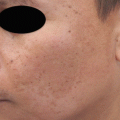
Fig. 4.1
Immunostaining for NKI-beteb and tyrosinase-related protein (TRP)-2 before and after treatment with a combination of soybean extract and niacinamide for 8 weeks. Immunostaining for (a) NKI-beteb before treatment (×100), (b) NKI-beteb after treatment (×100), (c) TRP-2 before treatment (×100), and (d) TRP-2 after treatment (×100) [38]
4.3.2.2 Basement Membrane Disruption
Some studies have investigated the status of the basement membrane in the melasma skin. Sanchez et al. demonstrated the presence of vacuolar degeneration of the basal cells and the focal vacuolar degeneration of the basement membrane in 3.9 % (3/76) of melasma skin specimens [39]. In contrast, Kang et al. did not observe any disruptions of the basement membrane in their evaluation of skin samples from 56 Korean patients with melasma using diastase-resistant periodic acid-Schiff (D-PAS) staining and electron microscopy [33]. However, the same authors reported that pendulous melanocytes associated with basement membrane abnormalities are a characteristic feature of melasma [6].
Another study of melasma patients with Fitzpatrick skin types IV and V revealed that PAS staining showed damage in the basal membrane in 95.8 % of the melasma lesions versus 58.3 % of the perilesional skin, and antibody to collagen type IV showed damage in 83 % of the melasma lesions versus 66 % of the perilesional skin [36].
Basement membrane disruption could be caused by elevated levels of matrix metalloproteinase (MMP)-2 and MMP-9, which degrade type IV and VI collagen during chronic UV exposure [40]. MMP-2 expression was found to be increased in the lesional skin compared with the perilesional normal skin (0.018 ± 0.014 vs. 0.004 ± 0.005, p = 0.006) [6]. Since free melanin and melanophages are present in the dermis of the melasma skin, disruption of the basement membrane could facilitate the descent or the movement of active melanocytes and melanin into the dermis, which is reflected in the constant hyperpigmentation of melasma [33, 36].
4.3.2.3 Solar Elastosis
Many observations strongly suggest that sun exposure is the primary trigger of melasma. Melasma affects particularly the face, a sun-exposed area, and the condition worsens in the summer. Solar elastosis is more marked in areas of melasma, compared with the unaffected facial skin [35]. Kang et al. reported a moderate-to-severe degree of solar elastosis in 93 % of melasma patients included in their study [33]. The melasma skin showed a significantly higher degree of solar elastosis than the perilesional normal skin (83 % vs. 29 %, p < 0.05) [36]. Furthermore, the amount of elastotic material was significantly higher in the melasma skin than that in the perilesional normal skin (13.3 ± 2.8 % vs. 10.2 ± 2.9 %, p < 0.001). Moreover, thick, highly curled, and more fragmented elastic fibers were observed in Verhoeff-van Gieson-stained sections of the melasma skin [33].
4.3.2.4 Increased Vascularization
Accumulating evidence has shown that the number of blood vessels is higher in the melasma lesions than in the perilesional normal skin [41–43]. The increase in the number of vessels was more prominent than the increase in vessel size. This finding represents that the erythema noticed in melasma patients could be due to angiogenesis and telangiectasia [41]. An immunohistochemical study of factor VIIIa-related antigen showed a considerable increase in the number of enlarged blood vessels, vessel size, and vessel density in the melasma skin compared with the perilesional normal skin [41].
The elevated expression of vascular endothelial growth factor (VEGF) in keratinocytes has led to the hypothesis that VEGF may play a role in the behavior of the melanocytes in the skin, because the functioning VEGF receptors were demonstrated in melanocytes in vitro [44]. Elevations in the levels of c-kit, SCF, and inducible nitric oxide synthase have also been observed, which could affect vascularization [5, 45]. Moreover, blood vessels or endothelial cells modified by UV irradiation may release cytokines and soluble factors, such as plasminogen, which is a possible cause of hyperpigmentation in melasma [46].
Tranexamic acid (TXA) inhibits plasmin, a key molecule involved in angiogenesis that converts extracellular matrix-bound VEGF into its free forms [47]. TXA has also been reported to suppress neovascularization-induced bFGF [48]. In a recent clinical trial that evaluated the efficacy of systemic TXA in the treatment of melasma, we demonstrated significant decreases in the lesional melanin index and in the erythema index after the oral administration of 250 mg TXA, three times per day, for 8 weeks [49]. A histologic analysis showed significant reductions in the level of epidermal pigmentation and vessel numbers (Fig. 4.2a–d). These findings suggest that the interactions between increased levels of vascularization and melanocytes may have an influence on the development of hyperpigmentation.
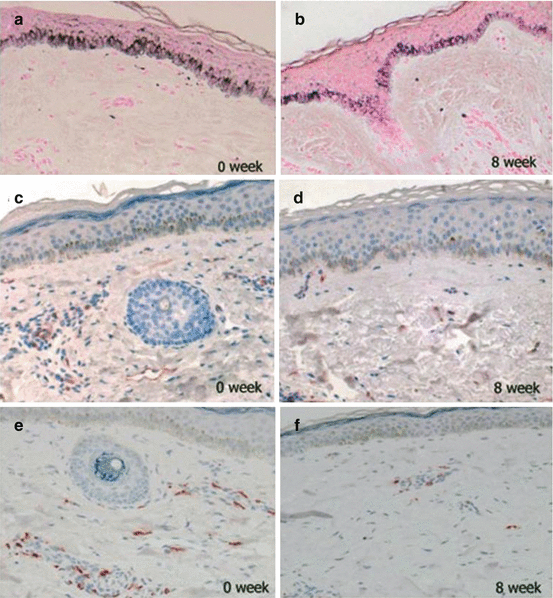

Fig. 4.2
Histologic changes after 8 weeks of treatment with tranexamic acid. (a, b) Fontana-Masson staining shows reduced epidermal pigmentation (×100). (c, d) Anti-CD31 staining shows reduced levels of vascularity (×100). (e, f) Antitryptase staining shows reduced mast cell numbers (×100) [49]
4.3.2.5 Mast Cell Prevalence
Mast cells are observed more frequently in the melasma skin than in the nonlesional skin, especially in the dermal elastotic areas (Fig. 4.2e, f) [49]. The median prevalence of dermal mast cells was significantly higher in the melasma skin than in the perilesional normal skin (173 ± 57 % vs. 145 ± 57 %, p = 0.04) [35]. Using an antitryptase antibody, the number of mast cells was 58 ± 39.9 cells/mm2 in the melasma skin compared with 37 ± 28.8 cells/mm2 in the perilesional normal skin (p < 0.04) [36].
The role of mast cells in the development of melasma has not been elucidated. Since repetitive UV irradiation induces the production of mast cell tryptase, which degrades type IV collagen, elevated mast cell numbers and tryptase levels could weaken the basement membrane in the melasma skin [50]. Mast cells could trigger solar elastosis by inducing the production of elastin by fibroblasts, either directly or via other cell types or cytokines [51, 52]. Solar elastosis did not develop in mast cell-deficient mice that were repeatedly irradiated with UV [53]. The elevated numbers of mast cells, together with the presence of infiltrating leukocytes and dilated blood vessels, might reflect chronic skin inflammation that underlies the development of melasma [35]. Mast cells can also induce vascular proliferation by secreting angiogenic factors, including VEGF, fibroblast growth factor-2, and transforming growth factor-β [54].
4.4 Conclusion
In addition to the genetic background and exposure to ultraviolet radiation, there may be an association between female sex hormones/hormonal receptors and melasma. Furthermore, a gene profiling study showed that melasma might be associated with changes in gene expression, which are related with melanogenesis, Wnt signaling pathway, and lipid metabolism. Although melasma is characterized by epidermal pigmentation, there are distinctive histologic findings in melasma, such as epidermal hyperpigmentation, basement membrane disruption, solar elastosis, increased vascularization, and high prevalence of mast cells. Such findings suggest that these have important and interactive roles in the pathogenesis of melasma.
References