Recipient Vessel Exposure—Internal Mammary and Thoracodorsal
Theodore A. Kung
Adeyiza O. Momoh
DEFINITION
Recipient vessels for microsurgical breast reconstruction are required to re-establish perfusion in transferred free flaps.
Perfusion through these vessels is most critical early on after free tissue transfers, when flaps are entirely dependent on the recipient vessels for inflow and outflow. Over time, collateral perfusion is typically established from the surrounding soft tissue bed.
The primary recipient vessels for breast reconstruction are the internal mammary and thoracodorsal vessels.
ANATOMY
Internal Mammary Vessels
The internal mammary artery (IMA) is a paired artery on both sides of the sternum that arises from the subclavian artery. It transitions into the superior epigastric vessels at about the sixth intercostal space.
The internal mammary vein (IMV) is also paired, runs parallel to the IMA, arising from the superior epigastric vein and ending in the brachiocephalic vein.
The vessels are 1 to 2 cm lateral to the sternum and run deep to the intercostal muscles and ribs.
Both vessels give off intercostal branches laterally that run inferior to the first six ribs. Perforators to the overlying breast and skin are also given off by the IMA and IMV in the first 5 to 6 intercostal spaces.
The right and left IMAs are of similar diameter (1.9-2.1 mm) at 3rd and 4th intercostal spaces where they are typically used.1,2 At the level of the 3rd intercostal space, the IMA lies lateral to the IMV or between two IMVs.
The left IMV bifurcates at a higher level than does the right IMV (3rd rib on the left vs 4th rib on the right). At the level of the 3rd ICS, the left IMV is on average smaller than the right (2.5 mm vs 3 mm).3
Thoracodorsal Vessels
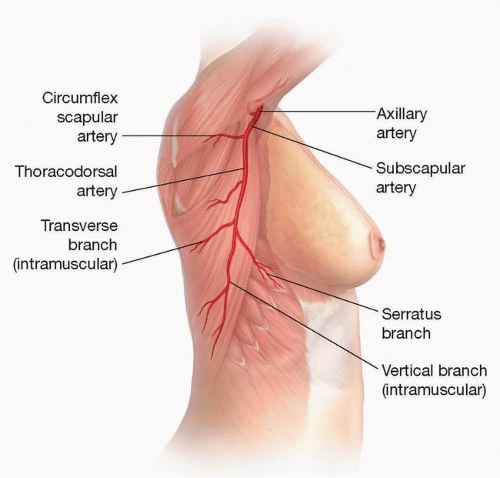
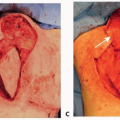
The thoracodorsal artery (TDA) and the circumflex scapular artery are the two main branches of the subscapular artery, which arises from the third division of the axillary artery (FIG 1).
The TDA travels along the lateral thoracic wall before diving into the latissimus dorsi muscle about 9 from its origin. Prior to its entry into the muscle, the TDA consistently (99%) gives off a branch to the serratus anterior muscle.4
The TD vessels are commonly divided for microvascular anastomosis just proximal to the serratus branch where the diameter of the artery is approximately 3 mm.5 Preservation of the serratus branch also allows for the use of a pedicled latissimus dorsi flap and an implant for breast reconstruction in case free tissue transfer is unsuccessful.
Proximally, there are often two veins that unify into a single TD vein. The TD vein travels with the TD artery and has an average diameter of 3.4 mm at its origin and 1.6 mm where it enters the latissimus dorsi muscle.6
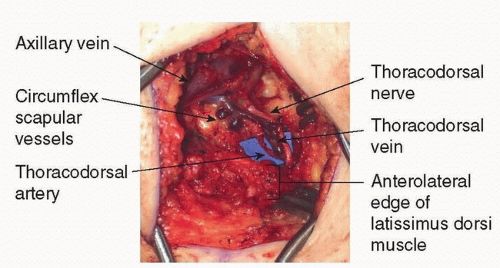
The TD nerve enters the axilla deep to the axillary vein at a point several centimeters medial to the origin of the subscapular vessels. The nerve then continues toward the subscapular system and eventually travels parallel with the TD vessels along the lateral thoracic wall (FIG 2).
PATIENT HISTORY AND PHYSICAL FINDINGS
A thorough history of prior thoracic and breast procedures including axillary lymph node dissections and prior chest wall radiation are important to determine possible compromise to the IM or TD vessels.
The continuity of IM vessels is seldom affected by oncologic breast procedures; however, a history of radical chest wall tumor resections or coronary bypass procedures may prevent use of the IM vessels as recipients.
Thoracodorsal vessels are at greater risk of injury during axillary lymph node dissections and should be approached with caution in the setting of delayed reconstruction in such patients.
A history of radiation should prompt an anticipation of friable recipient vessels. IM vessel friability introduces technical challenges with exposure and anastomosis but can be consistently overcome with careful vessel handling.
IMAGING
There is limited value to routine preoperative imaging of IM or TD vessels in the absence of a history that suggests potential compromise.
If a patient has had a preoperative breast MRI, that status of the mammary vessels may be evident.
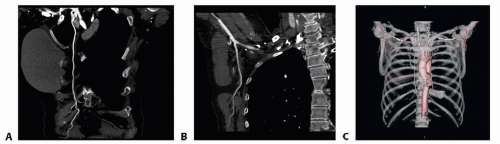
When needed, CT angiography provides detailed information on the vascular anatomy for surgical planning (FIG 3).
SURGICAL MANAGEMENT
The decision on which recipient vessel is to be used is made preoperatively. The internal mammary vessels are the authors’ preferred option for immediate and delayed microsurgical breast reconstruction with the TD vessels serving as a backup option.
If immediate reconstruction following mastectomy and axillary lymph node dissection is performed, the TD vessels are often already partially exposed and may be considered for anastomosis. However, the vessels must be carefully inspected for any injury or spasm resulting from the extirpation surgery. Additionally, the flap pedicle must be of sufficient length to reach the TD vessels for anastomosis.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree