5 Non-permanent fillers
Belotero®/Esthélis® and Teosyal®
Summary and Key Features
• Belotero®, Esthelis®, and Teosyal® are three hyaluronic acid families produced in Switzerland
• Good clinical data are available only for Belotero® Basic and Teosyal® Deep Lines
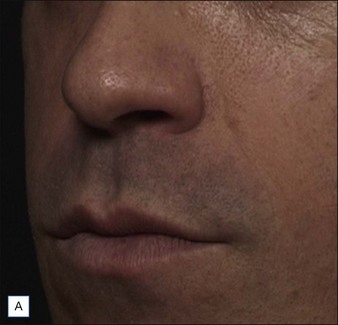
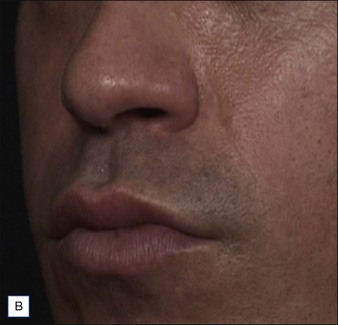
• At 6 months Belotero® Basic and Teosyal® Deep Lines still show at least a one-grade difference after treatment of the nasolabial folds
• Belotero® Basic and Teosyal® Deep Lines have a good safety profile
Hyaluronic acid dermal fillers
How to differentiate the different HA fillers? There are certain possibilities:
 Those with good clinical data and those without
Those with good clinical data and those without
 Those for more superficial and those for deeper injections
Those for more superficial and those for deeper injections
 Those with a few products and those with product families
Those with a few products and those with product families
 Those easy to inject and those more difficult to inject
Those easy to inject and those more difficult to inject
Belotero®
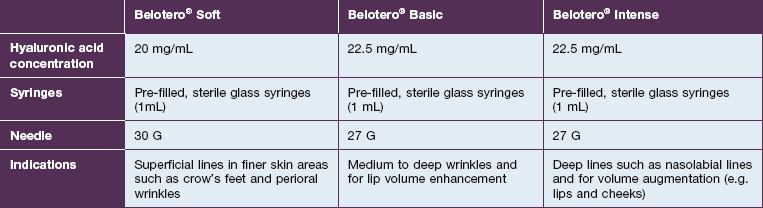
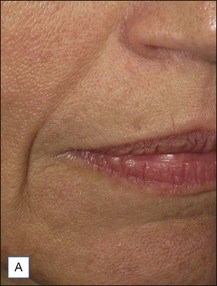
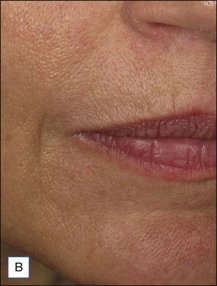
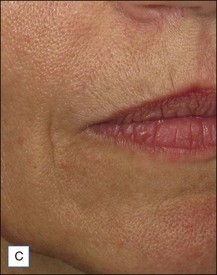
Belotero® is produced by the Swiss company Anteis SA and distributed by Merz Pharmaceuticals. The stabilizer is BDDE and according to the manufacturer two cross-linking processes are used leading to a cohesive and polydensified matrix, which is supposed to ease injection while maintaining long-lasting results. Belotero® offers three different formulations: Belotero® Soft, Belotero® Basic and Belotero® Intense (Table 5.1, Figs 5.1 and 5.2).