Reconstruction of Femur
Vishwanath R. Chegireddy
Michael J. A. Klebuc
Anthony Echo
DEFINITION
Femoral shaft bone defects usually result from tumor resection, traumatic loss, chronic osteomyelitis, congenital skeletal defects, failed allografts, or infected nonunions.1,2
Extensive resection of long bone can lead to large intercalary osseous defects necessitating reconstruction, which was traditionally achieved with allografts or allograft prosthetic composites. These allografts were associated with high rates of complications, including joint instability, fracture of the allograft, and infection of the allograft.3
Vascularized bone flaps are essential for successful bone reconstruction due to their ability to incorporate into the recipient site and provide early bone consolidation while maintaining stability of the appendicular skeletal structure. Owing to its independent blood supply, such a flap is well incorporated into the recipient bone despite compromised surrounding tissue from previous surgery or radiation therapy.
The advantage of vascularized bone flap over nonvascularized bone grafts is the intrinsic ability to integrate into the recipient bone similar to standard fracture healing instead of creeping substitution.
The fibula is an ideal vascularized bone flap for femur reconstruction due to its ease of access for harvest while preserving the stability of the proximal and distal tibial-fibular syndesmosis despite segmental resection. It has a negligible influence on knee and ankle joint stability without compromising weight-bearing ability or function of the lower extremity.
ANATOMY
The femur is the longest and strongest bone in the body and is subject to large amounts of axial loading as well as significant rotational angular stresses.
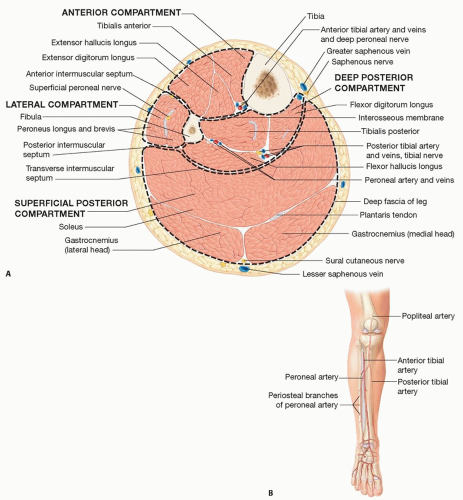
The fibula is a long and thin cortical bone with a small medullary component. In adults, the width of the fibula is 1.5 to 2 cm with a length of 35 cm of which approximately 25 cm of the fibular diaphysis can be harvested for free tissue transfer.
Vascular supply to the diaphysis of the fibula is via a dominant nutrient endosteal artery and minor musculoperiosteal branches from the peroneal artery and veins. These vessels run parallel to the fibula and course between the flexor hallucis longus (FHL) and tibialis posterior muscle group.
The dominant nutrient artery branches 6 to 14 cm from the peroneal artery bifurcation and enters the fibula posterior to the interosseous membrane. It then courses into the nutrient foramen located in the middle third of the diaphysis and further divides into an ascending and descending branch.
Musculoperiosteal vessels are derived from the peroneal artery and travel within the posterolateral septum, through the FHL and tibialis posterior muscle, giving segmental rise to four to eight branches that supply the muscles in the anterior compartment, soleus muscle, and lateral leg skin territory. The majority of the periosteal branches are located in the middle third of the fibula (FIG 1).
Soft tissue defect coverage is achieved with a skin paddle supplied by perforators traversing through the posterior crural septum with the majority concentrating at the proximal and distal ends of the fibula. These fasciocutaneous perforators supply a skin paddle up to 10 × 20 cm in size.7
PATIENT HISTORY AND PHYSICAL FINDINGS
Detailed preoperative evaluation is required for patients undergoing extensive long bone resection and intercalary bone defect reconstruction.
A thorough medical history should be obtained to evaluate the peroneal artery for free fibula graft. Pertinent history includes trauma, deep venous thrombosis, peripheral vascular disease, lymphedema, or venous insufficiency. Patients with peripheral vascular disease may not be a candidate for surgery.
Physical exam should include an evaluation of knee and ankle joints for range of motion and laxity, and a foot Allen test with a Doppler probe to access dorsalis pedis and posterior tibial arteries.
Peronea arteria magna is a congenital variant of the arterial inflow to the foot, which affects about 1% to 8% of the population, where both the anterior and posterior tibial arteries are hypoplastic or absent with the peroneal artery serving as the single arterial supply below the knee.
Patients with an abnormal vascular exam or a history of trauma to the affected extremity will need additional preoperative imaging such as an arteriogram, computed tomography angiogram (CTA), duplex ultrasound, or magnetic resonance angiography (MRA) to identify atherosclerotic occlusive disease or congenital anomalies.
IMAGING
Preoperative imaging of both the recipient and donor sites should include an evaluation of:
Recipient site:
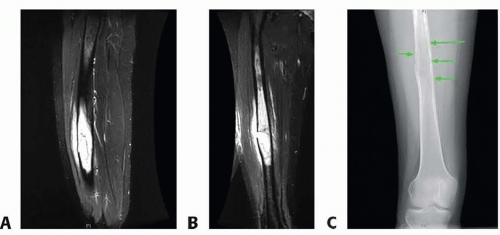
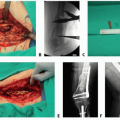
The extent of bone resection, length and diameter of the intercalary defect, and potential soft tissue defect (FIG 2). This allows the surgeon to determine the extent of the tumor burden, traumatic bone loss, or chronic osteomyelitis and plan for reconstructive options.
Donor site:
Evaluate vascular supply with Doppler ultrasound (anterior tibial, posterior tibial, and dorsalis pedis arteries), and exclude any fibular deformities.
Angiogram to look for vessel patency and flow to the foot.
SURGICAL MANAGEMENT
Preoperative Planning
Free fibula bone graft can be transferred with a generous skin paddle and adjacent muscles such as soleus, peroneal muscles, or FHL.
Either right or left leg can be used as a donor for fibula flap. The left side is preferable in most patients because it is the less dominant leg and used less frequently while driving a vehicle. However, thoughtful planning of vessel orientation in the recipient site should be considered, when deciding upon laterality.
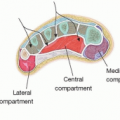
Prior to incision, anatomical landmarks including the head of the fibula, lateral malleolus, and anterior and posterior border of the fibula are marked. The posterior border of the fibula correlates with the posterior intermuscular septum encasing the vascular supply to the skin paddle (FIG 3).
Intraoperative Doppler probe is used to identify perforator vessels to design the skin paddle centered over the posterior crural septum. Sometimes, the perforators pass through the FHL and/or soleus muscles and lie posterior to the septum.
Area of fibula bone resection is marked with superior margin 4 cm below the head of the fibula and inferiorly 6 cm above the lateral malleolus. By preserving the proximal fibula, the stability of the knee is maintained without disrupting the attachment of the tibia to the fibula, fibular collateral ligament to the head of the fibula, and biceps femoris muscle. Also, the common peroneal nerve courses inferior to the head of the fibula, and identifying this location can prevent injury during dissection.
Positioning
Positioning can allow two surgical teams to work simultaneously, if the contralateral fibula flap is used (FIG 4):
The patient is placed in the supine position on the operative table with the donor side knee flexed 90 degrees. A roll is placed under the ipsilateral hip to internally rotate the pelvic girdle and keep the hip and knee flexed.
A heel stop can be placed on the operative room table to help knee at 90 degrees, when harvesting in the supine position.
The patient can also be placed in lateral decubitus position if donor and recipient site is in the same limb:
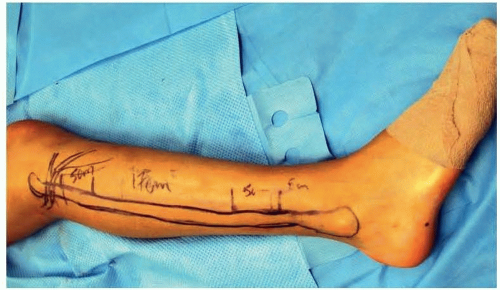
FIG 3 • Intraoperative marking of anatomical landmarks for an ipsilateral fibula harvest in the lateral decubitus position: head of the fibula, lateral malleolus, and anteroposterior fibular border.
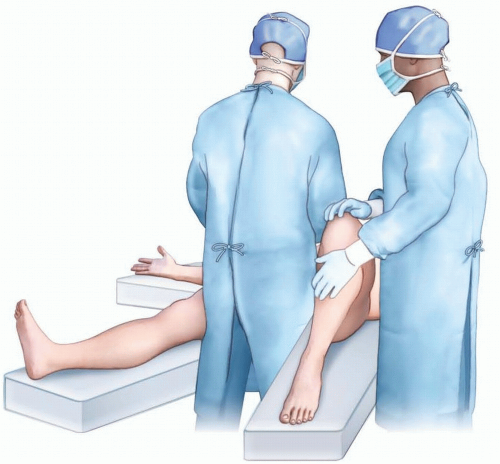
FIG 4 • If the contralateral fibula flap is desired, the patient can be placed in a supine position, which allows two teams to work simultaneously for recipient preparation and donor site harvest.
A bean bag can help keep the patient in the lateral position.
All pressure points should be carefully padded and protected.
Approach
Plastic surgeons and orthopedic surgical team will need to work together to coordinate resection of the affected femur segment and reconstruction of the subsequent intercalary defect. Preservation of potential recipient vessels is of the upmost importance.
Reconstruction of long femoral intercalary defects with vascularized fibula alone is not sufficient due to inadequate stability and fragility of the bone to tolerate early weight bearing. The Capanna method is ideal in this scenario, using a femoral allograft as a peripheral shell and telescoping the free fibula graft through the allograft segment.7
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree