Introduction
Peripheral nerve injuries are common and can be functionally devastating. Improved understanding of nerve topography, injury, and regeneration has led to advances in peripheral nerve surgery and increased treatment options for our patients.
Traditionally, when a tensionless repair cannot be performed, a nerve autograft is employed to bridge the defect. However, autografts can be associated with additional surgical sites, increased operative time for harvesting, and potential donor site morbidity, including sensory loss, scarring, and debilitating neuroma formation. Further, in the cases of large, segmental or multiple limb injuries, the amount of expendable autograft becomes a limitation. For these reasons, alternatives including nerve allotransplantation and processed nerve allografts were developed.
More recently, vascularized composite tissue allotransplantation has become a reality for the treatment of devastating injuries involving traumatic amputations or significant facial disfigurement. Since 1998, over 100 hand and 30 face transplants have been performed, with published reports of 72 hand and 19 face transplantations. The function of these transplanted parts is dependent on specificity and efficacy of the nerve regeneration. The purpose of this chapter is to explore and review nerve autograft alternatives. Specifically, we focus on the biology of nerve allografts, review the clinical experience of nerve allotransplantation, and discuss nerve regeneration in the setting of vascularized composite allotransplantation (VCA). We will also briefly describe the clinical evidence for the use of nerve conduits and processed nerve allografts.
Peripheral Nerve Regeneration
A peripheral nerve injury initiates a highly regulated and sophisticated sequence of events. This process is dependent on the degree of nerve injury. Establishing the severity of the injury will help predict if recovery is possible and if surgical intervention is needed. Seddon first classified nerve injury into neurapraxia, axonotmesis, and neurotmesis. Sunderland and Mackinnon later revised the classification system ( Table 68.1 ). A neurapraxia involves localized myelin damage. The axon is preserved and thus the nerve does not undergo degeneration. Axonal disruption, in contrast, leads to Wallerian degeneration distal to the site of injury with coordinated activation of Schwann cells (SCs) and macrophages to remove myelin and axonal debris.
| Seddon | Sunderland | Description | Recovery |
|---|---|---|---|
| Neuropraxia | I | Conduction block | Spontaneous recovery |
| Axonotmesis | II | Axonal Injury | Spontaneous recovery |
| III | Axonal and Endoneurium Injury | Partial recovery without surgical intervention | |
| IV | Axonal, Endoneurium, and Perineurium Injury | No recovery without surgical intervention | |
| Neurotmesis | V | Axonal, Endoneurium, Perineurium, and Epineurium Injury (Nerve Transection) | No recovery without surgical intervention |
| VI | Mixed pattern injury | Mixed recovery based on injury pattern |
Proximal to the injury, mitogenic cytokines released by the injured axons induce SCs to change into a proliferating phenotype. These proliferating SCs organize into columns along the basement membrane of endoneurial tubes and are referred to as “bands of Büngner” and they support axonal regeneration. Axons are initially pruned and then begin to sprout and regenerate along these bands to reestablish nerve continuity and reach the end-organ target. In humans, this regenerative rate is reported to occur at 1–3 mm/day.
Segmental nerve defects or tension at the site of injury will disrupt the normal cascade of events preventing regeneration. Traditionally, interposed autografts have served as a bridge or structural framework to allow regeneration to proceed and are considered the gold standard. They provide the necessary medium for regeneration, including viable SCs and extracellular matrix, despite undergoing Wallerian degeneration themselves. Autografts are typically chosen based on their caliber, the length of the nerve gap, donor site morbidity, and ease of harvest. Due to the ease of harvest, the long length, and minimal donor site morbidity, one of the most commonly used autografts is the sural nerve. Other suitable nerves for interpositional grafts are reviewed in Table 68.2 .
| Nerve graft | Potential length | Characteristics |
|---|---|---|
| Sural | 30–40 cm | Formed from the medial cutaneous sural nerve originating from the tibial nerve |
| Common donor nerve | ||
| Descends the leg between the two heads of the gastrocnemius becoming superficial just before it is joined by the sural communicating branch off the peroneal nerve | ||
| The donor defect involves the lateral and posterior part of the lower third of the leg but decreases in size over time | ||
| Medial antebrachial cutaneous | 20 cm | Travels down the arm medial to the brachial artery then becomes superficial to the deep fascia in the middle of the arm and runs distally with the basilica vein |
| Typically bifurcates proximal to the medial epicondyle into anterior and posterior branches | ||
| Harvesting the anterior branch avoids loss of sensation over the elbow | ||
| Lateral antebrachial cutaneous | 5–8 cm | Distal continuation of the musculocutaneous nerve |
| Exits from under the biceps tendon and splits into an anterior and posterior branch, running with the cephalic vein | ||
| Sensory defect involves the anterolateral forearm but can involve the volar radial or dorsoradial thumb | ||
| Significant or total overlap of sensory innervation with the superficial sensory radial nerve | ||
| Posterior cutaneous nerve of the forearm | 2–5 cm | Sensory branch of the radial nerve |
| Arises from the radial nerve in the spiral groove and passes through the lateral head of the triceps | ||
| Descends down the lateral aspect of the arm and terminates at the dorsal forearm and wrist | ||
| Provides sensation to the posterolateral forearm and is rarely used | ||
| Superficial radial nerve | 15–20 cm | Prone to painful neuroma formation, therefore not a first-line choice |
| It may be used for proximal radial nerve injuries | ||
| Separates from the radial nerve just distal to the elbow | ||
| Becomes superficial as it wraps around the brachioradialis 7 cm proximal to the wrist crease | ||
| Sensory defect corresponds to the dorsum of the thumb and the 1st and 2nd web spaces | ||
| Dorsal sensory branch of the ulnar nerve | 4–6 cm | Appropriate size and length for use as a digital nerve |
| Takeoff from the ulnar nerve 5 cm proximal to the wrist | ||
| Travels along the dorsomedial aspect of the wrist before dividing into two or three terminal dorsal branches | ||
| Painful neuroma formation is not typical | ||
| Donor defect involves the dorsal ulnar wrist, hand, and proximal parts of the ulnar two fingers | ||
| Posterior interosseous nerve | 6 cm | Terminal portion of the PIN provides wrist joint capsule proprioception |
| Lies deep to the 4th extensory compartment | ||
| Good donor for digital nerve defects | ||
| No apparent motor or sensory defect | ||
| Anterior interosseous nerve | 7 cm | Terminal portion of the AIN provides wrist joint capsule proprioception and innervates the pronator quadratus |
| Lies adjacent to the anterior interosseous artery on the volar aspect of the interosseous membrane as it enters the pronator quadratus | ||
| No clinically apparent motor or sensory defect | ||
| Superficial peroneal nerve | 30–35 cm | Major lateral branch of the common peroneal nerve innervating the peroneus longus and brevis, in addition to providing sensation to the lateral aspect of the lower leg and dorsal foot |
| Consistently long graft | ||
| Donor defect motor and sensory | ||
| Saphenous nerve | 40 cm | Largest cutaneous branch of the femoral nerve |
| Becomes subcutaneous between the sartorius and gracilis tendons | ||
| Advances down the leg next to the saphenous vein | ||
| Donor defect involves the medial aspect of the lower leg | ||
| Do not take the saphenous and sural nerve from the same leg as their cutaneous innervation overlaps and the resulting donor defect is significant | ||
| Intercostal nerves | 20 cm | Mixed motor/sensory nerve innervating accessory respiratory muscles of the thoracic wall and overlying skin |
| Donor site incision can be large and invasive | ||
| Risk of pneumothorax | ||
| Avoid harvesting T4 intercostal nerve to preserve sensation to nipple areola complex | ||
| Lateral cutaneous nerve of the thigh | 10–20 cm | Donor defect involves loss of sensation to the anterolateral aspect of the thigh. |
Nerve Allotransplantation
In large, segmental, complex, or multi-limb nerve injuries, the amount of available autograft sources may be limited. Nerve allografts, from either a cadaver or living donor provide an abundant source of nerve graft. They also avoid the donor site morbidity of nerve autografts, but require systemic immunosuppression for their use. In the presence of adequate immunosuppression, nerve allografts provide equal regeneration and function of an autograft. Studies even demonstrate enhanced regeneration in nerve allografts due to the regeneration-enhancing effects of the immunosuppressant, tacrolimus.
Systemic immunosuppression has known risks. These include, but are not limited to, increased risk for opportunistic infections, nephrotoxicity, and decreased cancer immunosurveillance, resulting in malignancy. Due to the toxicity and risks of immunosuppression, the indication for nerve allografts has been narrow, and careful selection of the patients is important. Currently, nerve allotransplantation is reserved for unique clinical situations of devastating nerve injuries that are not amenable to nerve transfers or nerve autografting, and conservative management would result in a nonfunctional limb or nonmanageable pain.
Nerve allotransplantation is unique when compared with solid organ and composite tissue transplantation. The nerve allografts act as a biological and temporary conduit through which host motor and sensory axons traverse to reach the host end-organ target. Thus, nerve allografts require only temporary systemic immunosuppression, which can be withdrawn once the axons have crossed the allograft.
Schwann Cells and Immunology of Nerve Allotransplantation
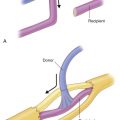
As previously discussed, SCs are an integral part of injured axonal recovery as they provided both neurotrophic factors and structural integrity. In both nerve autografts and allografts, SCs are essential for supporting axonal regeneration and subsequent remyelination. In nerve allografts, both donor and host SCs proliferate and support axonal regeneration as long as adequate immunosuppression is present ( Fig. 68.1A ).
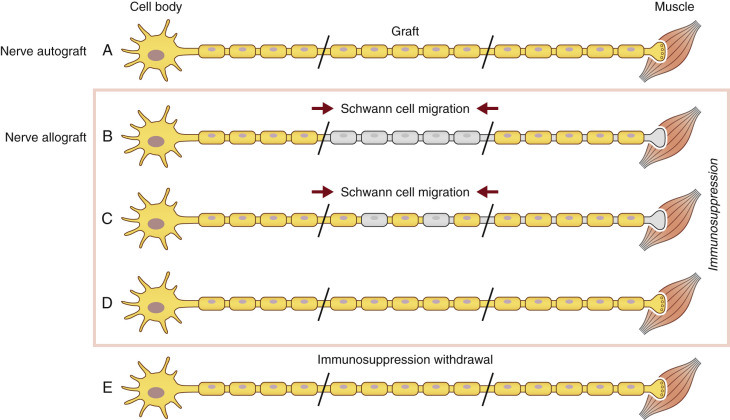
Host SC migration is essential in the process of nerve regeneration across a nerve allograft. It occurs from both the proximal and distal nerves. Eventually, donor SCs are lost, despite adequate immunosuppression, but this is from an unclear mechanism and may involve chronic rejection or episodes of subtherapeutic immunosuppression. Our laboratory explored SC migration and found, under immunosuppression, host SC migration into the graft is delayed. However, after regeneration was completed and immunosuppression was stopped, host SC migration occurred immediately to fill in the gaps left by the loss of the rejected donor SCs along the graft ( Fig. 68.1E ).
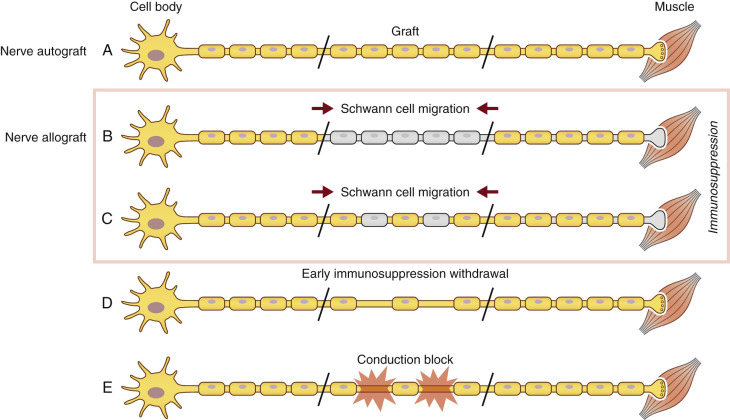
SCs are also known to play a large immunogenic role in nerve allograft rejection. They express major histocompatibility complex I (MHC-I), a molecule on cellular membranes that is responsible for displaying fragments of intracellular proteins to T cells to either trigger or prevent an immune response. Transplanted SCs also increase their expression of MHC II molecules, a family of molecules found on antigen-presenting cells and B-cell lymphocytes, increasing their host rejection response. For this reason, SCs are recognized as a prime target of the host alloimmune response. Because SCs play a large role in both axonal regeneration and allograft rejection, immunosuppression cannot be safely withdrawn until regeneration has reached the end-target organ. Functionally, removal of immunosuppression prior to repopulation of the donor graft by host SCs will result in donor SC death and a subsequent, devastating conduction block ( Fig. 68.2 ).
Current Strategies for Nerve Allotransplantation
Effort to mitigate donor alloantigenicity and host immune response to allografts is currently the source of significant clinical and laboratory investigation. In an attempt to prevent rejection of the allogenic nerve and simultaneously allow nerve regeneration, two strategies have been employed: (1) minimize the antigenicity of the nerve graft and (2) minimize the host immune response to the allograft tissue.
Strategies to decrease the antigenicity of the nerve graft include numerous decellularizing techniques such as cold preservation, radiation, lyophilization, and freeze-thawing. These attempts to minimize the induction of the host immune response created nerve grafts with inconsistent efficacy in supporting nerve regeneration. Due to their acellular nature and sometimes altered structural make-up, these treated grafts failed to reach results equivalent to autologous nerve grafts, especially over longer graft lengths. Thus, these techniques are not utilized independently in practice.
Cold preservation has successfully been shown to reduce allogenicity of nerve allografts. The effects are dependent on length of exposure. Increased storage time leads to decreased allogenicity (6 weeks) but also correlates with decreased axonal regeneration. One week of cold preservation, however, reduced the expression of MHC class II alloantigens but did not reduce the number of viable SCs. Thus, our current clinical protocol involves 7 days of cold preservation of the nerve allografts at 4–5°C in University of Wisconsin solution prior to implantation (see Table 68.4 ).
The advent of successful systemic immunosuppressive therapy has further allowed nerve allotransplantation to have clinical importance. Early work in peripheral nerve allograft immunosuppression involved the use of cyclosporine, but later shifted to the more potent calcineurin phosphatase inhibitor, FK-506. FK-506, also known as tacrolimus, inhibits the activation of T-cell proliferation, but has been shown in numerous animal models to enhance and accelerate nerve regeneration. Numerous calcineurin-independent pathways have been implicated in FK-506’s ability to enhance regeneration via mediators including growth associated protein 43 (GAP 43), FKBP-52, heat shock protein 90 (HSP-90), and cytoskeletal dynamics, but the exact mechanism has not yet been elucidated. Regardless of the mechanism, FK-506’s ability to speed regeneration and increase reinnervation of distal targets is well accepted.
Our clinical and laboratory experience with FK-506 has provided parameters for maximizing the benefits of this immunosuppressant. Low-dose/subtherapeutic doses of FK-506 equally provide regenerative enhancement to full dose administration. Thus, alternative immunosuppression regimens such as co-stimulatory blockade can be used with low-dose FK-506. However, the beneficial results of FK-506 were eliminated when both full dose and co-stimulatory blockade were given.
The timing of FK-506 administration is also important when considering the beneficial effects of the drug. Delay of administration should be avoided and preloading the drug before nerve repair should be considered. Animal studies revealed that the neuroregenerative effects diminished with delayed administration. If given after 5 days in a transection and repair model, no beneficial effects of the drug were seen. Preloading the drug 3 days before nerve transection and repair, in contrast, further enhanced the neuroregenerative effects. Although this timing is not possible in vascularized composite allotransplantation (VCA) due to the unpredictable nature of donor availability, there is a window of time – while the donor tissue is being procured and prepared – when administration of loading doses of FK-506 could be given. Ideally, FK-506 should be given for the duration of nerve regeneration and should be included in the maintenance regimens in VCA.
Our current immunosuppressive regimen involves both graft pretreatment and systemic immunosuppression. The senior author’s clinical experience and preferences will be elaborated on in the next section.
Clinical Experience with Nerve Allotransplantation
As discussed previously, the indications for nerve allotransplantation are limited due to the risks of immunosuppression. Currently, nerve allotransplantation is reserved for unique clinical situations of devastating nerve injuries that are not amenable to nerve transfers or nerve autografting. Patient selection and a multidisciplinary team approach are critical to the success of this procedure.
In contrast to solid organ or VCA transplantation, nerve allografts require only temporary systemic immunosuppression, which can be withdrawn once the axons have crossed the allograft. However, detection of rejection during regeneration is difficult with nerve allotransplantation. In organ transplantation, measurements of organ function, such as serum creatinine and liver transaminases, provide clinicians with objective data. In nerve allografts, the grafts are buried and functional assays do not exist. In nerve allotransplantation, once rejection is detected, either by local inflammation or erythema, it is usually too late to rescue the graft with modifications of the immunosuppression. Thus, having a sound immunosuppressive protocol, adequate patient selection and clinical suspicion, and ensuring patient compliance is essential.
The senior author (S.E.M.) has significant clinical experience with nerve allotransplantation. The results of the initial seven cases have previously been reported. Since that publication, an additional five patients have undergone reconstruction with nerve allografts. All of these cases are summarized in Table 68.3 . In the first 11 cases, nerve allografts were used to reconstruct large segmental defects in the extremities. Between 72 cm and 248 cm of nerve allograft pretreated with cold preservation were used in each case and in conjunction with the available nerve autograft in 9 of the 11 patients.
- •
Most cases were in younger patients (age range: 3–54 years).
- •
Patient 6 lost the graft due to acute rejection from non-compliance with immunosuppression.
- •
Most patients recovered protective sensation with varying degrees of motor reinnervation.
- •
Motor reinnervation was most robust in proximal muscle groups.
- •
The average length of time patients remained on FK-506 was 20.1 months. This was dictated by clinical evidence of nerve growth beyond the distal end of the allograft.
| Case | Surgery date | Age/Sex | Immunosuppression | Allograft total length | Defect gap | Outcome |
|---|---|---|---|---|---|---|
| 1 | Sept 1988 | 8-M | 26 months cyclosporine | 230 cm | Sciatic 23 cm | Regained protective sensation on plantar aspect of foot, no motor return |
| 2 | Sept 1993 | 12-M | 19 months cyclosporine | 160 cm | Post-tib. 20 cm | Regained 8/10 sensation in lower extremity but no motor |
| 3 | Feb 1994 | 15-F | 12 months cyclosporine | 178 cm (+59 cm Autograft) | Median 27 cm, Ulnar 15 cm | Excellent reinnervation of extrinsic ulnar/median muscles and good light touch sensation |
| 4 | Dec 1995 | 3-F | 18 months cyclosporine | 72 cm (+24 cm Autograft) | Ulnar 12 cm, SBR 12 cm, PIN 12 cm | Good sensory return with motor function in FDS and ulnar distribution |
| 5 | May 1996 | 26-F | 17 months FK-506 | 226 cm (+44 cm Autograft) | Radial 20 cm, Ulnar 18 cm, Median 22 cm | Motor recovery of the FCU, ulnar FDP and thenar muscles. Good sensory recovery in median distribution only |
| 6 | Nov 1996 | 16-M | Subtherapeutic cyclosporine (3 weeks) | 350 cm (+63 cm Autograft) | Median 23 cm, Ulnar 18 cm, Radial 27 cm | Acute rejection with subtherapeutic immunosuppression levels noted at 3 weeks. Salvage failed with increased doses |
| 7 | June 1998 | 24-M | 19 months FK-506 | 140 cm (+28 cm Autograft) | Post-tib. 14 cm (+28 cm Autograft) | Good sensory recovery on sole of foot |
| 8 | Feb 2000 | 35-M | 32 months FK-506 | 198 cm (+48 cm Autograft) | Median 24 cm, Radial 21 cm, Ulnar 18 cm | Recovered protective sensation to distal extremity with good proximal function |
| 9 | July 2000 | 15-M | 24 months FK-506 | 228 cm (+51 cm Autograft) | Post-tib. 20 cm, Peroneal 11 cm | Recovered protective sensation to distal dorsum and sole of foot |
| 10 | July 2005 | 19-M | 18 months FK-506 | ~100 cm Living Donor and Allograft (+74 cm Autograft) | Radial 20 cm, Median 8 cm (Autograft only), Ulnar 10 cm (Autograft only) | Excellent return of function to median and ulnar intrinsic musculature |
| 11 | Feb 2006 | 21-M | 18 months FK-506 | ~200 cm Living Donor and Allograft (+129 cm Autograft) | Median 19 cm, Radial 18 cm, Ulnar 18 cm (Autograft only) | Good functional recovery to ulnar and median nerve distribution, required tendon transfers for radial |
| 12 | Oct 2011 | 57-M | (surgery to present) FK-506 | 248 cm | N/A | Pain significantly decreased by 7 months after surgery |
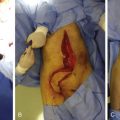
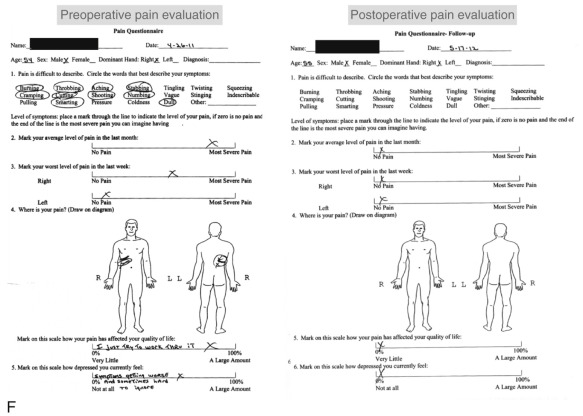
In contrast to the first 11 cases of allotransplantation, the most recent patient underwent nerve allotransplantation for significant pain after a thoracotomy. This 54-year-old man had recalcitrant right intercostal neuralgia following two thoracic operations 8 years prior to presentation to our clinic. Following his second thoracic procedure (a pleural leak repair after a right upper lobe resection), he developed severe pain localized to the dermatomes of T5-T8. His dysesthetic pain was unresponsive to conservative therapy ( Fig. 68.3 ). In a novel use of allograft transplantation, two 48 cm nerve allografts were looped between the T5-T6 and T7-T8 intercostal nerves following exposure and transection of the proximal nerve roots. Each was looped to connect two consecutive levels and allowed the regenerating intercostal nerves to taper off, preventing neuroma formation and pain. His neuralgia pain subsided by 7 months after his reconstruction. Autografts were avoided in this patient to avoid a second site of neuropathic pain. At 30 months postoperatively, the patient reported his pain was entirely resolved in the thoracic distribution and only limited to the spinal incision site. On a visual analog scale, he rated his pain a 3 out of 10. He controls his pain with topical anesthetic patches and activity modifications. He reported the procedure was “life-changing.”
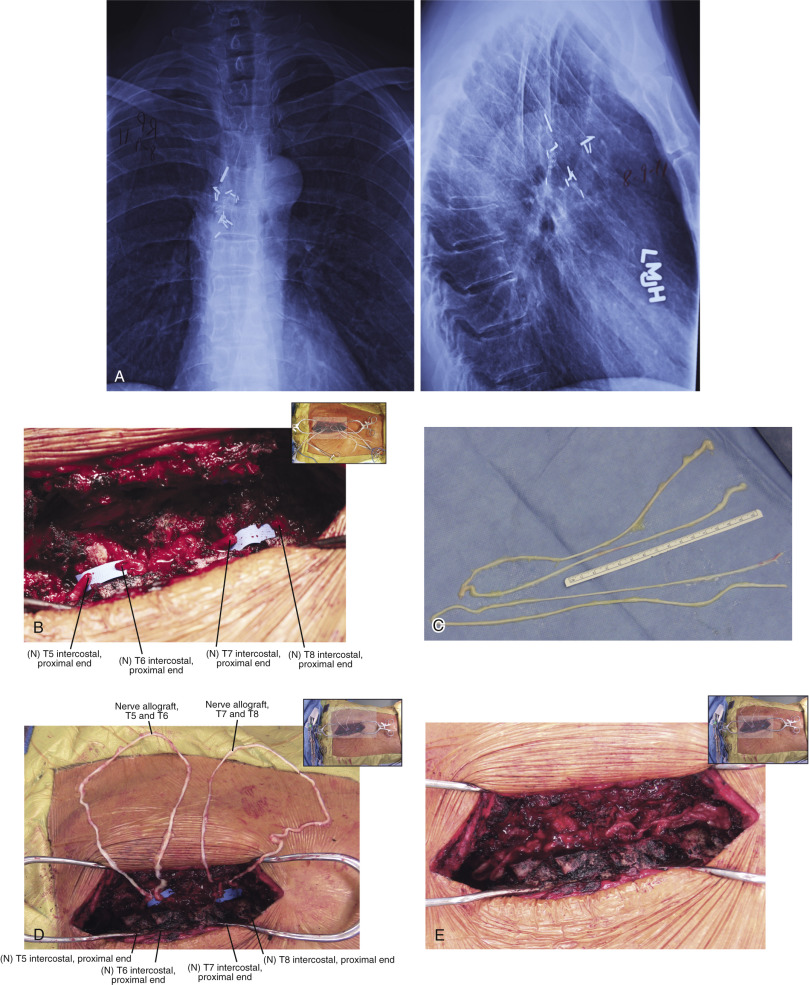
Our current clinical protocol involves exhausting all available autografts followed by the use of ABO-blood typed nerve allograft matched donors harvested within 24 h of death. More recently, living, related donors have also been used, as this can eliminate the waiting period for a matched donor and can decrease immunogenicity. To further reduce allogenicity, our protocol utilizes the University of Wisconsin Cold Storage Solution at 4°C for 7 days prior to implantation. We also use multiple small-diameter allograft cables as these revascularized more consistently than larger-diameter grafts. Our immunosuppression regimen incorporates the initiation of FK-506, 3 days preoperatively to maximize the regenerative potential. Our current protocol is detailed in Table 68.4 .
| Preoperative preparation | Intraoperative details | Postoperative care |
|---|---|---|
| Assess patient suitability | Resect patient’s injured nerve to healthy proximal and distal stumps | Induction immunotherapy (dose 2) |
| Medical comorbidities | Basiliximab 20 mg intravenously (postoperative day 4) | |
| Infection/Malignancy | ||
| Psychosocial fitness | ||
| Laboratory tests | Use patient’s autografts in addition to cadaveric allografts, donor-related or donor-unrelated allografts | Standard immunotherapy |
| ABO blood type | Continue FK-506 (goal level 5–8 ng/mL) | |
| CBC, CMP | Azathioprine 1–1.5 mg/kg per day | |
| HIV/Hepatitis | ||
| Nerve allografts | Insert extra allograft subcutaneously for rejection monitoring | Antibiotics |
| Consider related donors | Sulfamethoxazole-trimethoprim | |
| Harvest small-diameter grafts | Three times a week | |
| Cold preservation (4°C University of Wisconsin Solution, 7 days) | ||
| Immunotherapy | Stop immunotherapy | |
| FK-506 (tacrolimus) 1–2 mg PO BID (start 3 days preoperatively) | 6 months after Tinel’s crosses distal repair site | |
| Induction immunotherapy (dose 1) – basiliximab 20 mg intravenously (immediately preoperatively) |
Nerve Regeneration in Vascularized Composite Allotransplantation
The long-term functional outcome in vascularized composite allotransplantation (VCA) is dependent on the quality of nerve regeneration and muscle reinnervation of transplanted parts. With successful results from the multiple hand and face transplants performed to date, VCA is a viable option to treat devastating upper extremity and facial injuries. Understanding the nuances of nerve regeneration in these transplanted parts is necessary and critical to maintain the functional benefit of these procedures.
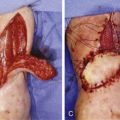
A number of differences between nerve regeneration in a nerve allograft versus that in VCA exist. Unlike isolated nerve allografting, which only requires temporary immunosuppression, VCA requires lifelong immunosuppression to prevent the rejection of the more immunogenic tissues such as skin, muscle, and bone. As mentioned earlier, nerve allografts depend on the migration of SCs from both the distal and proximal host nerve stumps into the graft to ensure axonal regeneration and ultimately function of the nerve. In VCA, there are no distal host SCs, as the entire distal neuromuscular unit is composed of donor tissue. Therefore, repopulation of the transplanted nerve with host SCs is dependent on the proximal stump only ( Fig. 68.4 ). Currently, it is unknown if there is a limit to how far host SCs are able to migrate to functionally repopulate a nerve in a transplanted part. Further, in nerve allografts, SC migration in the graft was stimulated by episodes of subclinical rejection in an effort to replace depleted donor SCs. In the presence of continuous immunosuppressive regimens, as required in VCA, it is unknown to what extent host SC migration will be stimulated and how this will affect long-term function.
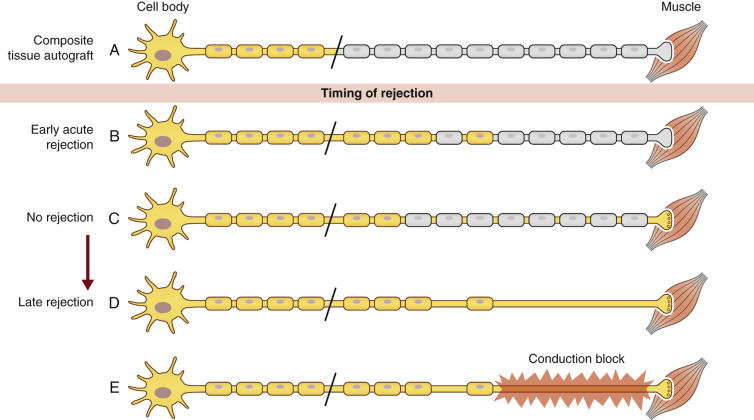
Long-term characterization of host and donor SCs in VCA is not yet fully understood. It is believed that donor SCs are no different from host SCs in their ability to support nerve regeneration. However, lapses in therapeutic immunosuppression causing acute rejection and early loss of donor SCs may have a more pronounced clinical effect in VCA, leading to demyelination and resulting in conduction blocks or a devastating permanent functional failure. Until immunosuppressive strategies are developed to induce tolerance to all donor tissue, the late loss of donor SCs from unexpected episodes of rejection is a significant concern.
Currently, VCA has a very specific application involving severely injured or disfigured patients who have adequate motivation to undergo a major surgical procedure, lifelong immunosuppression, and a lengthy rehabilitation process. Despite this, patients who undergo the procedure remain very satisfied and are recovering neurologic function. Length-dependent functional motor and sensory axonal regeneration has been observed in both facial and upper extremity transplants. Compared with more proximal transplants, distal hand transplants have demonstrated both a superior sensory and motor recovery, with reported return of intrinsic motor function, and discriminative sensation. Forearm and upper arm transplant patients have demonstrated return of sensation over longer time periods, but inconsistent and commonly protective sensation at best. The motor recovery of these more proximal transplants appears to continually improve over the first 5 years, but is commonly limited to elbow and forearm animation.
Future strategies to enhance nerve regeneration in VCA should consider the delicate balance between donor and host SCs. Strategies to optimize donor SC migration into regenerating transplanted nerves within a therapeutic immunosuppression environment should be developed. Keeping donor SCs alive to support axonal regeneration during this time period is important, and may help prevent rejection or functional failure over the lifetime of the patient.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree