Microtia is a congenital condition characterized by a spectrum of ear malformations, ranging from minor structural anomalies to anotia. The etiology is multifactorial, with interplay of genetic and environmental factors being implicated. Early multidisciplinary involvement should include pediatrics, plastic surgery, otolaryngology, audiology, and speech pathology, to ensure that the child is appropriately supported as they grow. This article provides an overview of the condition, a brief historic review, and illustrated descriptions of 2 common surgical approaches: porous polyethylene reconstruction with a temporoparietal fascia flap, and autologous reconstruction in 2 stages following Firmin’s modifications of the Nagata technique.
Key points
- •
Microtia is a congenital condition characterized by a spectrum of ear malformations, ranging from minor structural anomalies to anotia. Microtia can be isolated or associated with other malformations (20%–60% of cases).
- •
Microtia is concomitant with atresia in up to 90% of cases. The extent of hearing loss should be diagnosed and addressed early with consideration of a hearing aid. Surgical options to address atresia exist. Workup, timing, and coordination with reconstruction should be discussed with an experienced otology team.
- •
Autologous reconstruction usually involves 2 to 4 stages, starting at around the age of 7 to 10 years. It consists of harvesting rib cartilage, carving and assembling a construct, and placing it under mastoid skin. Autologous reconstruction offers a durable option with the patient’s own tissue, a low rate of late complications, and greater ability to salvage if there is exposure or infection. However, it is artistically demanding, results are operator dependent, has a steep learning curve, and carries the disadvantage of donor-site morbidity.
- •
Alloplastic reconstruction can be performed in a single stage at an earlier age (∼4 years), and even concomitantly with atresia surgery. It involves the implantation of a porous polyethylene framework under a temporoparietal fascia flap, followed by skin grafting. While it offers a reproducible esthetic result in experienced hands, exposure, infection, and complete reconstruction loss are possible even years after surgery and can be difficult to salvage. Secondary reconstruction after alloplastic failure is very challenging.
Definitions
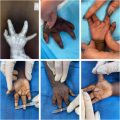
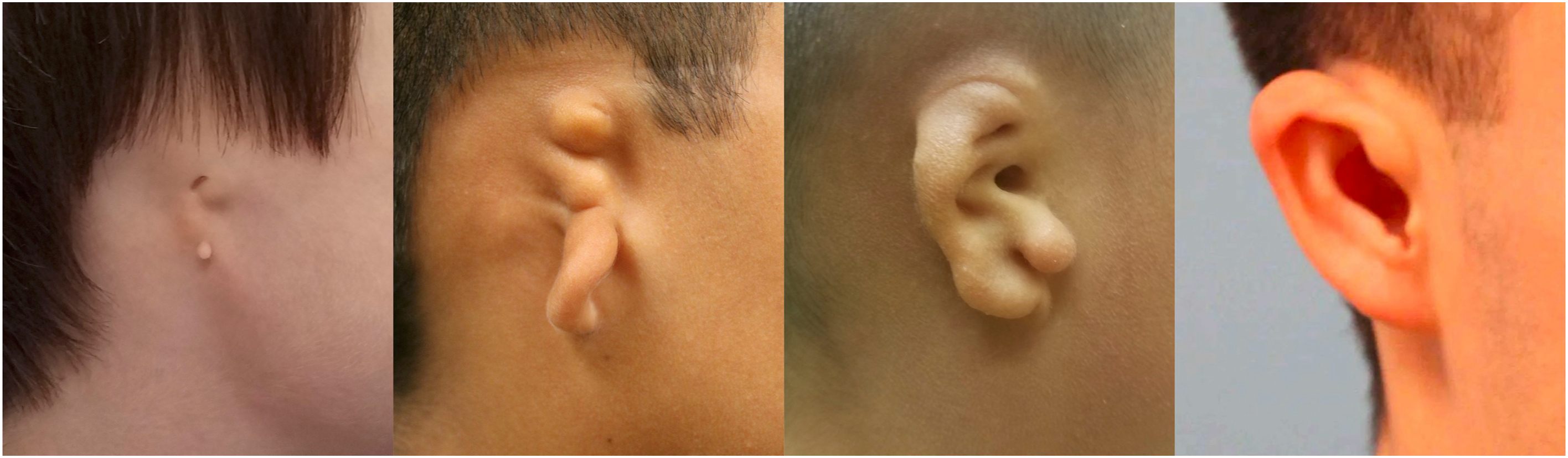
Microtia—stemming from the Greek words mikrós or “small,” and ōtikós “of the ear” —is a congenital anomaly characterized by a spectrum of malformations of the auricle, ranging from minor structural anomalies to complete absence of the ear (anotia). Anotia is usually considered to be under the same umbrella of the microtia spectrum, at its most severe end ( Fig. 1 ).
Embryology
The development of the ear is a complex process involving the coordinated formation of the external, middle, and inner ear. On day 23 of gestation, the inner ear begins to develop from a thickened portion of ectoderm adjacent to the hindbrain known as the otic placode. This invaginates to form the otic vesicle, which differentiates into the osseous and membranous labyrinths, with the osseous parts arising from the ossification of the capsule and the membranous part from the neuroectoderm of the otic placode. , These structures further differentiate into the cochlea, vestibule, and semicircular canals.
The middle ear consists of the tympanic cavity and ossicles. The tympanic cavity originates from the endoderm of the first pharyngeal pouch. The ossicles, composed of the malleus, incus, and stapes, are derived from different branchial arches around week 6 of gestation: the malleus and incus from the mesenchyme of the first branchial arch, and the stapes from the second branchial arch. ,
The pinna develops from the first and second branchial arches, and the external auditory canal from the first branchial groove. In the sixth week of development, 6 small prominences called the hillocks of His form on the dorsal surfaces of these arches. Hillocks 1, 2, and 3 arise from the first branchial arch, and hillocks 4, 5, and 6 form from the second branchial arch. These hillocks eventually fuse to form the auricle. In the original model by His, the first 3 hillocks contributed to the root of the helix, the anterior helical rim, the tragus, and the intertragic notch. The posterior hillocks from the second pharyngeal arch were through to form the remaining posterior portion of the auricle, including the concha, antihelix, the inferior crus, triangular fossa, superior crus, helix, antitragus, and lobule. However, newer studies have challenged this model, and the importance of the hillocks to specific structures is now downplayed. , In the eighth week of gestation, the groove between the first and second branchial arches invaginates to create the external auditory meatus. This invagination allows for the simultaneous migration and fusion of the 6 hillocks of His, forming the cartilaginous precursor to the auricle. By the 12th week of gestation, the auricle’s unique topographic characteristics begin to develop, and by the 18th week, it detaches from the head. While the ear begins its development in the anterior neck, it slowly migrates dorsal and cephalad to reach the adult position by about the 20th week of gestation. By the 20 second week, the auricle’s development is complete.
Embryologically, the external ear is formed earlier than the middle ear, so presence of microtia would then indicate a failure of development that heralds subsequent issues with development of the middle ear. Therefore, severity of the deformity of the external ear and middle ear are generally proportional to one another. The distant embryologic origin of the inner ear from the middle and external ear clarifies why inner ear structures are typically preserved in microtia, while the common origins of the middle and external ear from the first and second branchial arches explain the high coincidence of congenital aural atresia (CAA) and conductive hearing loss in microtia. The contribution of the first and second pharyngeal arches to the skeletal, muscular, and neural components of the face also explains the high coincidence microtia with facial clefts, macrostomia, facial palsy, and asymmetry, as seen in craniofacial microsomia (CFM) and the oculo-auricular-vertebral spectrum (OAVS).
Epidemiology
The prevalence of microtia has been reported to vary between 0.8 and 17.4 per 10,000 in different populations, being least common in Western Europeans and Caucasians, and more common in those of Andean, Native American, and Asian descent. Microtia is more common in male individuals, who comprise 54% to 73% of cases. , , , Involvement is unilateral in 80% to 90% of cases, and the right side is more commonly affected (60%). Although the contralateral ear appears normal in most cases, a study showed that the unaffected ear in patients with microtia is overall smaller than in the general population.
Etiology
The etiology of microtia is multifactorial, with both genetic and environmental factors being implicated. , , , Maternal diabetes type I, anemia, illness during the first trimester, advanced maternal and paternal age, first parity, higher maternal parity, and lower educational level have been deemed risk factors. Medications like isotretinoin, thalidomide, and mycophenolate mofetil have been implicated in multiple birth defects, including microtia. , , Alcohol consumption has also been shown to increase the risk, while folate consumption has been found to reduce it. Genetically, there is a significant contribution to microtia. Higher concordance has been seen monozygotic (38.5%) versus dizygotic twins (4.5%). Familial cases are common, with either an autosomal recessive or dominant mode of inheritance, with variable expression and incomplete penetrance. , Lastly, over 18 different microtia-associated syndromes with single-gene defects or chromosomal aberrations have been identified. Comprehensive lists of the most common microtia-associated disorders or syndromes can be found in several genetic reviews published to date. , , , , ,
Accompanying diagnoses and initial workup
Approximately 20% to 60% of patients with microtia have associated anomalies or an identifiable syndrome, warranting a thorough examination to detect any other dysmorphology. , Concurrent anomalies or a syndrome are more likely to be present in those with bilateral microtia. During initial workup of an anomalous ear (including microtia spectrum or lesser findings such as preauricular pits or tags), a thorough physical examination is paramount. Given the high incidence of concurrent ear and renal anomalies in several syndromes, a renal ultrasound is indicated when the ear malformation is accompanied by other dysmorphic features; a family history of deafness, auricular, and/or renal malformations; or a maternal history of gestational diabetes. In the absence of these additional findings, renal ultrasonography is not indicated.
Associated malformations most common with microtia include facial clefts (palate or others), cardiac defects, ophthalmologic defects, limb reduction defects, renal malformations, or holoprosencephaly. , Many of these anomalies are also associated with OAVS, a heterogeneous disorder involving the first and second branchial arch derivatives with a very wide clinical spectrum. Characteristic features of OAVS overlap with other named conditions, including CFM and Goldenhar syndrome. It is thought that these distinct entities are part of the OAV spectrum, with Goldenhar representing the most severe end. , Microtia is also a common feature of other syndromes, including Townes-Brocks syndrome, Treacher-Collins, Nager syndrome, Maier-Gorlin, and Kabuki syndrome. These conditions should be considered among the differential diagnosis when microtia is present.
In 55% to 90% of cases, microtia is accompanied by CAA, a malformed ear canal that can be stenotic or completely absent with varying grades of middle ear involvement. The degree of microtia and the severity of middle ear dysplasia are usually correlated, with more severe forms of microtia having more severe middle ear involvement. , , When evaluating a newborn with microtia, an audiologic baseline evaluation should be obtained within the first few months of life. , While 80% to 90% of patients have conductive hearing loss in the affected ear, 10% to 15% of patients can also present sensorineural hearing loss, warranting further workup. Most patients have normal hearing in the contralateral ear, which should allow for normal speech and language development. , However, efforts should be made to achieve binaural hearing during the critical period of brain development in early childhood, usually in the form a hearing aid. If there is any concern regarding the hearing status of the non-microtic ear (ie, a failed newborn hearing screen) or in bilateral microtia cases, diagnostic auditory brainstem response testing should be performed. The normal ear should be closely monitored throughout life, and prompt diagnosis and treatment of middle ear effusion or other issues should be undertaken to protect hearing.
Classifications
Several classification systems for microtia have been proposed overtime. The simplest classifications are based on the patterns of the dysmorphic ear. These are the least specific but most widely used. In 1926, Marx created the first classification for microtia dividing it into 3 grades, later modified by Rogers to include anotia , ( Table 1 ).
| Classification System | Categories Within System | Explanation |
|---|---|---|
| Marx/Rogers Classification | Grade I | A smaller auricle with all identifiable features of an ear |
| Grade II | An abnormal auricle with some recognizable normal structures | |
| Grade III | An abnormal auricle with no recognizable normal structures | |
| Grade IV | Anotia | |
| Nagata Classification | Lobule type | Cartilaginous remnant and vertical lobule without discernible canal concha, or tragus. |
| Concha type | Remnant ear has a concha and lobule, with or without a canal, tragus, or incisura intertragica. | |
| Small concha type | Remnant ear and lobule with only a small indentation representing the concha, although usually malpositioned (too anterior). | |
| Anotia | Absence of ear, or tissue present with no ear structure resemblance | |
| Atypical | Cases that do not fit the above classifications | |
| HEAR MAPS Classification | H earing | Bone conduction and hearing function. |
| E ar | Microtia Marx/Rogers grading | |
| A tresia | Jahrsdoerfer Scale based on temporal bone CT morphology and candidacy for external auditory canal reconstruction (EACR). | |
| R emnant earlobe | Grade 1: Normal Grade 2: Mildly reduced Grade 3: Moderately Reduced Grade 4: Severely reduced/absent | |
| M andible | Grade 1: Normal Grade 2: Mildly reduced Grade 3: Moderately reduced Grade 4: Severely reduced/absent | |
| A symmetry of soft tissue | Grade 1: Normal Grade 2: Mildly reduced Grade 3: Moderately reduced Grade 4: Severely reduced/absent | |
| P aresis facial nerve | House-Brackmann scale | |
| S yndromic? | Grade 1: No Grade 2: Yes |
Later, Nagata proposed a classification with descriptive terms based on the morphology of the auricular remnant in a way that directly influenced his approach. , Firmin also created a classification based on skin incision patterns rather than the ear morphology itself (see later discussion), which is used mostly by those who utilize her technique. More modern systems have attempted to include additional features besides ear morphology or surgical approach. Roberson and Reinisch devised the HEAR MAPS classification, using acronym that combines nor only the external ear, but also hearing, temporal computed tomographic (CT) scan results, facial morphology, and presence of a syndrome. While this classification is much more complex, it is comprehensive and allows teams to consider both the ear reconstructive and hearing aspects of the patient with microtia. Its application would also allow for better data collection for research.
Surgical techniques
Preoperative Assessment
In taking the medical history, medical problems or syndromes, any previous surgery on the head or chest area, and any developmental delays should be noted. Psychosocial implications of the deformity should be considered, including low self-esteem and bullying, which can be motivators for surgery. Hearing status should be clarified. The existence of a bone-anchored hearing aid (BAHA) should factor into surgical planning.
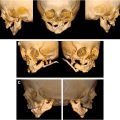
During physical examination, the type of microtia, skin quality around the ear, scars, low hairline, and any asymmetries of the face or chest should be noted. It is common to find undiagnosed hemifacial microsomia. One should look at the size and shape of the contralateral ear and palpate for any implanted hearing devices. A temporal bone CT scan may be desired by otolaryngology to assess internal ear anatomy for atresia reconstruction. If the patient is appropriate for ear reconstruction, the discussion of options follows.
An external prosthesis is an option, which allows for a very esthetically pleasing and symmetric ear. However, prosthesis needs to be surgically anchored, and maintenance is complicated; it can fall off during activity and does not change color like normal skin. Prosthetics are costly, often not covered by insurance, and usually need to be replaced every 2 to 3 years. While they are not ideal in pediatric patients, they are valuable in adults or in cases of severe scarring and failed reconstruction. , The second option is a porous polyethylene implant and temporoparietal fascia (TPF) flap. While it offers an off-the-shelf ear framework with a predictable appearance, complication rates can be high and difficult to manage. The third option is autologous reconstruction utilizing costal cartilage. This is the most widely utilized technique among ear reconstruction surgeons. This technique offers a durable option with the patient’s own tissue, and greater ability to salvage in case of exposure or infection at any point in life. However, it is technically demanding, has a steep learning curve, and usually requires at least 2 stages. The surgical plan discussion with the patient and parents often involves more than one consultation.
Autologous Reconstruction
History
Sir Harold Gillies was the first to use autologous costal cartilage for ear reconstruction in 1920. In 1950, following the foundations laid by Gillies, Tanzer reintroduced the use of rib cartilage for ear reconstruction with a 6 stage approach. He described cartilage as “the best material available for building lasting support.”
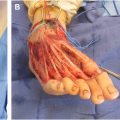
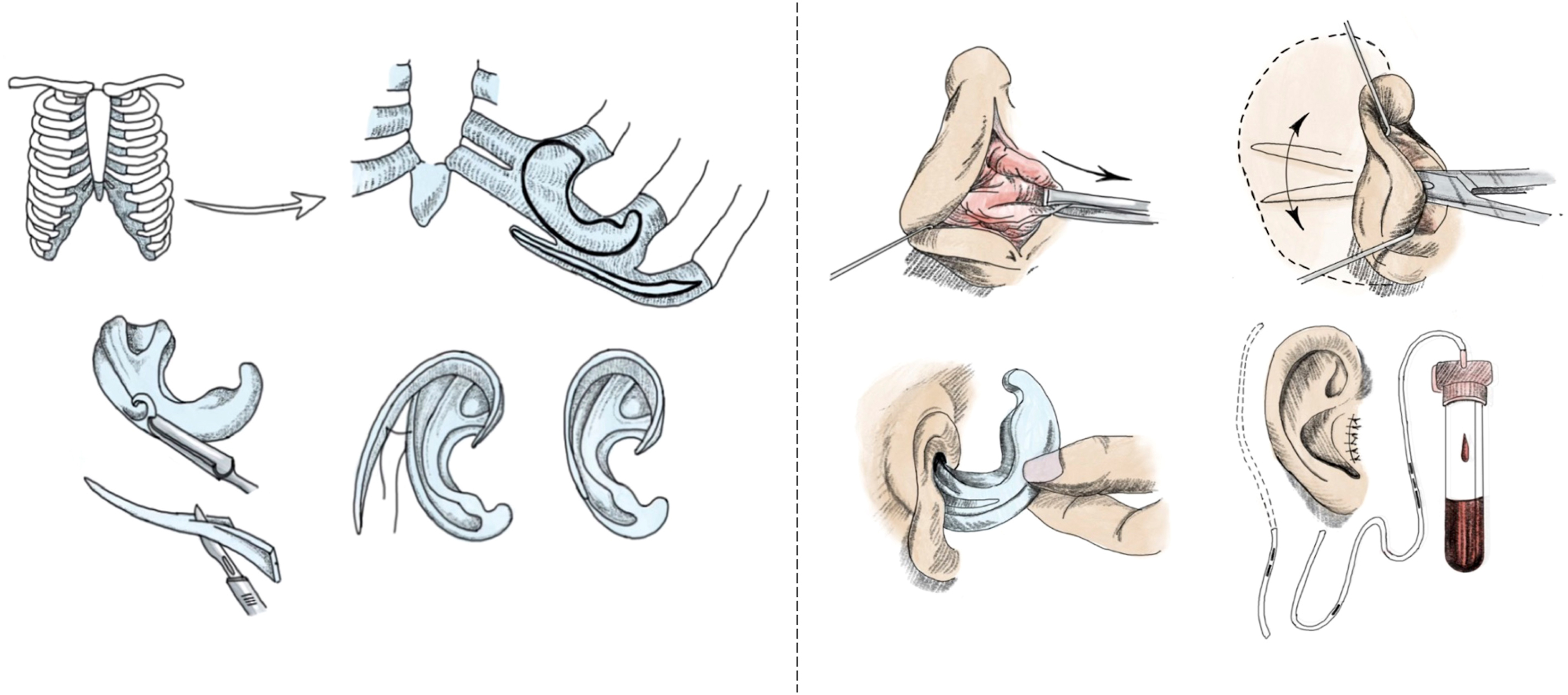
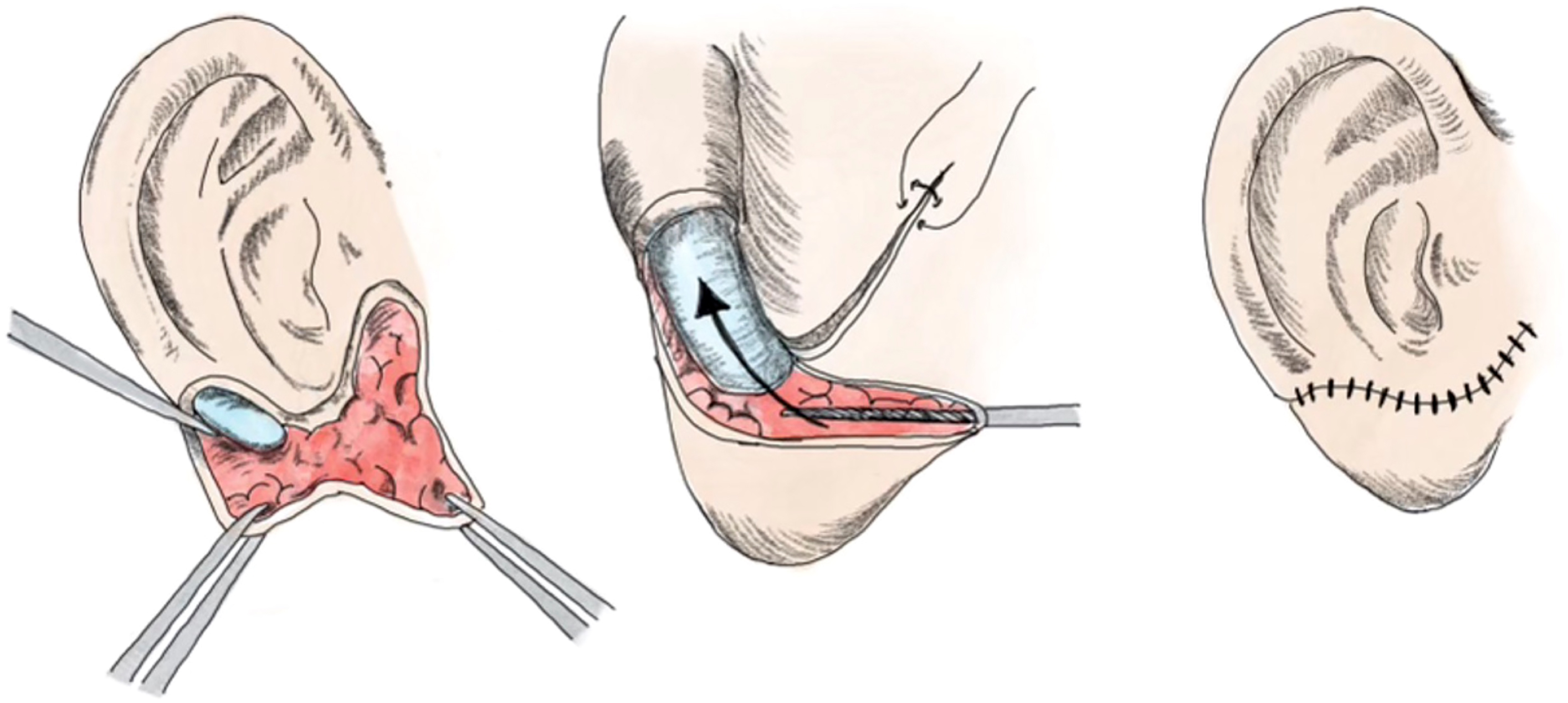
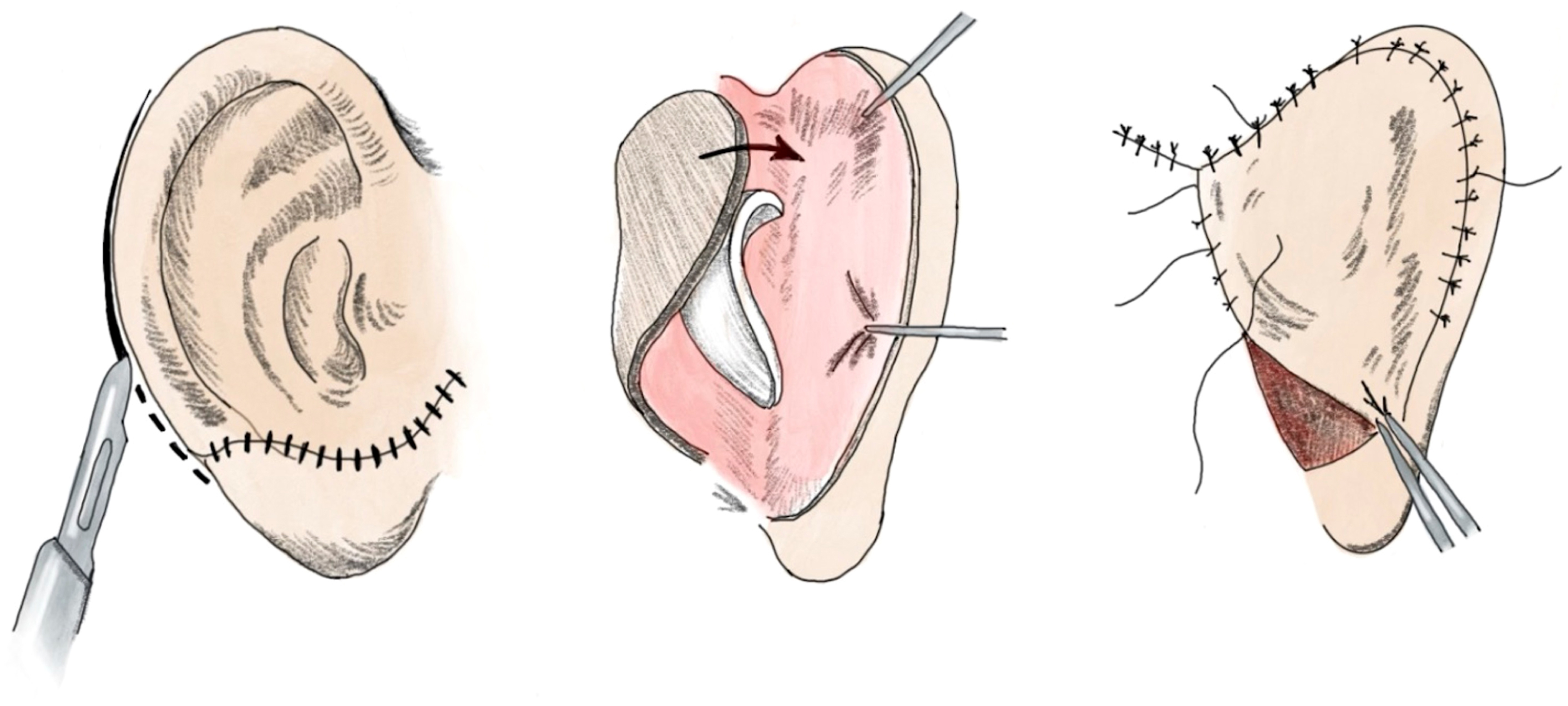
Brent refined Tanzer’s approach with the introduction of his own 4 stage technique in 1974. During the first stage, Brent’s technique includes harvesting part of the contralateral sixth, seventh, and eighth ribs; carving and assembling them into a framework and placing them under mastoid skin under suction ( Fig. 2 ). The second stage involves lobule transposition ( Fig. 3 ). The third is the elevation of the construct with the placement of a skin graft, and the fourth is creation of the tragus ( Fig. 4 ).
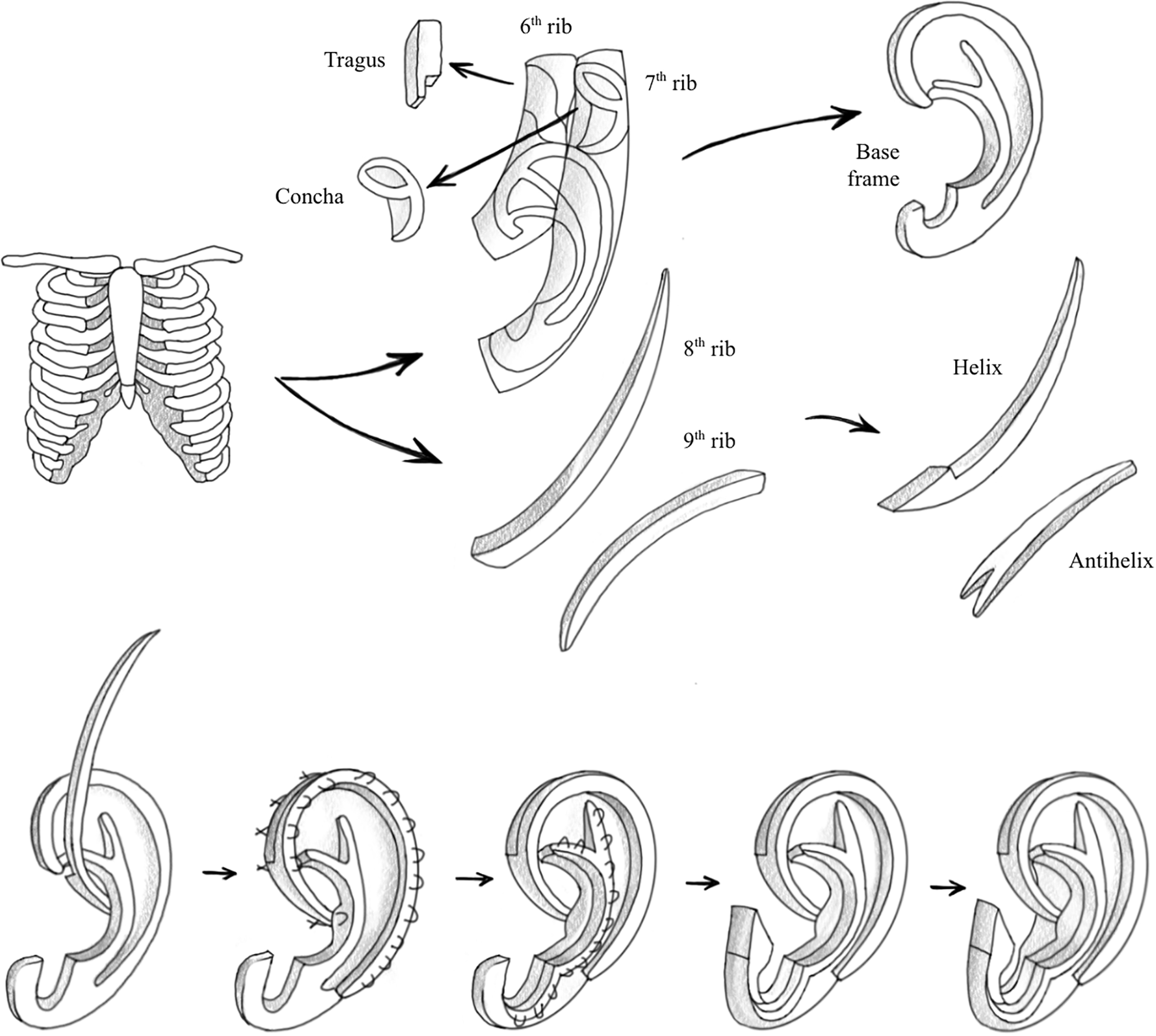
In 1993, Nagata introduced 2 stage approach that combined the first, second, and final stages of Brent’s technique, such that lobule transposition occurs in the same stage as framework fabrication and placement. , The second stage focuses on refining the contours and elevating the reconstructed ear to achieve a natural appearance. , His technique requires more cartilage and is best suited for older patients ( Fig. 5 ).
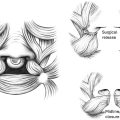
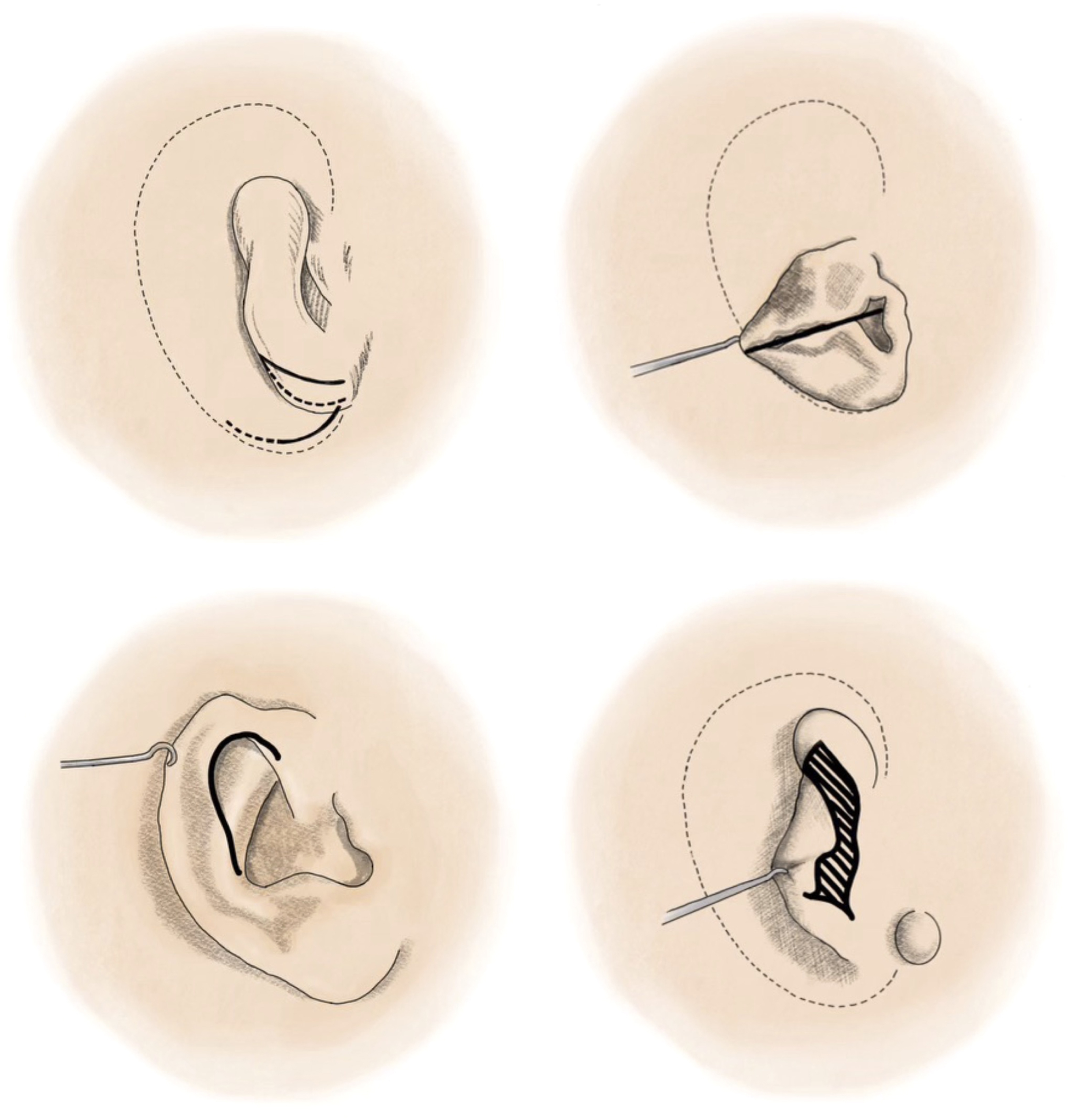
In 1998, Firmin proposed significant modifications to the Nagata technique to add greater versatility to the reconstruction depending on the type of microtia. Besides developing different frameworks, she also proposed different surgical approaches tailored for each type of deformity , ( Fig. 6 ). Firmin advocated for keeping the original tragus, antitragus, or both, rather than excising the native portions of the microtia remnant.
In 2000, Park introduced the expanded 2 flap method for microtia reconstruction, a 3 stage process that begins with the insertion of a subfascial tissue expander. , In the second stage, contralateral rib cartilage is harvested to create the ear framework, which is then placed with a skin flap covering the anterior surface, and a fascial flap and skin graft covering the posterior portion. The third stage involves small touch-ups to shape the fine details of the ear. ,
Autologous technique
The senior author prefers Firmin’s technique with her own adaptations, which will be discussed in detail.
While optimal timing for surgery is controversial, most surgeons prefer to do the surgery around the ages of 7 to 10 years, , , , because the human ear reaches maturity around the age of 7 years. It is also necessary to have sufficiently mature cartilage to create a construct that is stable and properly sized. Nagata cites a cutoff of the age of 10 years and chest circumference of 60 cm, while others have the age of 8 years or 55 cm chest circumference. , Brent described performing the surgery as young as the age of 6 years with an ideal of 7, though his technique requires less cartilage. Regardless, the child should be mature enough to participate in the decision and postoperative care. , ,
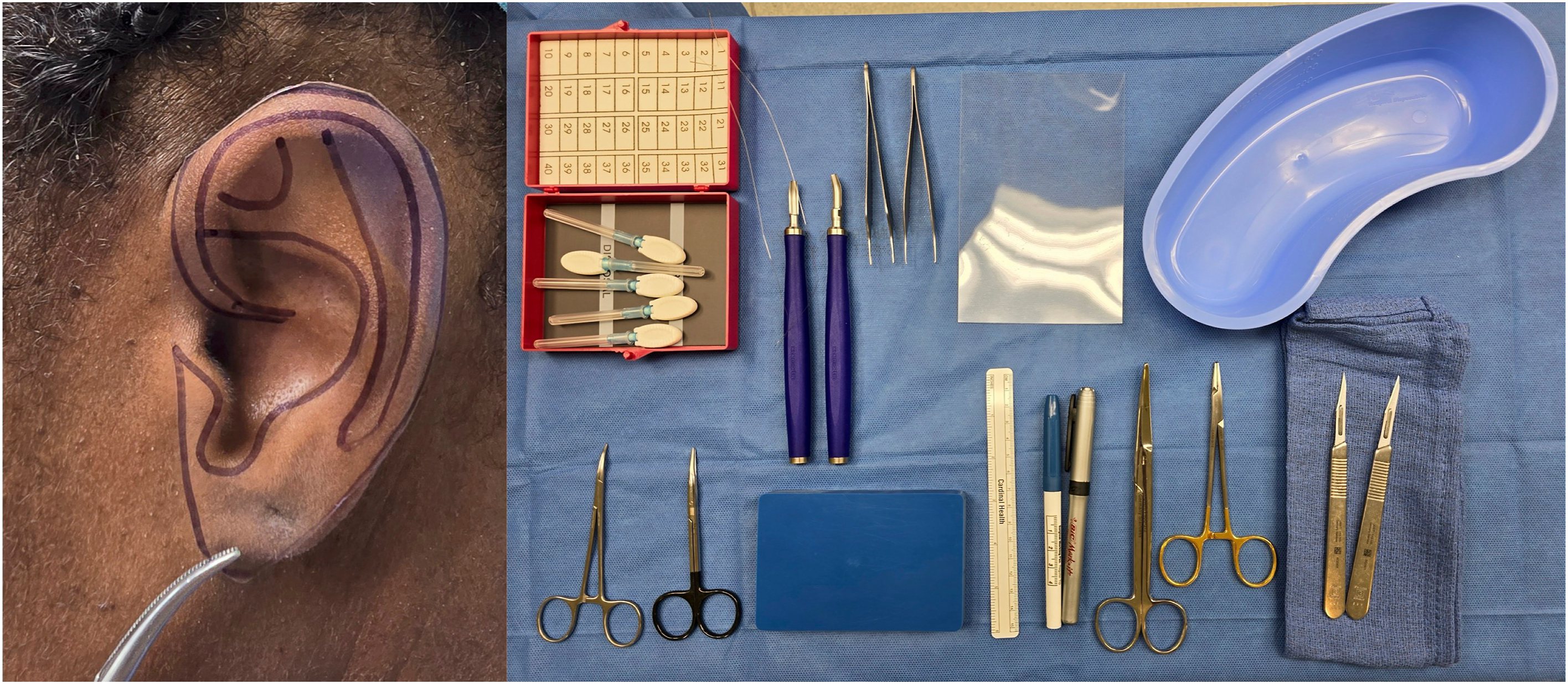
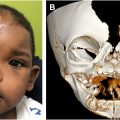
First stage
The initial surgery consists of planning the ideal placement for the ear construct, dissecting the skin envelope, harvesting rib cartilage, carving and building the construct, and securing it in the skin pocket. Like Brent, Firmin created a template utilizing radiograph film to trace the normal ear, , but one can utilize any other sterilizable and trimmable material. The normal ear is traced in the plastic film ( Fig. 7 ), which is placed between 2 transparent film dressings on the sterile field. A second template can be made and cut into the components that need to be carved. In bilateral microtia, the size and shape may be modeled after parent’s ears. Three dimensional (3D) printed models based off CT scans can also be used intraoperatively as templates.