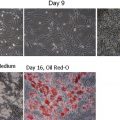
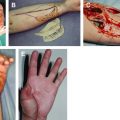
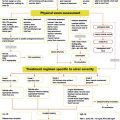
Wounds and damaged tissue become problematic when the tissue repair process does not proceed in a normal manner. Standard treatment of wounds entails topical dressings and devices in conjunction with good wound care practices. Good practices adequately support healing in most patients. Difficult, chronic, or recalcitrant wounds may require the use of more advanced technologies. Wounds that are full thickness or present with the absence of a matrix, may particularly benefit from regenerative materials. This article focuses on the use of cellular and acellular materials as well as chemical constructs to support granulation, tissue repair, and wound closure.
- •
The primary role of regenerative products is not to act as autologous skin graft, but to provide a means of dynamic interaction in the wound bed, thereby assisting and promoting tissue regeneration and wound closure.
- •
The clinician needs to differentiate between patients with normal and active cell responses versus those with impaired cellular activity, to assist with the choice of a living cell versus an acellular product.
- •
Not all acellular products function, integrate, or respond in the same manner, as a result of the variability of materials, structure, and components in each type of matrix.
- •
The variety and selection of regenerative materials, recently referred to as biomodulators, is so large that it has become necessary to classify them as either drugs, cellular products, or acellular products.
Regenerative materials that facilitate wound healing
The treatment of problematic and chronic wounds has evolved in the last 3 decades, progressing from the introduction of the first simple occlusive dressings to drugs and biologic materials. The addition of regenerative materials as well as biologic matrices has been a more recent addition to treatment modalities for wounds that do not respond to standard, conventional approaches for wound care. The rapid development and growth of cellular and acellular tissue replacements, matrices, and engineered products designed specifically for use in problematic wounds occurred after the introduction of living cell equivalents more than a decade ago. Since then, numerous acellular materials have been introduced and studied for use in tissue repair.
The variety and selection of regenerative materials, recently referred to as biomodulators, is currently so large that it has become necessary to classify them as either drugs, cellular products, or acellular products.
This article focuses on the use of true matrix replacements, including living cell and acellular wound materials. Applications of these types of products may extend beyond wound healing and are applicable to ulcers, tendon repair, hernia repair, tissue augmentation and other internal uses. This article discusses only the uses in wound healing in acute, chronic, and problematic wounds.
It is important to recognize that the primary role of regenerative products is not to act as autologous skin graft; they are designed to provide a means of dynamic interaction in the wound bed, thereby assisting and promoting tissue regeneration and wound closure. The term that best describes this process, biomodulation, was first used during an International Consensus on Acellular Matrices for the Treatment of Wounds.
The Extracellular Matrix
The extracellular matrix (ECM) is a key component of the tissue repair process. Its composition includes:
- •
Fibronectin
- •
Elastin
- •
Collagen
- •
Proteoglycans
- •
Hyaluronic acid.
The ECM is best compared with the framework or skeletal structure of a building in which all other components are added to the basic foundation and framework. Growth factors, fibroblasts, and other cellular components depend on the ECM for ongoing activity and tissue regeneration. The absence of an adequate ECM may significantly impair the natural sequence of tissue repair. The need for the presence of a matrix and regeneration in a wound bed has led to the development of regenerative products with a three-dimensional matrix (with or without a living cell component) that provide the framework for the repair process.
Most of the currently available materials are:
- •
Animal-derived or human-derived processed tissue
- •
Collagen-based or hyaluronic-based dressings
- •
Synthetic products
- •
Chemical constructs.
Living cells can also be used in conjunction with synthetic or collagen-based constructs. Because of the source of materials, many of these products are classified as biologic by the US Food and Drug Administration (FDA) (animal, human, or plant derived ).
Regardless of the manufacturing process, the tissue replacements and regenerative matrices are not antimicrobial agents. When placed in tissue defects that are not sterile or surgical uncontaminated wounds, bacterial growth becomes a barrier to effectiveness and outcomes. Aggressive debridement is needed to remove all nonviable and contaminated tissue. High levels of bacterial burden are known to impair the repair process, increase protease activity, and lead to further tissue breakdown. Most regenerative materials are protein based, therefore they are sensitive to protease levels, which may contribute to rapid degradation. Bacteria may also proliferate in a matrix and contribute to wound infection and further increase of matrix metalloproteinases (MMPs). Before application of any the products discussed in this article, bacterial presence must be significantly reduced or eliminated. The optimal environment for application of a wound matrix or regenerative product is the surgical setting, in which excisional and full-thickness debridement may be performed without difficulty. After application, protection from outside contamination, antibiotic use, and topical antimicrobials should always be considered to prevent new colonization or proliferation.
When choosing to use any newer technology, which inevitably comes with a significantly higher cost, clinicians should determine whether there are sufficient data to support the use of the product instead of more conventional modalities, and whether less expensive conventional treatments may provide similar results and time to wound closure.
- 1.
Remove all nonviable tissue
- 2.
Create a clean, bacteria-free environment
- 3.
Control the level of inflammation
- 4.
Protect product from external contamination
- 5.
Consider the concomitant use of antimicrobial agents
Living cell products
Cellular-derived wound healing products contain living cells such as fibroblasts, keratinocytes, or stem cells that are commonly embedded within a collagen or polyglactin matrix. Living cell products are designed to simulate the functional and biologic properties of human skin by:
- •
Providing a mechanical barrier to infection
- •
Encouraging extracellular matrix formation
- •
Stimulating keratinocyte growth and differentiation.
Living cell products are indicated for burn wounds, epidermolysis bullosa (EB), and most chronic wounds including diabetic, venous, and pressure wounds.
Only a few live-cell products are approved for clinical use ( Table 1 ). It is important for the clinician to understand the indications for each product and carefully evaluate any potential barriers to healing, such as infection, comorbidities, or patient noncompliance, to provide the best possible clinical outcome. Wounds of different cause, depth, age, and surface area must be treated differently and have dissimilar clinical prognoses. The use of adjunctive therapies such as compression, off-loading, and antibiotics should always be considered, if appropriate, to maximize healing success. Because living cell products are sensitive to bacteria and proteases, aggressive debridement is important, but only in the presence of adequate blood flow and the absence of any contraindication to sharp surgical debridement.
| Product | Company | Description | Live Cells | Uses |
|---|---|---|---|---|
| Apligraf | Organogenesis, Inc, Canton, MA | Bilayered epidermal and dermal equivalent | Neonatal keratinocytes | Chronic wounds: venous, diabetic |
| Dermagraft | Advanced Biohealing, Inc, La Jolla, CA | Single-layered dermal equivalent | Neonatal fibroblasts | Chronic diabetic wounds |
| Epicel | Genzyme Tissue Repair Corp, Cambridge, MA | Cultured autologous keratinocytes | Autogenous keratinocytes | Burn wounds >30% TBSA |
| Laserskin | Fidia Advanced Biopolymers, Abano Terme, Italy | Cultured autologous keratinocytes | Autogenous keratinocytes | Burn wounds >30% TBSA |
Wound Healing in Chronic Wounds
Normal wound healing requires a timely cellular response to injury by activation of keratinocytes, fibroblasts, endothelial cells, macrophages, and platelets, with resultant intercellular signaling through the coordinated release of growth factors and cytokines. Most chronic wounds are 1 of 3 categories:
- 1.
Pressure sores
- 2.
Diabetic ulcers
- 3.
Venous ulcers.
Prolonged chronic wounds have been shown to be deficient in growth factors (epidermal growth factor receptor, keratinocyte growth factor, platelet-derived growth factor [PDGF] and insulinlike growth factor [IGF]) and display :
- •
Decreased keratinocyte and fibroblast migration
- •
Increased reactive oxygen species
- •
Increased tissue proteases
- •
Microbial contamination.
Normal dermal fibroblasts synthesize and deposit critical extracellular components as well as secreting key growth factors important for intercellular signaling and repair. Fibroblasts from chronic wounds show pathologic changes in morphology, growth, and gene expression and have decreased or nonexistent replicative and functional ability. Keratinocytes are similarly dysfunctional, losing the ability to migrate from the wound edges and reepithelialize the wound surface. These key cells eventually become senescent and lose the capacity to react to growth factors that would normally stimulate a healing response.
Wound Healing in Diabetes
Wound healing in the diabetic patient is particularly challenging, because diabetic ulcers are slow to heal and prone to more serious complications such as osteomyelitis and amputation. There are more than 100 known physiologic factors that contribute to wound healing deficiencies in the diabetic patient, including :
- •
Derangement of cellular systems responsible for growth factor function
- •
Angiogenic response
- •
Macrophage function
- •
Collagen formation
- •
Epidermal barrier function
- •
Granulation tissue formation.
Regardless of the specific cause, it is vital to restore the continuity and integrity of the damaged skin in a timely fashion to minimize further morbidity and prevent complication. Regenerative live-cell products are designed to mimic the inherent cellular properties of the skin and provide temporary supplementation of critical functions lost by the chronic wound, such as keratinocyte and fibroblast proliferation and differentiation, extracellular matrix synthesis, and eventual reepithelialization.
The clinician needs to differentiate between patients with normal and active cell responses versus those with impaired cellular activity, to assist with the choice of a living cell versus an acellular product.
Apligraf
Apligraf (Organogenesis, Inc, Canton, MA, USA) is a bilayered skin equivalent designed to replicate the normal skin’s epidermis and dermis. The epidermal equivalent layer consists of a neonatal keratinocyte layer that is exposed to oxygen during the manufacturing process, giving rise to a stratified monolayer similar to the stratum corneum. The dermal equivalent layer contains neonatal fibroblasts impregnated on an extracellular collagen matrix composed of both bovine and human type I collagen. It is void of antigenic cells such as Langerhans cells, melanocytes, lymphocytes, macrophages, hair follicles, blood vessels, or sweat glands. Although the mechanism of action is not fully understood, it is thought that Apligraf creates a microenvironment that provides a physical and biologic barrier against wound infection and also produces a variety of MMPs, cytokines, and growth factors responsible for keratinocyte migration and extracellular matrix formation.
Use and Application of Apligraf
Apligraf is approved by the FDA for chronic venous ulcers of greater than 1 month’s duration and for diabetic ulcers of more than 3 weeks’ duration.
- •
It is supplied as a circular disk with a diameter of 7.5 cm and is 0.75 mm thick.
- •
It has a shelf life of 10 days and must be stored at 20 to 23°C until used.
- •
Apligraf may be applied every 4 to 6 weeks depending on the wound type, location, and clinician preference.
- •
As with most graft applications, wound bed preparation is critical and must involve proper debridement and control of edema and infection.
- •
Apligraf can be meshed or slit to facilitate drainage and is laid flat directly over the wound bed with the dermal side (glossy side) down.
- •
The graft should overlap the wound margin by 2 to 3 mm and care should be taken to smooth any wrinkles or air pockets. The graft is secured in place with staples or adhesive strips and protected with a soft primary dressing.
- •
Application of the product to the plantar diabetic foot requires special off-loading precautions to prevent disruption with ambulation.
- •
When applied to venous ulcers, compression is still required to address venous return. Compression needs to be applied carefully to prevent product disruption.
Apligraf Studies
Chronic venous leg ulcers
In 2000, Falanga published a prospective, randomized study of 214 patients with chronic venous leg ulcers treated with Apligraf with compression therapy versus compression therapy alone. Those patients treated with Apligraf were 3 times more likely to heal wounds older than 1 year ( P = .008) and 2 times more likely to attain complete wound healing by 24 weeks ( P = .002).
Diabetic foot ulcers
Veves and colleagues studied 208 patients in a multicenter, randomized, controlled trial in 2001 that compared Apligraf with moist gauze dressings for the treatment of diabetic foot ulcers. At 12 weeks, 56% of patients treated with Apligraf had complete wound healing versus 38% in the control group ( P = .004). The Apligraf-treated patients also had a faster median wound closure time of 65 days versus 90 days ( P = .003).
EB
Apligraf has also been used in the treatment of EB. Fivenson and colleagues published a small study of 9 patients with 96 sites of skin loss, of which, 90% to 100% healing was observed by 5 to 7 days, with clinically normal-appearing skin present by days 10 to 14. Falabella and colleagues also reported success in treating 69 acute EB wounds with no adverse events related to the application of Apligraf.
Dermagraft
Dermagraft (Advanced Biohealing, Inc, La Jolla, CA, USA) is a cryopreserved, single-layered, dermal substitute containing human-derived neonatal fibroblasts cultured on a bioresorbable polyglactin 910 scaffold. It stimulates the ingrowth of fibrovascular and epithelial tissue by depositing extracellular matrix components such as collagens, vitronectin, and glycosaminoglycans, and also secretes a variety of cytokines and growth factors including vascular endothelial growth factor (VEGF), PDGF, IGF-1, and granulocyte/macrophage colony-stimulating factor (GM-CSF). The fibroblasts continue to secrete growth factors and recruit host cells until fibrovascular ingrowth gradually replaces the donor cells and tissue. Dermagraft is void of antigenic cells and does not seem to stimulate rejection.
Use and Application
Dermagraft is primarily indicated and approved for the treatment of full-thickness diabetic foot ulcers of more than 6 weeks’ duration that are not overlying bone, tendon, muscle, or joint capsule.
- •
Dermagraft is cryopreserved and must be stored at −70 to −80°C until ready for use
- •
It is supplied in a clear bag containing 1 piece approximately 5 cm × 7.5 cm
- •
The graft must be thawed by submerging in water at 34 to 37°C for approximately 2 minutes and can be held aside in saline for up to 30 minutes
- •
The graft is laid flat on the wound bed (either side of the graft may be placed down) and trimmed to the approximate circumference of the wound margins
- •
Care should be taken to smooth out any wrinkles or air pockets to maximize surface area contact
- •
The graft is secured in place with staples or adhesive strips and a soft primary dressing should be applied directly over the graft
- •
Control of edema or proper off-loading is accomplished with an appropriate secondary dressing
- •
The primary dressing should be left in place for a minimum of 72 hours
- •
Dermagraft can be applied weekly for a total of 8 applications over a 12-week period
- •
When applying Dermagraft to the plantar diabetic foot, off-loading and removal of all pressure from the wound site is imperative to prevent disruption of the material when ambulating.
Dermagraft Studies
Diabetic foot ulcers of more than 6 weeks’ duration
Dermagraft was approved by the FDA in September 2001. The pivotal Dermagraft study was a multicenter, randomized, controlled study of 314 patients published by Marston and colleagues that compared Dermagraft with conventional wound care methods over 12 weeks. Patients included in the final analysis had ulcers of greater than 6 weeks’ duration. Twenty-eight percent of patients treated with Dermagraft achieved complete healing versus 14% in the control group ( P = .035). Wounds were 1.6 to 1.7 times more likely to heal in the Dermagraft group and the median wound closure was 91% versus 78% in the control group ( P = .44). The incidence of ulcer-related adverse events was also lower in the Dermagraft group (19%) compared with the control (32%; P = .007). Other non–FDA-approved uses included venous ulcers, fasciotomy wounds, buccal fat pad donor site healing, pediatric postsurgical abdominal wound healing, and vestibuloplasty.
Cultured epidermal autograft
A cultured epidermal autograft (CEA) is a single-layered epidermal substitute comprising the patient’s own keratinocytes, which are cultured ex vivo, together with mouse fibroblasts to form a thin sheet of skin. The total body surface area of a human (1.8 m 2 ) can be produced in just 4 weeks, although the minimum required preparation time for normal-sized wounds is 16 days. CEAs must be used in conjunction with a dermal substitute, which makes it fragile. It is currently approved for full-thickness burns of total body surface area greater than 30% and large congenital nevus excisions, although some success has been reported with treatment of leg ulcers. Although CEAs have shown some limited success with burn wounds, most of the literature concludes that it is generally unpredictable and inconsistent and should be used as an adjunct to conventional burn wound coverage with split-thickness autografts. Various improvements to cultured epidermal autografts are currently being studied, including a hyaluronic acid membrane carrier and a spray-on application.
- 1.
In the absence of contraindications and the presence of adequate blood flow for healing, debride the wound bed of all nonviable tissue
- 2.
Ensure that the material is in full contact with the wound bed and apply a nonadhesive wound contact layer over the product before dressing
- 3.
Securely attach the material to the wound margins
- 4.
Address non–weight bearing, compression, and other related treatment factors
- 5.
Avoid disruption of the dressing for up to 1 week if possible
The Future
Pilot studies are underway for the development of new live-cell regenerative wound care products. One such possibility is the use of multipotent adult stem cells, which have shown promise in accelerating wound repair and reconstituting the wound bed. Although considerable focus has been placed on bone marrow–derived mesenchymal stem cells, other types of stem cells are being studied, including those derived from hair follicles and adipose tissue. Currently there are no FDA-approved products available, and randomized clinical trials are still needed.
Living cell products
Cellular-derived wound healing products contain living cells such as fibroblasts, keratinocytes, or stem cells that are commonly embedded within a collagen or polyglactin matrix. Living cell products are designed to simulate the functional and biologic properties of human skin by:
- •
Providing a mechanical barrier to infection
- •
Encouraging extracellular matrix formation
- •
Stimulating keratinocyte growth and differentiation.
Living cell products are indicated for burn wounds, epidermolysis bullosa (EB), and most chronic wounds including diabetic, venous, and pressure wounds.
Only a few live-cell products are approved for clinical use ( Table 1 ). It is important for the clinician to understand the indications for each product and carefully evaluate any potential barriers to healing, such as infection, comorbidities, or patient noncompliance, to provide the best possible clinical outcome. Wounds of different cause, depth, age, and surface area must be treated differently and have dissimilar clinical prognoses. The use of adjunctive therapies such as compression, off-loading, and antibiotics should always be considered, if appropriate, to maximize healing success. Because living cell products are sensitive to bacteria and proteases, aggressive debridement is important, but only in the presence of adequate blood flow and the absence of any contraindication to sharp surgical debridement.
| Product | Company | Description | Live Cells | Uses |
|---|---|---|---|---|
| Apligraf | Organogenesis, Inc, Canton, MA | Bilayered epidermal and dermal equivalent | Neonatal keratinocytes | Chronic wounds: venous, diabetic |
| Dermagraft | Advanced Biohealing, Inc, La Jolla, CA | Single-layered dermal equivalent | Neonatal fibroblasts | Chronic diabetic wounds |
| Epicel | Genzyme Tissue Repair Corp, Cambridge, MA | Cultured autologous keratinocytes | Autogenous keratinocytes | Burn wounds >30% TBSA |
| Laserskin | Fidia Advanced Biopolymers, Abano Terme, Italy | Cultured autologous keratinocytes | Autogenous keratinocytes | Burn wounds >30% TBSA |
Wound Healing in Chronic Wounds
Normal wound healing requires a timely cellular response to injury by activation of keratinocytes, fibroblasts, endothelial cells, macrophages, and platelets, with resultant intercellular signaling through the coordinated release of growth factors and cytokines. Most chronic wounds are 1 of 3 categories:
- 1.
Pressure sores
- 2.
Diabetic ulcers
- 3.
Venous ulcers.
Prolonged chronic wounds have been shown to be deficient in growth factors (epidermal growth factor receptor, keratinocyte growth factor, platelet-derived growth factor [PDGF] and insulinlike growth factor [IGF]) and display :
- •
Decreased keratinocyte and fibroblast migration
- •
Increased reactive oxygen species
- •
Increased tissue proteases
- •
Microbial contamination.
Normal dermal fibroblasts synthesize and deposit critical extracellular components as well as secreting key growth factors important for intercellular signaling and repair. Fibroblasts from chronic wounds show pathologic changes in morphology, growth, and gene expression and have decreased or nonexistent replicative and functional ability. Keratinocytes are similarly dysfunctional, losing the ability to migrate from the wound edges and reepithelialize the wound surface. These key cells eventually become senescent and lose the capacity to react to growth factors that would normally stimulate a healing response.
Wound Healing in Diabetes
Wound healing in the diabetic patient is particularly challenging, because diabetic ulcers are slow to heal and prone to more serious complications such as osteomyelitis and amputation. There are more than 100 known physiologic factors that contribute to wound healing deficiencies in the diabetic patient, including :
- •
Derangement of cellular systems responsible for growth factor function
- •
Angiogenic response
- •
Macrophage function
- •
Collagen formation
- •
Epidermal barrier function
- •
Granulation tissue formation.
Regardless of the specific cause, it is vital to restore the continuity and integrity of the damaged skin in a timely fashion to minimize further morbidity and prevent complication. Regenerative live-cell products are designed to mimic the inherent cellular properties of the skin and provide temporary supplementation of critical functions lost by the chronic wound, such as keratinocyte and fibroblast proliferation and differentiation, extracellular matrix synthesis, and eventual reepithelialization.
The clinician needs to differentiate between patients with normal and active cell responses versus those with impaired cellular activity, to assist with the choice of a living cell versus an acellular product.
Apligraf
Apligraf (Organogenesis, Inc, Canton, MA, USA) is a bilayered skin equivalent designed to replicate the normal skin’s epidermis and dermis. The epidermal equivalent layer consists of a neonatal keratinocyte layer that is exposed to oxygen during the manufacturing process, giving rise to a stratified monolayer similar to the stratum corneum. The dermal equivalent layer contains neonatal fibroblasts impregnated on an extracellular collagen matrix composed of both bovine and human type I collagen. It is void of antigenic cells such as Langerhans cells, melanocytes, lymphocytes, macrophages, hair follicles, blood vessels, or sweat glands. Although the mechanism of action is not fully understood, it is thought that Apligraf creates a microenvironment that provides a physical and biologic barrier against wound infection and also produces a variety of MMPs, cytokines, and growth factors responsible for keratinocyte migration and extracellular matrix formation.
Use and Application of Apligraf
Apligraf is approved by the FDA for chronic venous ulcers of greater than 1 month’s duration and for diabetic ulcers of more than 3 weeks’ duration.
- •
It is supplied as a circular disk with a diameter of 7.5 cm and is 0.75 mm thick.
- •
It has a shelf life of 10 days and must be stored at 20 to 23°C until used.
- •
Apligraf may be applied every 4 to 6 weeks depending on the wound type, location, and clinician preference.
- •
As with most graft applications, wound bed preparation is critical and must involve proper debridement and control of edema and infection.
- •
Apligraf can be meshed or slit to facilitate drainage and is laid flat directly over the wound bed with the dermal side (glossy side) down.
- •
The graft should overlap the wound margin by 2 to 3 mm and care should be taken to smooth any wrinkles or air pockets. The graft is secured in place with staples or adhesive strips and protected with a soft primary dressing.
- •
Application of the product to the plantar diabetic foot requires special off-loading precautions to prevent disruption with ambulation.
- •
When applied to venous ulcers, compression is still required to address venous return. Compression needs to be applied carefully to prevent product disruption.
Apligraf Studies
Chronic venous leg ulcers
In 2000, Falanga published a prospective, randomized study of 214 patients with chronic venous leg ulcers treated with Apligraf with compression therapy versus compression therapy alone. Those patients treated with Apligraf were 3 times more likely to heal wounds older than 1 year ( P = .008) and 2 times more likely to attain complete wound healing by 24 weeks ( P = .002).
Diabetic foot ulcers
Veves and colleagues studied 208 patients in a multicenter, randomized, controlled trial in 2001 that compared Apligraf with moist gauze dressings for the treatment of diabetic foot ulcers. At 12 weeks, 56% of patients treated with Apligraf had complete wound healing versus 38% in the control group ( P = .004). The Apligraf-treated patients also had a faster median wound closure time of 65 days versus 90 days ( P = .003).
EB
Apligraf has also been used in the treatment of EB. Fivenson and colleagues published a small study of 9 patients with 96 sites of skin loss, of which, 90% to 100% healing was observed by 5 to 7 days, with clinically normal-appearing skin present by days 10 to 14. Falabella and colleagues also reported success in treating 69 acute EB wounds with no adverse events related to the application of Apligraf.
Dermagraft
Dermagraft (Advanced Biohealing, Inc, La Jolla, CA, USA) is a cryopreserved, single-layered, dermal substitute containing human-derived neonatal fibroblasts cultured on a bioresorbable polyglactin 910 scaffold. It stimulates the ingrowth of fibrovascular and epithelial tissue by depositing extracellular matrix components such as collagens, vitronectin, and glycosaminoglycans, and also secretes a variety of cytokines and growth factors including vascular endothelial growth factor (VEGF), PDGF, IGF-1, and granulocyte/macrophage colony-stimulating factor (GM-CSF). The fibroblasts continue to secrete growth factors and recruit host cells until fibrovascular ingrowth gradually replaces the donor cells and tissue. Dermagraft is void of antigenic cells and does not seem to stimulate rejection.
Use and Application
Dermagraft is primarily indicated and approved for the treatment of full-thickness diabetic foot ulcers of more than 6 weeks’ duration that are not overlying bone, tendon, muscle, or joint capsule.
- •
Dermagraft is cryopreserved and must be stored at −70 to −80°C until ready for use
- •
It is supplied in a clear bag containing 1 piece approximately 5 cm × 7.5 cm
- •
The graft must be thawed by submerging in water at 34 to 37°C for approximately 2 minutes and can be held aside in saline for up to 30 minutes
- •
The graft is laid flat on the wound bed (either side of the graft may be placed down) and trimmed to the approximate circumference of the wound margins
- •
Care should be taken to smooth out any wrinkles or air pockets to maximize surface area contact
- •
The graft is secured in place with staples or adhesive strips and a soft primary dressing should be applied directly over the graft
- •
Control of edema or proper off-loading is accomplished with an appropriate secondary dressing
- •
The primary dressing should be left in place for a minimum of 72 hours
- •
Dermagraft can be applied weekly for a total of 8 applications over a 12-week period
- •
When applying Dermagraft to the plantar diabetic foot, off-loading and removal of all pressure from the wound site is imperative to prevent disruption of the material when ambulating.
Dermagraft Studies
Diabetic foot ulcers of more than 6 weeks’ duration
Dermagraft was approved by the FDA in September 2001. The pivotal Dermagraft study was a multicenter, randomized, controlled study of 314 patients published by Marston and colleagues that compared Dermagraft with conventional wound care methods over 12 weeks. Patients included in the final analysis had ulcers of greater than 6 weeks’ duration. Twenty-eight percent of patients treated with Dermagraft achieved complete healing versus 14% in the control group ( P = .035). Wounds were 1.6 to 1.7 times more likely to heal in the Dermagraft group and the median wound closure was 91% versus 78% in the control group ( P = .44). The incidence of ulcer-related adverse events was also lower in the Dermagraft group (19%) compared with the control (32%; P = .007). Other non–FDA-approved uses included venous ulcers, fasciotomy wounds, buccal fat pad donor site healing, pediatric postsurgical abdominal wound healing, and vestibuloplasty.
Cultured epidermal autograft
A cultured epidermal autograft (CEA) is a single-layered epidermal substitute comprising the patient’s own keratinocytes, which are cultured ex vivo, together with mouse fibroblasts to form a thin sheet of skin. The total body surface area of a human (1.8 m 2 ) can be produced in just 4 weeks, although the minimum required preparation time for normal-sized wounds is 16 days. CEAs must be used in conjunction with a dermal substitute, which makes it fragile. It is currently approved for full-thickness burns of total body surface area greater than 30% and large congenital nevus excisions, although some success has been reported with treatment of leg ulcers. Although CEAs have shown some limited success with burn wounds, most of the literature concludes that it is generally unpredictable and inconsistent and should be used as an adjunct to conventional burn wound coverage with split-thickness autografts. Various improvements to cultured epidermal autografts are currently being studied, including a hyaluronic acid membrane carrier and a spray-on application.
- 1.
In the absence of contraindications and the presence of adequate blood flow for healing, debride the wound bed of all nonviable tissue
- 2.
Ensure that the material is in full contact with the wound bed and apply a nonadhesive wound contact layer over the product before dressing
- 3.
Securely attach the material to the wound margins
- 4.
Address non–weight bearing, compression, and other related treatment factors
- 5.
Avoid disruption of the dressing for up to 1 week if possible
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree