3 NASHA™ family
Summary and Key Features
• NASHA™ are a safe, reliable class of dermal fillers that have low potential for allergic response
• NASHA™ fillers are non-permanent and thus ideal to use for treatment-naïve patients and for beginning injectors as they can be easily dissolved with hyaluronidase
• The type of injection technique selected (serial puncture, linear, fanning or cross-hatching) should be based on anatomical site
• Common areas for injection include the nasolabial folds, followed by the melolabial folds, jowls, and lips. Injection into the glabella is contraindicated owing to the risk of necrosis unless it is used with a tiny needle and injected superficially pulling back on the plunger before injection
• Depth of the fold or amount of volume replacement required are the key components to consider prior to product brand selection
• NASHA™ fillers can be layered on top of each other or on top of other semipermanent fillers
• Hyaluronic acid fillers can last from 3 to 6 months depending on location and amount of product previously injected; they last for longer in areas of low mobility and shorter in areas of greater movement
• Postoperative sequelae are usually edema and bruising lasting only a few days
Basic science
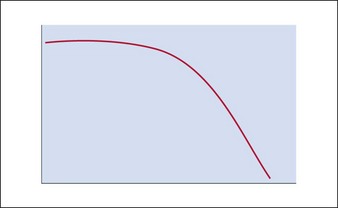
The hyaluronic acid molecule is stabilized with cross-linked hydroxyl groups. It is this cross-linking that confers longevity to the product in the skin and the degree of cross-linking one of the distinguishing features of NASHA™ products. Because of this structural matrix, the degradation curve of implanted HA is not linear. Rather, the product retains its effect until the structural complex around the HA molecule is broken down. Once the unbound HA is exposed, efficacy is then lost (Fig. 3.1). As a result, patients will often suddenly notice a change in their appearance rather than experience a gradual loss over time. In addition, patients sometimes feel they look worse after HA injection than they did prior to it; however, this is more of a function of selective memory. This is why taking pre-injection photographs is so important. As with natural hyaluronan, HA fillers can be instantly dissolved with the enzyme, hyaluronidase.
Choosing the right NASHA™
The main difference between commercially available NASHA™ products on the current US market is the viscosity of the product. The higher the concentration of HA, the stiffer is the product and the longer it lasts in tissue (Table 3.1). The larger the particle and the greater the concentration of the filler, the greater the lift you get. It should be noted, however, that not all of the HA in a given filler is cross-linked. Some is free, fragmented or only lightly cross-linked so that the gel can actually flow out of the syringe with ease. As discussed above, the cross-linking is what confers longevity to the product and this does vary between brands. In Restylane® and Juvéderm®, 1,4-butanediol diglycidyl ether is the cross-linking agent, which forms ether linkages in the HA chain. This same type of ether linkage is formed by diglycidyl sulfone found in Prevelle® Silk. In addition to cross-linking, it is worth mentioning that not all HA products are packaged in the fully hydrated state. As mentioned earlier, NASHA™ fillers create volume via hydration and, for this reason, patients will often look better or more filled 24 hours after treatment with products like Restylane® and Juvéderm, which are not completely saturated in the syringe. With that in mind, it is prudent not to overcorrect when using NASHA™ products.
Table 3.1 Hyaluronic acid (HA) concentration of commercially available NASHA™ products
| Trade name | HA concentration | Particle size* |
|---|---|---|
| Restylane® | 20 mg/mL | ~260 µm |
| Perlane® | 20 mg/mL | ~1000 µm |
| Juvéderm® | 24 mg/mL | 50–500 µm (very cohesive) |
| Juvéderm® Ultra Plus | 24 mg/mL | Homogeneous gel |
| Prevelle® Silk | 5.5 mg/mL | ~250 µm |
| Hydrelle® | 28 mg/mL | Not available |
| Hylaform® Plus | 5.5 mg/mL | ~700 µm |
* Even very cohesive products are particulate. The particle sizes of commercially available NASHA™ fillers are shown. In comparison, the particulate sizes of non-NASHA™ products are as follows: Radiesse® (25–45 µm), Artefill® (over 30–50 µm), Sculptra® (40–60 µm), Dermalive® (20–120 µm).
Patient evaluation
Prior to injection with any dermal filler, a careful patient history should be obtained. Pertinent items to note in this history include a history of herpes simplex virus, pregnancy or breastfeeding, history of keloid formation, presence of any autoimmune disease, and allergy to lidocaine as most of the fillers now come pre-mixed with lidocaine (Box 3.1). A medication history should include use of multivitamins, fish oil, vitamin E, blood thinners, aspirin, ibuprofen, and Gingko biloba, as all of these can predispose the patient to bruising. Unless medically necessary, the patient should be advised to discontinue these medications 14 days prior to treatment. In addition, alcohol use within 48 hours of injection can cause an increase in bruising tendency.