific CD8+ cytotoxic T lymphocytes. It is reversed by halting the immune attack and by activating melanocyte precursors in the bulge and hair follicle infundibulum, to proliferate, migrate, and differentiate through the process called repigmentation. Although repigmentation refers to the replenishment of pigment cells only, keratinocytes in vitiligo skin demonstrate architectural abnormalities and are also likely to be directly involved in repigmentation. Changes of the keratinocytes architecture seem to appear in the absence of basal melanocytes, in the sun-exposed skin. Therefore, significant increase in thickness of both stratum corneum and viable epidermis in vitiligo-depigmented skin, as compared with the adjacent, normal-appearing skin, was reported. This increase likely occurs as an adaptive response to lack of melanin that can minimize and counteract the harmful UV effects on the skin.
Based on current knowledge, vitiligo repigmentation depends on available melanocytes from 2 sources:
- •
The hair follicle, which is the main source of pigment cells and is often unaffected by the T cell–mediated attack, likely because the hair follicle bulge is an immune privileged location
- •
The epidermis at the lesional borders, which contains a pool of functional melanocytes and represents a secondary source for repigmentation
Melanocyte activation, followed by migration, proliferation, and differentiation is triggered by several stimuli, such as UV radiation (delivered as treatment or by natural sunlight) and drugs (systemic and topical steroids and topical calcineurin inhibitors).
Introduction
The loss of epidermal mature melanocytes in vitiligo depends on melanocyte-specific CD8+ cytotoxic T lymphocytes. It is reversed by halting the immune attack and by activating melanocyte precursors in the bulge and hair follicle infundibulum, to proliferate, migrate, and differentiate through the process called repigmentation. Although repigmentation refers to the replenishment of pigment cells only, keratinocytes in vitiligo skin demonstrate architectural abnormalities and are also likely to be directly involved in repigmentation. Changes of the keratinocytes architecture seem to appear in the absence of basal melanocytes, in the sun-exposed skin. Therefore, significant increase in thickness of both stratum corneum and viable epidermis in vitiligo-depigmented skin, as compared with the adjacent, normal-appearing skin, was reported. This increase likely occurs as an adaptive response to lack of melanin that can minimize and counteract the harmful UV effects on the skin.
Based on current knowledge, vitiligo repigmentation depends on available melanocytes from 2 sources:
- •
The hair follicle, which is the main source of pigment cells and is often unaffected by the T cell–mediated attack, likely because the hair follicle bulge is an immune privileged location
- •
The epidermis at the lesional borders, which contains a pool of functional melanocytes and represents a secondary source for repigmentation
Melanocyte activation, followed by migration, proliferation, and differentiation is triggered by several stimuli, such as UV radiation (delivered as treatment or by natural sunlight) and drugs (systemic and topical steroids and topical calcineurin inhibitors).
Clinical patterns of repigmentation and the repigmentation sources
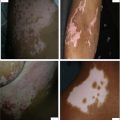
There are 4 classic repigmentation patterns observed on clinical examination : perifollicular (most common) ( Fig. 1 A-i ), marginal (see Fig. 1 A-ii), diffuse (see Fig. 1 A-iv), and combined, which includes more than one pattern (an example of marginal combined with perifollicular pattern is provided in Fig. 1 A-v). A fifth newly described repigmentation pattern, the medium-spotted patern, is presented in Fig. 1 A-vi.
The perifollicular pattern presents as small, round, pigmented macules around the hair follicles (see Fig. 1 A-i). This clinical observation was confirmed by numerous previous in vivo studies that identified amelanotic, inactive, 3,4-dihydroxy-L-phenylalanine (DOPA)(−) melanocytes in the infundibulum outer root sheath of hair follicles collected from healthy individuals or from vitiligo patients. The origin of DOPA(−) melanocytes was later identified as the hair follicle bulge (both in the transgenic mouse model and then in human skin). From this location melanocyte precursors ascend to repopulate the depigmented epidermis ( Fig. 1 B, right side) in the ultraviolet radiation (UVR)-treated vitiligo. Activation by UV treatment or ionizing radiation induces these precursors to migrate, proliferate, and differentiate, finally expressing the full pigmentation pathway in the interfollicular epidermis. Data confirming the follicular reservoir also came from animal and human models of pigmentation, which is revisited in this review.
The marginal pattern is observed as a repigmentation rim at the borders of the lesions (see Fig. 1 A-ii). It is the result of activation of functional epidermal melanocytes in the lesional borders. Some investigators have proposed an epidermal repigmentation reservoir, consisting of more immature melanocyte precursors with supposed migratory, differentiation, and perhaps proliferative abilities (see Fig. 1 B, left side).
The diffuse repigmentation pattern appears as generalized darkening across the patches of vitiligo (see Fig. 1 A-iv), whereas, in the combined pattern (see Fig. 1 A-v), the repigmentation does not fit into any single type or where more than one pattern contributes to the repigmentation process. The diffuse pattern suggests that repigmentation can arise from interfollicular melanocyte precursors either in the dermis or interfollicular epidermis (by reactivation of DOPA(−) melanocytes, which are hypothesized to persist in the center of the lesions).
A very recent study reported in pediatric vitiligo patients a medium-spotted repigmentation pattern, located in non–hair-bearing to minimal hair-bearing sites, such as the palms, soles, lips, ankles, and anterior wrists. This uncommon pattern begins as larger spots that are not centered on any particular hair follicle. Consistent with these findings, a previous study reported a rare pattern of repigmentation on the palms of a patient, consisting of irregular brown macules developed after a course of psoralen plus UVA (PUVA) therapy (see Fig. 1 A-vi). Tyrosinase (TYR)(+) melanocytes were found along the basement membrane of the repigmenting lesions, in contrast to the adjacent vitiliginous skin, which lacked these cells. Based on observation of repigmentation that occurred in the center of an initially fully depigmented lesion on the palm (a region devoid of hair follicles), the investigators hypothesized that the melanocyte precursors/stem cells can remain in vitiliginous lesions serving as repigmentation reservoir. The pattern seen in this case seems to support medium-spotted repigmentation, recently reported in children with vitiligo.
Other reports identified melanocytes within the depigmented vitiligo, and one of them claimed that melanocytes in depigmented epidermis were “never completely absent.” The investigators hypothesized that these melanocytes can recover their functionality in vivo and in vitro on the removal of hydrogen peroxide, which they propose is the major mediator of cytotoxicity.
Interestingly, in some of the frozen epidermal sections of depigmented untreated skin collected from vitiligo patients of skin types II and III, the authors’ group observed large cells, with fragmented bodies, and diminished and abnormal, fragmented dendrites, expressing a Premelanosome protein (PMEL)(+)/Tyrosine kinase receptor (C-KIT)(−) phenotype ( Fig. 2 A). The authors noticed similar aspects in paraffin sections of depigmented skin, in which epidermal cells carried a dopachrome tautomerase (DCT)(+)/C-KIT(−) phenotype (see Fig. 2 B). These rare cells were amelanotic (Fontana-Masson(−) staining, see Fig. 2 B) and located not only close to lesional borders but also within the lesions. It was previously proposed that these cells might be residual nonfunctional, senescent cells (progressing toward apoptosis) rather than poorly differentiated melanocyte precursors with potential to form functional melanocytes. Loss of C-KIT expression is an interesting and expected finding, knowing the major implication of stem cell factor (SCF)/c-kit in melanocyte survival and migration. The authors think that acquisition of C-KIT expression is essential in the repigmentation process associated with the migratory, proliferative, and differentiating phenotype of post–stem cell melanoblasts.
Based on indirect evidence of stem cell markers, tissue culture studies, and repigmentation patterns observed in patients with vitiligo, another previous report hypothesized the existence of a pool of extrafollicular melanocyte stem cells in a well-protected area of the dermis with the ability to replace any damaged melanocytes in the basal layer of the epidermis. More recently, a dermal source of melanoblasts (DCT(+)) was identified in the secretory portion of the eccrine sweat glands after skin exposure to ionizing radiation. It seems that the precursors of these cells colonize sweat glands during development, being maintained in an immature, slow-cycling state; they were shown to renew themselves in response to genomic stress (eg, ionizing radiation) having the capacity to provide their differentiating progeny to the epidermis. It has been hypothesized that these melanoblasts can provide an anatomic niche for melanocyte-melanoma precursor cells ; however, their implication in regeneration of vitiligo epidermis awaits investigation.
Clinical particularities of repigmentation and their correlation with cellular and molecular changes
The Lag Time Between Initiation of Stimulus (Ultraviolet) and Visible Repigmentation
Repigmentation is unpredictable, not proportional to the magnitude of the lesions, and often cosmetically insufficient. Curiously, in the same patient, vitiligo repigmentation in some regions can commonly parallel active depigmentation of other regions. Neither sex nor skin type are associated with differences in onset or pace of repigmentation following UVR treatment of vitiligo. Patients’ clinical response to narrow band UVB (NBUVB) exposure, applied twice weekly, has a lag time ranging between 4 weeks and 4 months, variable between body areas and from patient to patient. This lag time may depend on the integrity of the bulge stem cell reservoir, on the melanocyte precursors susceptibility to activation, and on their migratory and proliferative abilities. If there is a lack of any visible repigmentation after 6 months of treatment, further therapy is discouraged, in both adult and pediatric patients, and consideration of a different alternative can be advised.
Interestingly, in animal models, the cellular response to UVB seems to appear shortly after exposure. Therefore, in the C57BL/6 mouse, an increase of Tyrosinase protein 1 (TRP1)(+) epidermal melanocytes was first identified on the 5th day after UVB (one exposure of 0.18 J/cm 2 of energy, corresponding to 1.5 minimal erythema dose for the C57BL mice) and reached a cellularity 4 times as great as that of the normal control on the 14th day.
Repigmenting Versus Treatment-Resistant Lesions
- •
Repigmentation develops best in the hair-bearing regions. Areas with a higher density of hair follicles (face, arms, forearms, thighs, legs, abdomen, back) respond more rapidly to treatment, and those with lower density (dorsum of hands, fingers, feet, and toes) respond more slowly.
- •
Depigmented areas where hair follicles are absent or in low density (palms, soles, volar wrists, genital sites, mucosal or semimucosal surfaces) rarely respond to treatment; the response, if present, is slow and incomplete. The potential treatment response relies on the epidermal source of melanocytes from the lesional borders (that can migrate ∼ 4–5 mm into the depigmented area), on epidermal melanocytes that are hypothesized to persist in the center of the depigmented lesions, or on melanocytes with extrafollicular dermal origin.
- •
Depigmented areas with white terminal hairs (leukotrichia) are poor responders to medical treatment with minimal chances of repigmentation. Leukotrichia is perhaps an indicator of more severe, long-lasting, and active CD8+ T-cell immune-mediated attack on melanocytes, which progresses gradually downward, from the epidermis to the bulge. Depletion of the bulge melanocyte stem cells leads to exhaustion of secondary hair germ in the bulb, which is composed of melanocytes precursors more committed to melanocyte differentiation.
Cellular and Molecular Changes of Repigmented Skin
The sequences of repigmentation process
There has been surprisingly limited research addressing the repigmentation process in vitiligo, despite impressive progress studying the mechanisms of vitiligo immunology and genetics. It is known that the UV-induced repigmentation process includes keratinocyte stimulation and melanocyte activation by UV light ( Fig. 3 A) melanocyte migration, proliferation, and differentiation (differentiation implies cell melanization). Melanocyte migration involves the following steps: melanocyte decoupling from the basement membrane and from keratinocytes, cell movement, and recoupling to the basement membrane and to keratinocytes. The intimate melanocyte-keratinocyte anatomic and functional interactions in the hair follicle and epidermis are essential for epidermal repopulation in vitiligo. However, how these processes develop during repigmentation and the triggers that initiate one process in favor of the other have not yet been identified.
There are 2 major pathways, p53 and Wnt/β-catenin, that strongly activate the pigmentation process. It has been clearly shown that cellular and DNA damage by UVR induces p53 activation. In normal skin repeatedly exposed to UVR, p53 orchestrates melanocyte activation by coordinating the release of keratinocyte-paracrine/growth factors with melanogenic activity, which induces microphthalmia-associated transcription factor (MITF) expression in melanocytes (see Fig. 3 A). Similarly, in vitiligo, repigmentation occurs following melanocyte activation by NBUVB orchestrated by p53 and its downstream effector, α-melanocyte stimulating hormone, which is a potent inducer of MITF.
The Wnt/β-catenin signaling pathway has also been implicated in the pigmentation process. UVB irradiation of the mouse F1 of HR-1 × HR/De induced robust expression of Wnt7a and subsequent β-catenin translocation into the nucleus in the melanocyte stem cells (the model is presented in the Transgenic mouse models section). In the same model, intradermal injection of inhibitor of Wnt response 1(IWR-1) (a chemical inhibitor of β-catenin activation), and small interfering RNA against Wnt7a inhibited the proliferation of epidermal melanocytes. It was demonstrated that Wnt7a triggered melanocyte stem cell differentiation through β-catenin activation. The Wnt/β-catenin pathway seemed to be modulated by the UVB treatment in the mouse model, but the effects of UVB on the Wnt/β-catenin pathway on melanocytes and keratinocytes in the bulge of human vitiligo skin await to be studied. Wnt signaling is essentially required for melanocyte development; its activation results in stabilization of β-catenin/lymphoid enhancer factor (Lef) complex, which leads to transactivation of downstream target genes, such as Mitf , to promote melanocyte-fate specification and melanocyte differentiation. Moreover, epidermal Wnt controls hair follicle induction by orchestrating dynamic signaling crosstalk between the epidermis and dermis. Epithelial Wnt/β-catenin signaling is required for hair matrix cell proliferation, and β-catenin is required in bulge stem cells and also for epidermal proliferation.
More recently, transcriptome analysis on lesional, perilesional, and nondepigmented skin from patients with vitiligo and from matched skin of healthy subjects identified decreased Wnt activation in depigmented vitiligo skin. In addition, using ex vivo explants of depigmented human vitiligo skin, the authors found that treatment with Wnt agonists or glycogen synthase kinase 3 β (GSK3β) inhibitors induce increased expression of melanocyte-specific markers, triggering the differentiation of resident melanocyte stem cells in pre-melanocytes expressing paired box protein-3 (PAX3) and DCT. All these findings raise the possibility that Wnt normalization is important for the repigmentation process.
p53 seems to crosstalk with β-catenin in several cell lines, including the hair follicle stem cells. It has been reported that β-catenin and pygopus homolog 2 (Pygo2) (the latter with regulatory roles on Wnt/β-catenin target genes) converge to induce p53 in cultured keratinocytes and in cycling hair follicles. These findings identify Pygo2 as an important regulator of Wnt/β-catenin function in skin epithelia and p53 activation as a prominent downstream event of β-catenin/Pygo2 action in stem cell activation. Whether p53-β-catenin crosstalk intervenes during the repigmentation process awaits further study.
Human and animal models to study repigmentation
There are several mouse models and few human models used to study repigmentation, which the authors summarize later. They have revealed valuable information about the sequence of melanocyte proliferation, migration and differentiation.
Transgenic mouse models
SLFTg1-1 mice injected with anti-c-Kit monoclonal antibody ACK2
Investigation using this model has revealed the melanocyte precursors in the hair follicle, their migratory capacity, and participation to the repigmentation process. This model is a nonvitiligo repigmentation model, using the SLFTg1-1 humanized mouse, expressing steel factor (SLF) in the basal layer, under control of the Keratin (Krt)14 promoter. Treatment with the anti–c-Kit (ACK2) antibody eliminated the c-Kit(+) melanoblasts in the hair follicle and induced complete skin and hair depigmentation. In the next hair cycles, perifollicular repigmentation occurred, based on a population of residual melanocyte stem cells Dct(+)/c-Kit(−) maintained in the hair follicle, which showed migratory abilities.
K14-SLF/ +; Dct-lacZ/ + transgenic mouse model
The experiments done on this model identified the melanocyte precursors in the bulge, their migratory capacity under SLF stimulation, and their differentiation ability. This model is a non-vitiligo repigmentation model that, in addition to the SLFTg1-1 mutation, expresses the lacZ reporter gene under the control of the Dct promoter. Histologic examination of this mouse skin demonstrated that the progeny of surviving melanoblasts in the bulge could migrate upward to the epidermis in the presence of SLF, indicating that bulge stem cells are the source of melanocytes in the epidermis. Increasing numbers of lacZ+ cells appeared in the epidermis as pigmented spots in a concentric pattern around the terminal hairs.
F1 hairless HR-1 x HR/De mouse model
The experiments done on this model reveal the differentiation ability of melanocyte precursors in the bulge, their ability to migrate along the hair follicle outer root sheath, and also to repigment the interfollicular epidermis following UVB stimulation. This model is also a non-vitiligo model, in which homozygous mutants ( Hr hr /Hr hr ) show normal development of the first hair cycle. They become completely hairless at 3 weeks of age; at 4 weeks of age they become depigmented. In this model, delayed pigmented spots are induced long after UV irradiation. Following UVB exposure, the melanocyte precursors were observed to proliferate in the bulge and differentiate to melanoblasts that migrated to the epidermis and became melanotic cells.
Dct-LacZ + mouse model
The experiments done on this model revealed the migratory ability of bulge melanocyte precursors along the hair follicle outer root sheath, achieving proliferative and differentiation ability only after they reached interfollicular epidermis. This model is a non-vitiligo transgenic mouse model, expressing the lacZ reporter gene under the control of the Dct promoter. Following exposure to UVB or on induction of a wound on the back of the mouse, melanocyte stem cells were shown to exit the bulge and migrate along the outer root sheath infundibulum without proliferation; they proliferated and differentiated in the epidermis.
Mc1r e/e transgenic mouse model
The experiments done on this model revealed the migratory ability of melanocyte precursors along the hair follicle outer root sheath under melanocortin 1 receptor (Mc1r) stimulation. This mouse is a non-vitiligo, non-humanized mouse expressing a non-functional Mc1r. After wounding on the back of Mc1r e/e mice, a lower number of epidermal melanocytes were noticed as compared with their control littermate, Mc1r +/+ mice; the difference in melanocyte number in the epidermis was caused by impaired melanocyte migration from the bulge to the interfollicular epidermis, which was attributed to the lack of Mc1r function in the mouse expressing the defective gene.
Human models of repigmentation
Human vitiligo model using punch grafts
Punch grafts were performed on depigmented vitiligo lesions, and then they were exposed to khellin and UV light. Immunostaining experiments revealed the migratory capacity of melanocytes (horizontal migration to depigmented areas).
Human vitiligo model using punch biopsies
The experiments done on this model revealed the proliferative, migratory, and differentiation ability of melanocyte precursors in both hair follicle and interfollicular epidermis. Skin biopsies taken from patients with untreated vitiligo and from patients treated with NBUVB for 3 and 6 months were immunostained with melanocyte markers (DCT, C-KIT, TYR, or PAX3), markers of proliferation (KI-67), and/or of migration (melanoma cell adhesion molecule [MCAM]), and a keratinocyte specific marker (K14). NBUVB was associated with a significant increase in the number of melanocytes in the infundibulum and with restoration of the normal melanocyte population in the epidermis (see Fig. 3 B).
Repigmented narrow band ultraviolet treated–skin versus normal skin and untreated vitiligo skin
Using immunostaininig techniques coupled with collection of skin biopsies from patients with vitiligo, the authors showed that in the hair follicle bulge, NBUVB treatment stimulated a slight increase of 2 populations of immature melanocytes, a stem cell population C-KIT(−)/DCT(+), and a melanoblast population C-KIT(+)/DCT(+) (see Fig. 3 B). The targeted immature melanocyte in the hair follicle bulge and infundibulum of untreated vitiligo contained only amelanotic melanocytes (ie, they expressed the melanocyte markers DCT and/or C-KIT and/or PAX3, but they were TYR(−) and Fontana-Masson(−)); these immature populations remained amelanotic in the bulge after 3 to 6 months of NBUVB treatment. Fontana-Masson(+) cells and TYR(+) cells were seen only in the upper infundibulum and epidermis after treatment. NBUVB treatment was associated with significantly increased expression of melanocyte markers in the vitiligo treated skin, the most striking contrast being observed between the untreated depigmented epidermis (devoid of melanocytes) and the treated pigmented epidermis, which was heavily DCT(+), C-KIT(+), PAX3(+), TYR(+), and strongly Fontana-Masson(+).
Melanocyte proliferation after NBUVB was indirectly supported by the observation of increased melanocyte numbers in all regions tested and was directly quantified by KI-67 coexpression with C-KIT, DCT, and TYR. The authors identified a presumed migratory population of melanocytes (DCT(+)/MCAM(+)) that was minimally expressed in the bulge but showed increased expression in the infundibulum and epidermis. The melanocyte precursors showed differentiation abilities in the upper infundibulum by gradually exhibiting TYR expression, a process that paralleled proliferation and migration and that continued in the epidermis (population TYR(+)/MCAM(+)/KI-67(+)). The authors found no significant difference in melanocyte marker expression between NBUVB-treated vitiligo skin and normal skin, which suggested that NBUVB exposure for 3 to 6 months returns depigmented skin to a normal status in respect to pigmentation.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree