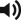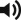
Introduction
The conventional latissimus dorsi muscle or myocutaneous flap has undoubtedly served an important role as one of the most common flaps in reconstructive microsurgery. However, its bulkiness and the sacrifice of donor structures often preclude its use. In this respect, the reduction in volume and complete preservation of the muscle and its motor nerve that can be achieved using the perforator concept are indeed worthwhile.
Cutaneous branches from the thoracodorsal and lateral thoracic artery were first mentioned by de Coninck et al. and the flaps based on these arteries have been known as thoracodorsal skin flaps, thoracodorsal axillary flaps, and lateral thoracic flaps. Since the introduction of the perforator concept by Koshima , the perforator flap from the lateral thoracic region was attempted by Angrigiani et al. under the name of the latissimus dorsi musculocutaneous flap without muscle. It has since been developed as a thin pattern for a resurfacing tool by Kim et al. ; another septocutaneous perforator has also proven to be a useful pedicle of the perforator flap. Three distinct rows of perforator patterns are present and interconnected with each other in the lateral thoracic region. According to the author’s nomenclature system, they may be given different names, such as the “latissimus dorsi perforator flap” based on a musculocutaneous perforator, the “thoracodorsal perforator flap” based on a septocutaneous perforator, and the “lateral thoracic perforator flap” based on a direct cutaneous perforator.
These perforator flaps from the lateral thoracic region region can provide many options for reconstruction, such as resurfacing a wound with a thin cutaneous flap, reconstructing a head and neck defect including intraoral resurfacing, and reconstructing defects requiring different tissue components by harvesting the flap as a chimeric flap.
Flap Anatomy ( Figs 56.1 , 56.2 and Fig 11.1 , Fig 11.2 , Fig 11.3 , Fig 11.5 , Fig 11.8 , Fig 11.9 )
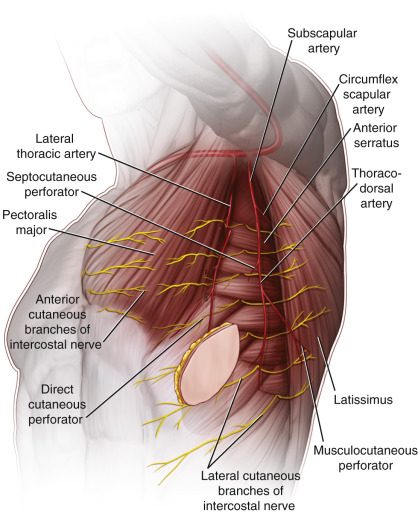
The subscapular artery arises from the axillary artery and divides into the thoracodorsal and the circumflex scapular arteries The thoracodorsal artery penetrates the latissimus dorsi muscle about 8–14 cm from the bifurcation of the subscapular artery into the circumflex scapular and thoracodorsal. Shortly before it enters the muscle, the vascular bundles send a branch to the serratus anterior muscle. The latissimus dorsi muscle is nourished by two main muscular branches from the thoracodorsal artery: a lateral branch running parallel to the anterior border and a horizontal branch passing obliquely to the dorsal and medial part of the muscle. They give multiple terminals to the skin as perforators through the muscle along the course of branches, and these musculocutaneous perforators are located at intervals in the back area. Septocutaneous perforators arise from the branch to the serratus anterior or other cutaneous branches, and they reach the skin between the latissimus dorsi and serratus anterior muscles. Cutaneous branches originate from the main thoracodorsal artery or from the branch to the serratus anterior. They also supply septocutaneous perforators or direct cutaneous perforators.
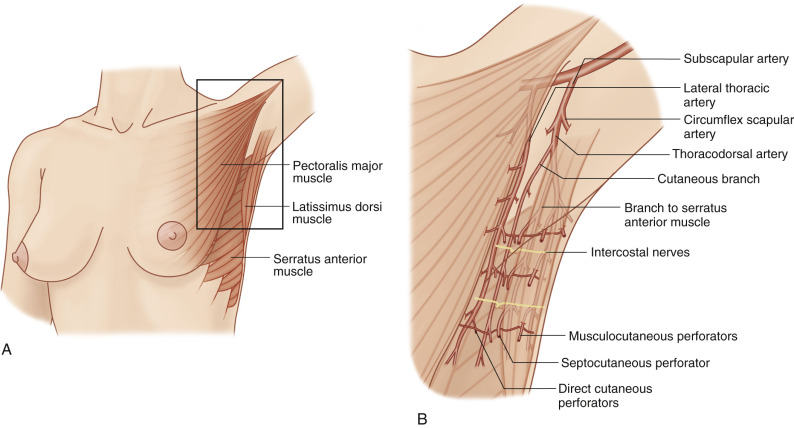
Additional cutaneous branches are supplied from the lateral thoracic artery, which arises as a third branch from the axillary artery at the lateral border of the pectoralis major muscle. It supplies the pectoralis major, the pectoralis minor and upper digitations of the serratus anterior muscle, while running distally along the lateral border of the pectoralis major and superficially on the serratus muscle fascia, anterior to the long thoracic nerve, and terminating in the skin as direct cutaneous perforators. In females, this branch is prominently developed to supply the lateral portions of the breast. The additional accessory lateral thoracic artery is present as a direct branch in 70% from the axillary artery between the origin of the lateral thoracic artery and the subscapular artery. It runs along the midaxillary line, terminating with several direct cutaneous perforators.
Arterial Supply of the Flap (see Figs 56.1 , 56.2 and Fig 11.1 , Fig 11.2 , Fig 11.3 , Fig 11.5 )
Three distinct rows of perforator groups are well detected in the lateral thoracic region, at intervals parallel to the anterior border of the latissimus dorsi muscle. The most frontal line is a longitudinal series of direct cutaneous perforators arising from the lateral thoracic vessels, and is located on the serratus anterior muscle along the lateral border of the pectoralis major muscles. The middle row, about 2 cm anterior to the latissimus dorsi muscle border, is composed of septocutaneous (or direct cutaneous) perforators from the cutaneous branches of thoracodorsal vascular systems. The most posterior row is composed of musculocutaneous perforators through the latissimus dorsi muscle, and its anterior group is located within 2 cm of the anterior border of the latissimus dorsi muscle.
There are horizontal communications among these perforators; therefore, the criss-cross network is formed by the connection of a musculocutaneous perforator via a septocutaneous perforator going to a direct cutaneous perforator in each row ( Fig. 56.2 ). Intercostal vessels are also connected in each horizontal row, and intercostal nerve branches are collateral with this network. The perforator from the intercostal vessels is very tiny, but it can be available for the pedicle of an island pattern for regional coverage.
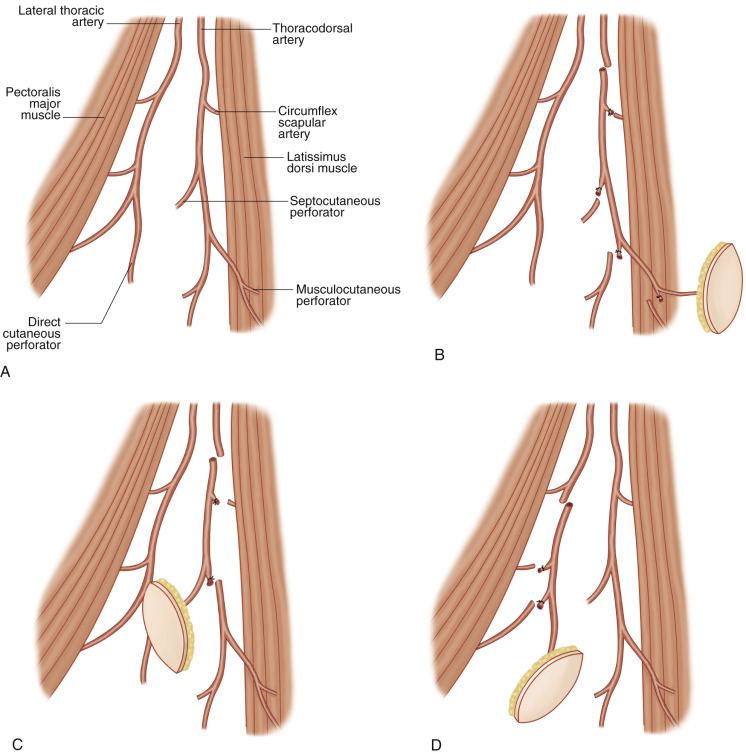
To discriminate between these perforator flaps that have the same donor skin territory, a different nomenclature is required to better understand the pedicle base or the course dissected. According to the author’s new version of the nomenclature system, the perforator flap based on a musculocutaneous perforator is named the latissimus dorsi perforator flap, and the flap based on a septocutaneous perforator, is named the “thoracodorsal perforator flap.” An additional perforator flap is available in this area based on a direct cutaneous perforator from the lateral thoracic artery, and it is named the lateral thoracic perforator flap ( Fig. 56.3 ).
Latissimus Dorsi Perforator Flap
Dominant:
musculocutaneous perforators from the thoracodorsal artery through the latissimus dorsi muscle
Length: 12 cm (range 8–14 cm from the origin of the thoracodorsal artery)
Diameter: 1 mm (range 0.8–2 mm at the level of the proximal thoracodorsal artery)
Thoracodorsal vessels are divided into a muscle branch to serratus anterior and latissimus dorsi muscles ( Fig. 56.3B ) and the latter is divided into a horizontal and a vertical branch inside the muscle (see Ch. 11 and Ch. 41 ).
The pedicle can be lengthened an additional 6–8 cm, by including the proximal source vessel of the subscapular artery or shortened to the level where the thoracodorsal artery enters the latissimus dorsi muscle, according to the condition and diameter of recipient vessels. Sometimes, its length can be controlled by the perforator chosen (proximal or distal) and the location of the perforator on the flap (central or peripheral).
Minor:
there are several minor perforators distributed several centimeters apart from the reliable perforator.
One of these can be included and thus pedicle length and diameter may be changed according to the perforator selected.
Thoracodorsal Perforator Flap ( Fig. 56.3C )
Dominant:
septocutaneous perforators from the thoracodorsal artery
Length: 10 cm (range 7–12 cm from the origin of the thoracodorsal artery)
Diameter: 1 mm (range 0.8–2 mm at the level of the thoracodorsal artery)
Septocutaneous perforators arise from the branch to the serratus anterior, cutaneous branches, or thoracodorsal vessels. The origin of cutaneous branches is also variable from the main thoracodorsal artery or from the branch to the serratus anterior. Several direct cutaneous perforators may originate from the cutaneous branch. In 100 cadaver dissections by Rowsell et al. , direct cutaneous branches were identified in the subscapular–thoracodorsal arterial system in 81% of dissections. A total of 7% originated from the axillary artery, 28% from the subscapular artery, and 47% from the thoracodorsal artery, with an average diameter of 1.0 mm. This study was published before the advent of the perforator concept and some branches might be confused with perforators. The branch was introduced in another study as a direct cutaneous branch from the extramuscular course of the thoracodorsal artery, reported in 55% of dissections. The anatomic basis of the subscapular arterial system has been ascribed by Cabanie et al. to a consistent, direct cutaneous branch of the thoracodorsal artery.
To review the evolution of using the pedicle in flaps ( Fig. 56.4 ), conventional flaps were based on the arterial concept and later were developed on the branch-based concept. Recently, it was clinically confirmed that the same dimension of the flap can be reliably based on only one available perforator arising from the branch. Moreover, the perforator flap can be harvested as a thin pattern based on the single, reliable perforator, excluding the underlying deep fascia. Its dimension may extend over its angiosome, compared with the conventional flap. In our clinical experience, all three perforators are always present, but the location of the reliable perforator in each row varies according to the patient.
Minor:
there are several minor perforators several centimeters apart from the reliable perforator.
One of these can be included and its pedicle length and diameter are similar to that of the dominant perforator.
Lateral Thoracic Perforator Flap ( Fig. 56.3D )
Dominant:
direct cutaneous perforators from the lateral thoracic artery
Length: 8 cm (range 5–10 cm from the subclavian artery)
Diameter: 1 mm (range 0.5–2 mm at the origin of the lateral thoracic artery)
In the latissimus dorsi perforator flap, the pedicle is usually reliable in its diameter and comitant veins. Conversely, the pedicles of the septocutaneous and direct cutaneous perforators may be smaller in diameter, and they have several tiny branches to the surrounding adipose tissues. Sometimes, they have the constant diameter of an artery in spite of the proximal dissection of the pedicle, and end-to-side arterial anastomosis may be required. The pedicles of the lateral thoracic and the thoracodorsal perforator flap are shorter than that of the latissimus dorsi perforator flap.
Minor:
there are several minor perforators several centimeters apart from the reliable perforator.
One of these can be included and its pedicle length and diameter are similar to that of the dominant perforator.
Venous Drainage of the Flap (see Fig. 11.8 )
Primary:
comitant veins of the thoracodorsal, subscapular or lateral thoracic artery
Length: same length as the artery it accompanies
Diameter: 1 mm (range 0.8–5 mm)
The venous drainage of the latissimus dorsi perforator flap through the comitant veins is quite reliable, with a reliable proximal diameter. However, in the perforator-based flap preserving the proximal thoracodorsal vessels, the distal comitant vein beneath the latissimus dorsi muscle is often too small to be used for vascular anastomosis, and further proximal dissection of the vein might be required to insure safer venous anastomosis. Conversely, the venous drainage of the thoracodorsal or lateral thoracic perforator flap may be variable, ranging from tiny comitant veins to a large, parallel vein.
Secondary:
lateral thoracic vein or other superficial veins
Length: 8 cm (range 6–10 cm)
Diameter: 1.2 mm (range 1–1.5 mm)
In the thoracodorsal and lateral thoracic perforator flaps, the comitant veins may be too narrow to use for anastomosis, and a separate large cutaneous vein may be included in the pedicle. The separate vein, draining into the proximal axillary vein, has a variable course and multiple branches. The lateral thoracic vein runs parallel and anterior to the muscle border of the latissimus dorsi with a reliable diameter. Other superficial cutaneous veins are also optional; they mainly drain directly into the axillary vein.
Flap Innervation (see Figs 11.2 , 11.9 )
Sensory:
A lateral branch of the intercostal nerve is recommended for the flap sensation because these branches can be easily detected during dissection of a reliable perforator.
The sensation is quite useful in resurfacing reconstructions, especially in the neck, hand, foot, and oral cavity, including the tongue.
Motor:
The perforator flaps are harvested without muscle, and motor nerve preservation is attempted during intramuscular dissection.
Minor tributaries are sometimes inevitably sacrificed at some points along the perforator pedicle; however, functional deficit is not apparent. The thoracodorsal and lateral thoracic perforator flaps do not require an intramuscular dissection and no injury occurs.
Flap Components
Perforator flaps in the lateral thoracic region are elevated as cutaneous flaps but can be composed of the latissimus dorsi muscle, the serratus anterior muscle, the rib or scapular bone and regional fat tissues based on a separate branch of the pedicle (see Flap modification/flap handling ).
Advantages
- •
Reduced donor site morbidity .
- •
Perforator dissection allows for preservation of muscle and motor nerves and minimizes donor site morbidity.
- •
Controlled resurfacing with a thin flap (see Flap modification/flap handling ).
- •
Various patterns and compositions can be harvested and used to reconstruct defects requiring different tissue arrangements and tissue types. Composite flaps based on the perforator concept are easier to handle and the components of the flap can be arranged in the recipient site without restriction. More delicate, controlled contouring is possible in three-dimensional reconstruction (see Flap modification/flap handling ).
- •
Sensate flap (see Flap modification/flap handling ).
- •
Long pedicle and proximal branching system . An additional length of 5–6 cm (>15 cm in total) can be obtained through transmuscular perforator pedicle dissection (“transmuscular” is the term we prefer over the more commonly used term “intramuscular” since the dissection is made through the muscle to the proximal source vessel). Further lengthening is possible by designing the flap with the perforator in an eccentric position.
- •
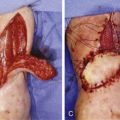
Minimal morbidity of the donor scar . The donor site can be closed primarily up to 10 cm (12 cm in older patients), resulting in an acceptable linear scar and no functional deficits.
- •
Availability of secondary options . Other perforator patterns or conventional flaps, such as the scapular, parascapular, and muscle flaps, can be considered as secondary options in the worst cases.
- •
Short and simple intramuscular dissection due to the thinness of the latissimus dorsi muscle.
- •
The operation can be performed in the supine position when a large flap is not required.
Disadvantages
- •
Inconsistent anatomy . The learning curve for identifying a reliable perforator with ease may be steep. Preoperative ultrasound studies prove helpful in alleviating intraoperative difficulties. It is possible to convert the flap into the muscle-sparing technique involving a minimal segment of the muscle.
- •
Long operation time . Searching for a reliable perforator and, in some cases, changing position of the patient can increase operation times.
- •
Pedicle injury during dissection and potential for compression and kinking during the inset. Extreme care is required during the dissection and later positioning of the pedicle during the inset. Postoperatively, controlling the position of the patient and movements is critical.
- •
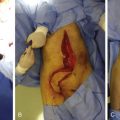
Initial temporary vascular insufficiency . Initial temporary flow disturbance, including flap ischemia and flap congestion, has been observed. This is considered to be an adaptive phenomenon to overcome the flow overload through a narrow perforator pedicle in the new flow redistribution. Its clinical course is variable. If a sluggish venous return or indistinct bleeding at the flap margin is observed after anastomosis, arterial insufficiency is suspected in spite of a positive milking test. As the distal perforator pedicle is very narrow in diameter and the dissection time of the pedicle may be long, the arterial component of the pedicle can easily undergo spasm. Loss of visible pulsation in the reliable perforator can be seen during dissection; the color of the flap can turn slowly from the pedicle side to the distal end of the flap after anastomosis. Initial temporary venous congestion is sometimes observed because of the centralization of venous drainage into tiny comitant veins of the perforator pedicle, such as in a traffic jam. Before final clipping of the pedicle for flap harvesting, a gradual color change should be observed in the flap, from slight congestion to the acceptable hue, in the process of adapting to the new flow distribution. These phenomena are particularly pronounced in large flaps, 25 cm or more in length, or in very long flaps (compared with their narrow widths), and they are more prevalent in the thoracodorsal and lateral thoracic perforator flaps (14.3%) than in the latissimus dorsi perforator flap (11.9%). Because the reliable perforator in the lateral thoracic region has a smaller diameter than that in the lateral thigh or abdomen, the initial temporary congestion or insufficiency can be more observed here. The return of the color takes from several hours to several days after anastomosis. Initial temporary ischemia may progress to mild congestion and this may last for several days or more.
- •
Deviation of the breast . In female patients, primary closure of the donor site may result in deformity of the breast with the thoracodorsal or lateral thoracic perforator flap; however, this can be released over time.
Preoperative Preparation
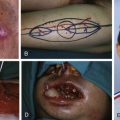
The reliable perforator is located alongside the muscle border using an audible Doppler and is marked. No evident pulsating sound is detected around a perforator, in contrast to the source vessel which has a longitudinal pulsating course. This method is not specific. Color Duplex or power Doppler imaging can aid in locating the perforators. The arterial or venous component of the pedicle can be confirmed in the spectral Doppler mode. According to these studies, the musculocutaneous perforator runs through the muscle obliquely, and the septocutaneous perforator has a more tortuous course. If one is well acquainted with perforator identification, this equipment may no longer be required preoperatively because the reliable perforator can be found in the expected area and other perforator rows can be found in this region.
There are no age limitations, but obese patients can be moderately contraindicated for harvesting a thin flap, especially in the thoracodorsal or lateral thoracic perforator flap. The reason for this is the fact that the purpose of harvesting flaps from this region is the ease of handling and the thinness of the flaps as compared to other parts of the body, and this is not the case in obese patients. However, thinning can be performed safely if necessary. In children or in elderly patients with atrophied adipose tissue, flap harvest is easier than usual. Diseases such as diabetes, heart disease, and paralysis are not major obstacles in the harvest of perforator flaps.
Flap Design
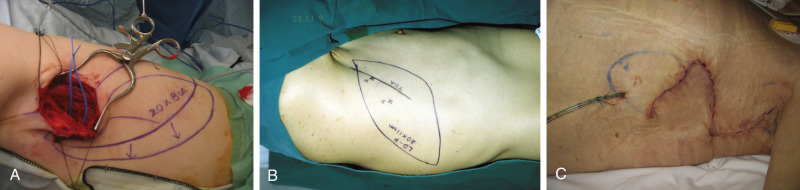
Anatomic Landmarks ( Fig. 56.5 )
The anterior border of the latissimus dorsi muscle and the center of the axillary fossa are marked. The anterior border of the muscle is made prominent by upward traction of the arm in the lateral decubitus position. Locating a musculocutaneous perforator near the proximal thoracodorsal vessels is of practical use because of its shorter transmuscular dissection. The expected area is 8–10 cm (6–8 cm in children) from the center of the axillary fossa and within 2 cm posterior to the anterior border of the muscle; the next is seen 2–5 cm inferior to the first. According to a study by Ishida, the distance from the axilla is variable, from 8 cm to 17 cm, and the distance from the muscle border is reported to be 1–4 cm. At the same level, anterior to the muscle border, the septocutaneous or direct cutaneous perforator can be found.
General Thoughts About Flap Design
All perforators can be used confidently as a pedicle for the perforator flap; however, their reliability is still questionable for the perfusion of a large flap extending over its original angiosome territory. Even though each perforator dominates a measure of skin territory, a reliable perforator has the great ability of being able to expand its perfusion over its own angiosome. Such a perforator is called a “reliable perforator” and is defined as a perforator arising from the carrier muscle with a “visible pulsation.” It has a larger than normal diameter (0.5–1.5 mm) and is often combined with the collateral sensory nerve, a useful indicator by which to find it. Despite the evident lack of hemodynamic studies, such a reliable perforator with a “visible pulsation” has a clinically peculiar ability to overcome the angiosome barrier through the subdermal vascular network. As the identification of this reliable perforator is critical, the flap design may be modified intraoperatively based on its size and location ( Fig. 56.6A ). The design can be oriented in various directions but horizontal or longitudinal elliptical design is mostly acceptable based on a single reliable perforator. However, the design is made according to the most relaxed or redundant skin tension by pinch test ( Fig. 56.6B ) because primary closure is better than skin graft in terms of postoperative scarring. The design may be modified with fishmouth ends to decrease the size of skin graft when a large flap is required. After harvest, a local flap is designed for the primary closure of a large donor site ( Fig. 56.6C ).