Anatomy of peripheral nerve
Peripheral Nerve Physiology
In the peripheral nervous system, effector information is transmitted through motor units, each of which is composed of an alpha motoneuron, its axon, and whatever number of extrafusal muscle fibers it innervates. Afferent information is integrated through sensory receptors, its axons, and its cell bodies, which are located in dorsal root ganglia.
At rest, nerve fibers maintain their resting membrane potential. Generation of a stimulus results in changes in ion flow from outside to inside the axon. This increases positivity within the interior of the nerve fiber, decreasing the potential difference between the interior and the exterior, thereby transmitting the impulse that generates an action potential [2].
Basic Principles of Pathophysiology, Mechanisms, and Classification of Peripheral Nerve Injuries
Demyelination and Axonal Damage
After an injury, a nerve trunk reacts with two separate pathophysiological processes that can occur individually or concurrently and that are independent of the etiology and mechanism of injury.
The first process is demyelination (or loss of the myelin layer), either isolated or associated with axonal damage. If its Schwann cell remains viable and just demyelination is observed, the nerve will ultimately recover its function completely, merely by remyelinating the affected segment.
Any injury that causes disruption of an axon’s integrity results in degeneration of the distal segments through a process called Wallerian degeneration, which is completed within 3 weeks of an injury. This degeneration process can also occur in the cell body, proximal axon, and distal target organs. Simultaneously to this degeneration process, there is a regeneration process, influenced by the remaining Schwann cells, which influences the formation of endoneurial tubes, which act to guide outgrowing axons searching for the distal stump, so they ultimately may reinnervate their target organ(s) [2].
Mechanisms of Injury
A PNI occurs when a nerve is transected, stretched, crushed, or compressed. Therefore, the normally existing communication that is conducted by the nerve, between the peripheral and central nervous system, is lost. Other less-frequent mechanisms of nerve injury, like toxicity by substance injections, have been described too. Silicone migration and granuloma formation are considered another special type of nerve injury, though still related to one of the previously described mechanisms [1].
Such injuries often have consequences for the affected individual, including potential losses of sensory, motor, and autonomic function. Pain is another component of certain PN syndromes, commonly caused by sustained compression when scars or other external agents are present [3].
In these ways, peripheral nerve injuries may lead to both suffering and altered quality of life for the patient [4].
Grading of Lesions
Regardless of etiology, two current classification systems for grading nerve injuries, used since the last century and described by Seddon and Sunderland, can be applied to all pathologic processes that affect the PN, not only the traumatic ones for which they were originally described. These systems’ most important applications are predicting the probability of spontaneous recovery and, thus, the need for surgical exploration and repair and estimating a patient’s prognosis.
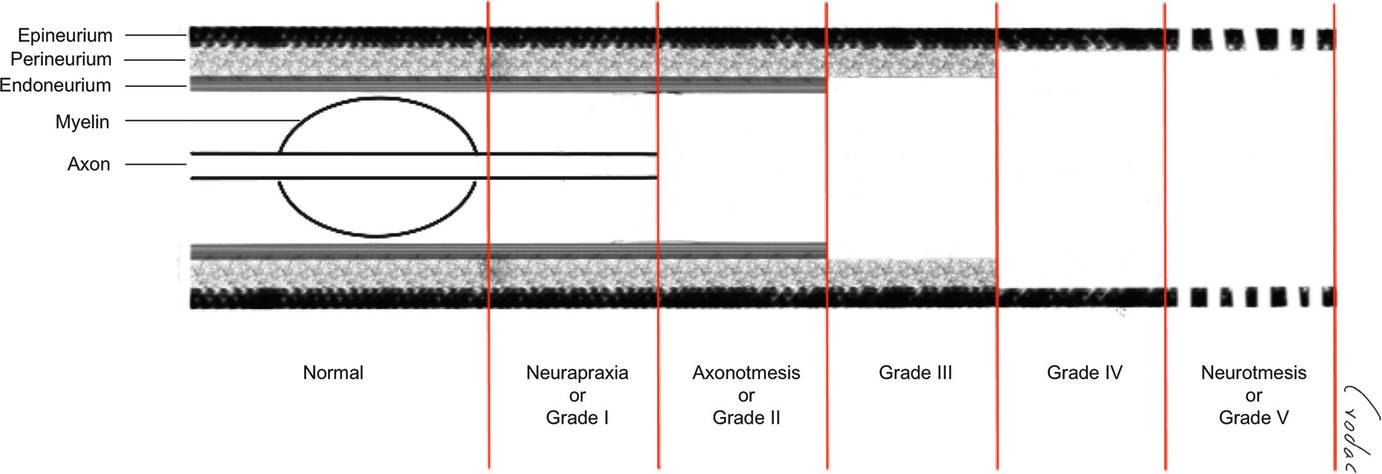
Classification of nerve injuries (Seddon & Sunderland)
It is important to understand these basic principles of nerve injuries, because they must be appreciated to comprehend peripheral neuropathy secondary to the cosmetic use of silicone and the subsequent management of that neuropathy.
Inflammatory Responses to Biomaterials
Silicone Toxicology and Immunology
Silicone is a derivative of silicon that is used for a variety of medical applications, because it has been claimed to be biologically inert. This said, it has been linked to local inflammatory and fibrotic reactions and is believed to be associated with a variety of autoimmune diseases. Silicone derivatives are polymerized, through chemical reactions, to form chains of varying length; and as the chain length increases, the polymer becomes increasingly viscous and hydrophobic. Silicone can be produced either in a liquid or solid state, depending on the size and extent of cross-linking of the polymer. Polydimethylsiloxane is the most common polymer used in medical products [8].
As mentioned previously, although silicones appear to be nonantigenic, they are not completely biologically inert, and this can be explained by the presence of contaminants. Nevertheless, what is not fully understood at present is if the adverse events related to silicone injection result from an aberrant host response in a susceptible individual, from an infection with an unusual response, or from a normal host response to a contaminant [9].
All foreign bodies can elicit an immunologic reaction, and granulomas are a generic response to foreign materials of all types. The tissues surrounding a silicone implant typically experience an inflammatory response. It is the degree of this response that determines whether or not local symptoms occur. The cellular inflammatory response, including the production of macrophages, can lead to the development of fibrosis around the implant. In addition, the capsule may elicit a contractile or retractive fibrotic process. The scar contracture that results varies in degree, from mild to severe, and is frequently accompanied by symptoms of pressure sensation and pain [9, 10, 11].
Silicone Migration and Granuloma Formation
Other responses to silicone implants include the development of silicone granulomas outside the area of implantation or injection and the involvement of regional lymph nodes; such responses have been shown to result after rupture of an implant and the extracapsular spread of silicone [10, 12].
Unpredictable reactions can occur weeks to years after silicone implantation. Migration of the product to distant sites can produce serious systemic complications, which include granulomatous hepatitis, organ compression, respiratory problems, and silicone emboli.
Silicone granulomas and migration are the most commonly reported complications associated with the use of injectable liquid silicone. These lesions manifest clinically as recurrent cellulitis-like reactions, characterized by pain, tissue induration, nodules, ulceration, and/or local lymphadenopathy. Systemic signs like fever, weight loss, nausea, and vomiting may also be present.
It is known that silicone undergoes biological oxidation to silica, which becomes incorporated into the reticular endothelial system, eliciting a granulomatous response. Some authors argue that the likelihood of migration is linked to the amount of silicone injected and to its location [13].
Treating systemically spread silicone granulomas is usually complex and includes a combination of strategies. Different medications—like topical and systemic steroids, minocycline, allopurinol, methotrexate, and tacrolimus, among others—have been used. However, these treatments will not be reviewed in this chapter [14].
Effects on Peripheral Nerves
To our knowledge, Sanger et al. are the only ones who have described the PN changes that result from silicone use complications. The main mechanism for PN injury in such cases is secondary nerve compression, caused by the inflammatory response this substance triggers in adjacent tissues. These effects were studied in rats by experimentally injecting silicone in extraneural and intraneural locations. Extraneural silicone elicited an inflammatory response mediated by histiocytes, eosinophils, lymphocytes, and foreign-body giant cells; this response peaked at 4 weeks, after which perineural fibrosis was established without silicone penetrating the epineurium. Intraneural silicone caused a similar inflammatory response, but the reaction was more delayed and characterized by more extensive fibrosis. No evidence of direct toxicity to peripheral nerves has been documented after silicone migration or direct injection [15, 16].
Depending upon the location and amount of migrating material, silicone can spread to certain regions more easily than others, causing adjacent peripheral nerve disfunction, the clinical presentation of which will largely depend upon the function(s) and location of the affected nerve. Compromise of the brachial plexus, median nerve, radial nerve, and tibial nerve in the tarsal tunnel, among others, has been described [17, 18].
Evaluation of and Diagnostic Procedures to Determine Peripheral Nerve Involvement Post-Silicone Injection
Initial Approach
As described previously, since the mechanism of silicone-linked nerve injury is related to compression of the affected nerves by silicone in the absence of intrinsic toxicity, the clinical picture tends to present insidiously, as with any other chronic nerve compression. Depending on severity, clinical features include impaired sensory, motor, and/or autonomic function. Of course, a history of silicone implants or silicone injections weeks, months or, more frequently, years or decades before the onset of symptoms must be present, as this is generally the only way in which silicone enters human tissues. As silicone migration to distal sites is accompanied by granuloma formation, usually nodular masses are identifiable along an affected nerve’s trajectory.
Burning pain is a recognized symptom in chronic PN compression syndromes. It is initially intermittent and localized at the compression site but typically irradiates through the nerve’s sensory distribution, accompanied by sensory disturbances like dysesthesia or paresthesia. The pain that irradiates within a nerve’s territory, called neuropathic pain, has certain characteristics (e.g., often described as electric or burning) that must be distinguished from those produced by local irritation of the soft tissues by the silicone material, which tend to be more diffuse, lack sensory abnormalities, and be accompanied by trophic or cutaneous changes. A positive Tinel’s sign (digital percussion at the compression site elicits neuropathic symptoms) may confirm the compression site with superficial nerves.
Less commonly, motor impairment may be present, which may manifest as weakness and/or atrophy, both of which depend upon the severity and chronicity of compression. The presence of either weakness or atrophy is an unequivocal sign of severe compromise.
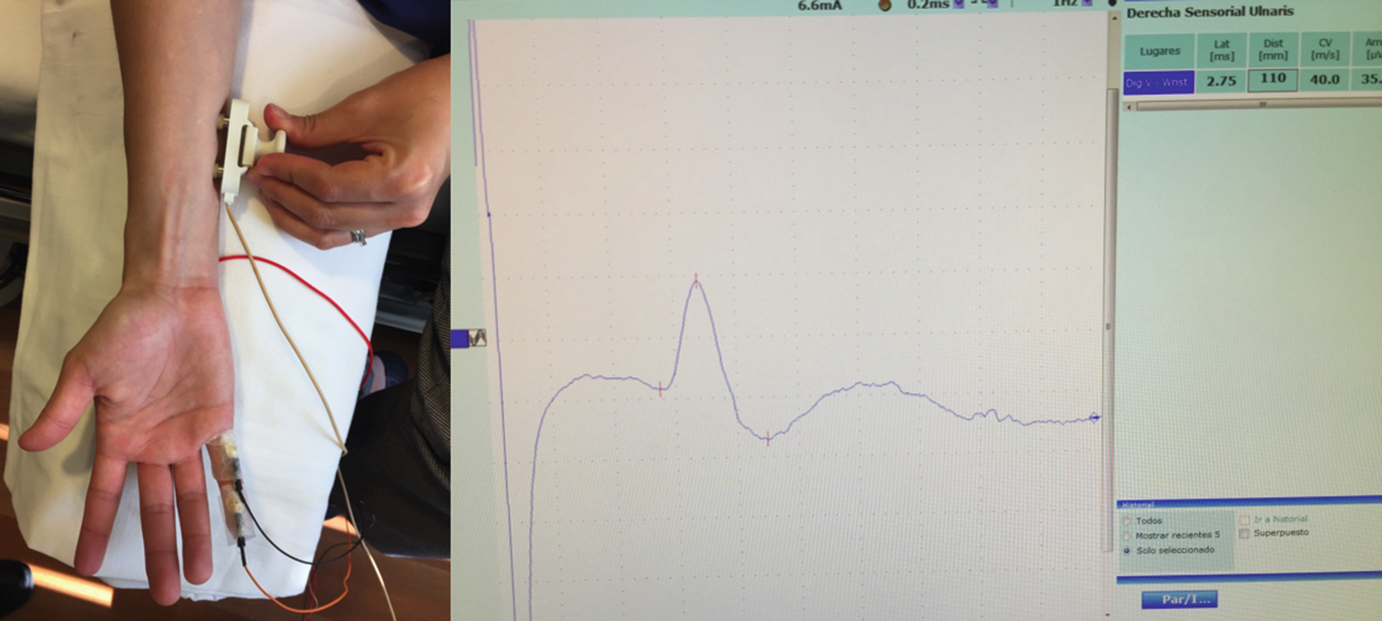
Once a physician suspects peripheral nerve compression from silicone material, the diagnosis is confirmed by electrodiagnostic studies and imaging studies.
Electrodiagnostic studies (EDS) are an essential tool for evaluating peripheral nerve diseases and useful complements to the clinical evaluation that may help to detect signs not confirmed by neurological examination and guide diagnosis and treatment. The utility of EDS is broad and includes pinpointing the site of injury; identifying the underlying pathophysiological process; establishing the timing, severity, and extent of injury; generating a list of possible diagnoses; and assessing symptom progression, thereby helping to predict prognosis and establish the need for and timing of surgical exploration.

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree