Vascular anomalies represent a heterogeneous group of disorders arising from developmental abnormalities in the endothelial and lymphatic systems, affecting various anatomic locations, and ranging in severity from benign to complex with significant morbidity. Vascular anomalies frequently affect visible areas of the body, such as the head and neck, leading to esthetic and functional concerns in addition to profound psychosocial impacts.
Key points
- •
A comprehensive, multidisciplinary approach is integral for evaluation and treatment of vascular anomalies given the complexity of these lesions and associated syndromes.
- •
Ultrasound is a useful diagnostic imaging modality evaluating the flow characteristics.
- •
Treatment may entail pharmacotherapy, laser therapy, interventional radiology-related procedures, and/or surgical management.
- •
Newer pharmacotherapy targeted at underlying molecular and genetic mechanisms are revolutionizing treatment of some complex vascular anomalies.
Introduction
Vascular anomalies represent a heterogeneous group of disorders arising from developmental abnormalities in the endothelial and lymphatic systems, affecting various anatomic locations, and ranging in severity from benign to complex with significant morbidity. Vascular anomalies are most often diagnosed in infancy or early childhood, with overall prevalence estimated to be approximately 5.5%. Vascular anomalies frequently affect visible areas of the body, such as the head and neck, leading to esthetic and functional concerns in addition to profound psychosocial impacts. Pediatric plastic surgeons play a pivotal role in the multidisciplinary management of vascular anomalies, especially those that result in physical deformities, functional impairment, or complications such as bleeding and infection.
Terminology and classification
Historically, the nomenclature surrounding vascular anomalies has been fraught with confusion, resulting in frequent misdiagnoses and suboptimal treatment strategies. For instance, the term “hemangioma” was often used as a catch-all label for various vascular lesions, regardless of biological behavior or clinical course. In the early 1980s, Mulliken and Glowacki introduced a biological classification system based on cellular characteristics and clinical behavior. Under their system, vascular anomalies were divided into 2 major categories: tumors, which exhibit endothelial proliferation and growth, and malformations, structural blood vessel anomalies that are present at birth and do not involve cellular proliferation.
The Mulliken–Glowacki classification was later refined by the International Society for the Study of Vascular Anomalies (ISSVA), which further subdivided vascular malformations into “low-flow” and “high-flow” lesions based on their hemodynamic characteristics. The development of these distinctions was a transformative step, not only for standardizing the terminology surrounding vascular anomalies but also in terms of their clinical significance. However, while the ISSVA classification remains the gold standard for diagnosing vascular anomalies and guiding appropriate therapeutic interventions, some anomalies—such as combined malformations or those associated with certain syndromes—do not fit neatly into any system and require a more individualized approach.
Pathogenesis and genetic basis
The pathogenesis of vascular anomalies is rooted in genetic abnormalities that disrupt normal vascular development, and advances in molecular biology have elucidated several key mutations—both inherited and somatic—involved in their formation and progression. Inherited mutations tend to display familial patterns and have been implicated in syndromic conditions involving multiple organ systems, such as capillary malformation-arteriovenous malformation (CM-AVM) and hereditary hemorrhagic telangiectasia. In contrast, somatic mutations—such as those associated with PIK3CA or TEK genes—result in mosaicism, in which only a subset of cells carry the mutation and are, therefore, more commonly associated with nonsyndromic malformations.
Vascular tumors, particularly infantile hemangiomas, are thought to arise from aberrant angiogenesis. The rapid growth phase seen in infantile hemangiomas is driven by increased expression of angiogenic factors, such as vascular endothelial growth factor (VEGF) and basic fibroblast growth factor. Tissue hypoxia is believed to be a key stimulus for the initial development of hemangiomas, as hypoxia-induced factors promote endothelial cell proliferation and new vessel formation. Over time, as the hemangioma matures, there is a shift toward antiangiogenic signals, resulting in lesion regression—the so-called “involutional phase” and replacement by fibrofatty tissue.
Lymphatic malformations, on the other hand, result from the disruptions in lymphangiogenesis and can be macrocystic or microcystic. Mutations in the VEGFR3 gene, which encodes a receptor for VEGF-C, have been identified in some cases of lymphatic malformations. These mutations lead to abnormal lymphatic vessel growth and dilation, resulting in the cystic spaces characteristic of these malformations.
The genetic basis of vascular anomalies continues to be an area of active research, with the hope that further discoveries will lead to more targeted and effective therapies.
Clinical features and diagnosis
The clinical presentation of vascular anomalies varies widely but—as discussed previously—can be broadly categorized into 2 distinct groups: vascular tumors, such as hemangiomas, which are characterized by proliferative growth, and vascular malformations, congenital anomalies of blood vessels that grow proportionally with the patient but do not involute spontaneously.
Vascular Tumors
Infantile hemangiomas are the most common type of vascular tumor, affecting up to 10% of infants, especially those born prematurely or with low birth weight. , They typically present within the first few weeks of life as small red or bluish lesions on the skin or mucosal surface. They undergo a rapid growth phase during the first few months of life, followed by a plateau phase and eventual involution, typically beginning around 12 months of age. While most hemangiomas are benign and self-limiting, those located near critical structures such as the eyes, airway, or liver may cause functional impairment and require medical or surgical intervention.
Congenital hemangiomas, in contrast, are fully formed at birth and do not follow the growth-involution pattern seen in infantile hemangiomas. These lesions can be classified as either rapidly involuting congenital hemangioma (RICH) or noninvoluting congenital hemangioma (NICH). RICH lesions typically regress within the first year of life, whereas NICH lesions persist and may require surgical excision for cosmetic or functional reasons.
In addition to the more common vascular tumors, there are several others of clinical significance with distinct clinical behaviors, presentations, and management challenges.
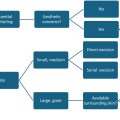
Kaposiform hemangioendothelioma (KHE) is a rare, but aggressive, vascular tumor, typically presenting in infancy or early childhood. It is locally invasive and is most often associated with Kasabach–Merritt phenomenon (KMP), a life-threatening coagulopathy characterized by severe thrombocytopenia and consumptive coagulopathy that results from platelet trapping within the abnormal tumor vasculature and an ensuing clotting cascade. KHE typically presents as a firm, violaceous mass on the trunk or extremities. Unlike hemangiomas, however, KHE tends to grow slowly and progressively, eventually violating tissue planes and invading surrounding tissues such as muscle and bone. Imaging, particularly MRI, is crucial for assessing the extent of the tumor and guiding treatment planning. The association of KHE with KMP necessitates early diagnosis and prompt medical intervention with treatment strategies focused on controlling tumor growth and pharmacologic management of the coagulopathy with corticosteroids, vincristine, and, most recently, sirolimus (ie, rapamycin). While surgical excision may be considered in cases of a well-defined and accessible tumor, this is often not feasible due to its infiltrative nature.
Tufted angioma (TA) is another rare vascular tumor, closely related to KHE, that presents primarily in infants and young children. It is characterized by the development of slow-growing, reddish-purple nodules or plaques, most commonly located on the neck, upper trunk, or extremities. Histologically, TA consists of small, angiomatous lobules (“tufts”) that are dispersed throughout the dermis and subcutis. Unlike KHE, TA are usually benign and do not invade surrounding tissues, but they can be associated with pain and significant discomfort in areas prone to friction or trauma. In some cases, TA is also associated with KMP, though less commonly than KHE. Management of TA depends on tumor size, location, and symptoms but, given their benign nature, often consists of observation or pharmacologic management. Surgery is typically unnecessary unless the lesion becomes symptomatic.
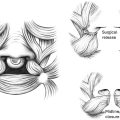
The term pyogenic granuloma (PG) is a misnomer, given that they are neither infectious nor a response to chronic inflammation. Rather, this vascular entity—more appropriately termed lobular capillary hemangioma—represents a rapid overgrowth of capillaries that develops in response to minor trauma or irritation. PG typically presents as small, red, and friable nodules on the skin or mucous membranes. PG is particularly common in children and pregnant women and may arise spontaneously or in response to factors such as hormonal changes (eg, during pregnancy), infections, or drug reactions. While benign, PG often requires treatment due to their tendency to bleed. First-line management of PG typically involves surgical excision or curettage, with or without electrocautery for hemostasis. In cases where surgical excision is not feasible or a less invasive approach is preferred, laser or cryotherapy may be effective. PG recurrence is common if the lesion is not fully excised (eg, “shave excision”), and repeat treatments may be necessary. In cases of recurrent or persistent PG, a biopsy is recommended to rule out possible mimics including Spitz nevus, amelanotic melanoma, or other skin malignancies.
Vascular Malformations
Vascular malformations, while less common, are present at birth and tend to persist or progress over time, growing proportionally with the child. Capillary malformations, commonly referred to as “port-wine stains,” present as flat, pink or red patches on the skin, most commonly on the face or limbs. Over time, these lesions may darken and thicken, causing significant cosmetic concerns. Venous malformations present as soft, compressible masses that may enlarge with activity or dependent positioning. These lesions can cause pain, swelling, and, in some cases, thrombosis or bleeding. Lymphatic malformations are characterized by cystic masses that may fluctuate in size, especially in response to infection or trauma. In contrast to these 3 categories of vascular malformations—all of which are considered “low-flow,” arteriovenous malformations (AVMs) possess high-flow hemodynamic properties, resulting in pulsatile masses that may be associated with overlying skin changes or, in severe cases, signs of high-output cardiac failure.
Combined Malformations
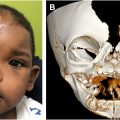
Combined malformations are complex vascular anomalies involving more than one type of vascular channel, including capillary, venous, lymphatic, and/or arteriovenous components. These malformations often require a nuanced diagnostic and management approach due to their multifaceted nature and potential for significant morbidity. Furthermore, combined malformations may be associated with various known syndromes. Each syndrome is characterized by a set of distinct clinical features, involving multiple organ systems and a range of disease processes ( Table 1 ). , As such, multidisciplinary care is essential to coordinate surgical, medical, and genetic treatment strategies.
| Syndrome/Condition | Key Vascular Malformations | Associated Systemic Findings |
|---|---|---|
| PHACE Syndrome |
|
|
| Sturge–Weber Syndrome |
|
|
| Klippel-Trénaunay Syndrome (KTS) |
|
|
| Parkes Weber Syndrome |
|
|
| Megalencephaly-Capillary Malformation (M-CM) |
|
|
| Capillary Malformation-Arteriovenous Malformation (CM-AVM) |
|
|
| Cutis Marmorata Telangiectatica Congenita (CMTC) |
|
|
| PTEN Hamartoma Tumor Syndrome |
|
|
| CLOVES Syndrome |
|
|
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree