Ulnar collateral ligament injuries are prevalent among baseball players with an increasing incidence, especially in younger athletes. Diagnosis relies on history, examination, and MRI, with dynamic imaging playing an expanding role. Nonoperative management prioritizes cessation of throwing, correcting kinetic chain deficiencies, and gradual rehabilitation. The decision between reconstruction and repair is based on tear characteristics, tissue quality, patient goals, position, and timing of injury. Primary surgery generally results in high return-to-play rates. Revision surgery has less favorable outcomes with more complications. Emerging trends include the use of hybrid reconstruction techniques for challenging cases and platelet-rich plasma to augment nonoperative care.
Key points
- •
The incidence of ulnar collateral ligament (UCL) injuries is continuing to rise, especially in younger athletes.
- •
MRI scans provide definitive diagnosis with dynamic imaging providing increasing utility.
- •
Kinetic chain deficiencies should be addressed as part of comprehensive rehabilitation for both nonoperative and operative treatment.
- •
UCL reconstruction remains the gold standard for surgical treatment, however, UCL repair augmented with an internal brace has become increasingly popular because of promising outcomes in appropriately selected patients.
Introduction
Ulnar collateral ligament (UCL) injuries of the elbow are common in overhead athletes, particularly baseball players. The UCL consists of 3 bundles including the anterior, posterior, and transverse bundle with the anterior bundle serving as the primary stabilizer to valgus stress. A significant valgus force is generated across the elbow joint during the throwing motion and repetitive tensile stress on the UCL leads to both acute and chronic injuries. Ultimately, the destabilizing effect of a UCL injury results in medial elbow pain and valgus instability during the late cocking and early acceleration phase of throwing, leading to decreased throwing performance.
Before the first UCL reconstruction performed by Dr Frank Jobe in 1974, these injuries were considered career-ending for overhead athletes. Partial UCL injuries and proximal tears can be successfully treated nonoperatively. However, complete UCL tears, especially in competitive overhead athletes, are generally indicated for operative treatment. Reconstruction techniques have evolved over the years to include different approaches to managing the flexor pronator (FP), ulnar nerve, graft type, tunnel configuration, and graft fixation. UCL repair has seen resurgence with the addition of internal brace augmentation demonstrating good outcomes in appropriately selected patients. With improved surgical techniques and more standardized rehabilitation protocols, the outcomes of surgical treatment have improved considerably with return-to-play (RTP) rates around 80% to 95%.
Recently, there has been an increasing trend in UCL reconstructions performed, particularly at a younger age. As a result, there has been a rise in the number of revision surgeries performed. This article aims to provide a comprehensive review of UCL injuries emphasizing nonoperative and operative management in the primary and revision setting.
Epidemiology
Elbow injuries are common in overhead athletes, particularly baseball players, because of the large and repetitive torque seen across the elbow joint. Elbow injuries in youth make up about 18% of all baseball-related injuries and more than 25% of pitchers will experience elbow pain during a season, further highlighting the high prevalence. , In a study of baseball players entering the Major League Baseball (MLB) draft, elbow injuries and UCL reconstructions were the most common.
The annual incidence of patients undergoing UCL reconstruction is 4 per 100,000 patients. The number of surgeries has increased over the last several years with a disproportionate number being performed in younger patients. More than 50% of all UCL reconstructions are performed in athletes between the ages of 15 and 19 with an overall annual incidence of 22 per 100,000 patients. In collegiate baseball players, 15% of elbow injuries require season-ending surgery with pitchers most frequently affected. At the professional level, the number of UCL reconstructions performed also continues to rise. More than 10% of all professional baseball players and 25% of MLB pitchers have undergone a UCL reconstruction.
Multiple risk factors have been linked to UCL injuries. Higher velocity and pitch counts are well-known predictors of UCL injuries. Poor pitching mechanics and kinetic chain deficiencies have also been implicated. Side-to-side differences including a decrease in the total arc of motion and internal rotation, as well as an increase in humeral retrotorsion of the dominant arm have been found to increase the risk of UCL injuries.
Anatomy and biomechanics
The overhead throwing motion places an enormous amount of torque on the medial elbow, particularly during the late cocking and early acceleration phases. Angular velocities through the elbow have been estimated to reach nearly 5,000° per second with a peak elbow torque of 64 Newton-metre. , Greater than 50% of the torque is transmitted through the UCL resulting in forces greater than 32 Newton-metre. The ultimate load to failure of a native UCL is about 34 Newton-metre, indicating that the forces seen during throwing can approach the forces required to result in a UCL tear.
Static and dynamic stabilizers of the elbow play a critical role in resisting valgus stress. Static stabilizers include the UCL, anterior capsule, and both the radiocapitellar and ulnohumeral articulations. The UCL is comprised of the anterior, posterior, and transverse bundles. ( Fig. 1 ) The anterior bundle is the primary restraint against valgus stress, while the posterior and transverse bundles provide minimal contributions. Callaway and colleagues demonstrated that the anterior bundle is functionally divided into the anterior and posterior bands, which tighten reciprocally with the anterior bundle tauter in extension and the posterior bundle tauter in flexion. They also showed progressively worsening valgus instability with increasing elbow flexion starting at 30° up to 90° after sequential cutting the ligament. The anterior bundle originates from the anteroinferior surface of the medial epicondyle. The UCL has a broad and elongated insertion on the ulnar ridge at the sublime tubercle. The osseous stabilizers provide constraint mainly at end ranges of motion including less than 20° and greater than 120° of elbow flexion.
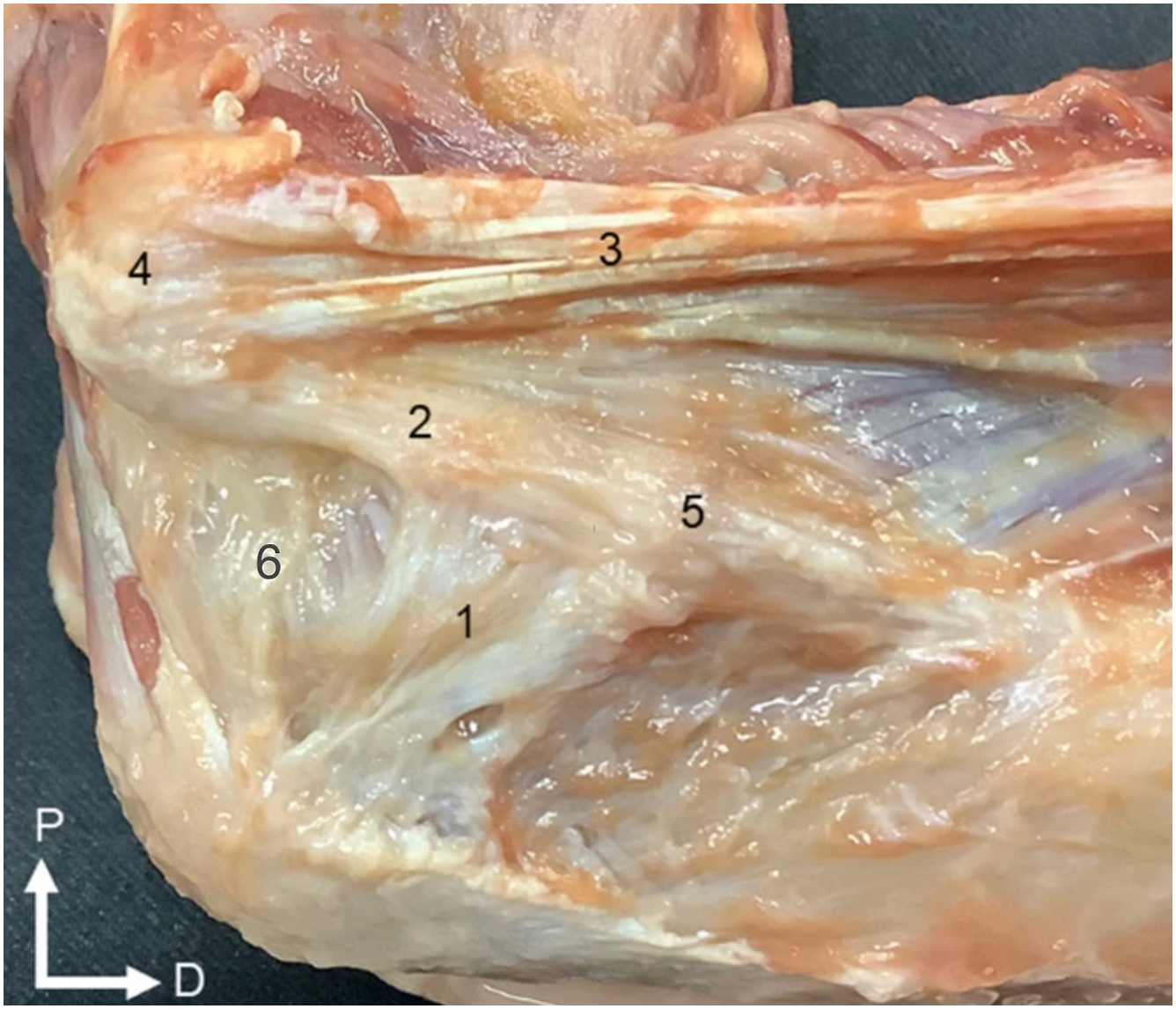
Dynamic stabilizers of the medial elbow primarily consist of the flexor carpi ulnaris (FCU), flexor digitorum superficialis (FDS), and pronator teres (PT), which comprise of the FP tendinous complex. The FCU is the primary dynamic stabilizer, the FDS is a secondary stabilizer, and the PT contributes the least. The FCU originates proximally at the common flexor insertion and distally along the ulnar ridge, while the FDS overlaps with the UCL insertion, providing significant restraint against valgus stress. Recent literature underscores the pivotal role of dynamic stabilizers, notably the FCU and FDS, in countering valgus stress. Lin and colleagues demonstrated that the FCU and FDS provided the most stability between 45 and 90°. Furthermore, electromyography studies have shown increased activation of the FCU during the throwing motion. Anatomic studies have demonstrated the intimate relationship between the tendinous complex, joint capsule, and the UCL, essentially forming a confluence of structures over the medial elbow, underscoring the clinical importance of strengthening these muscles during rehabilitation. ( Fig. 2 )
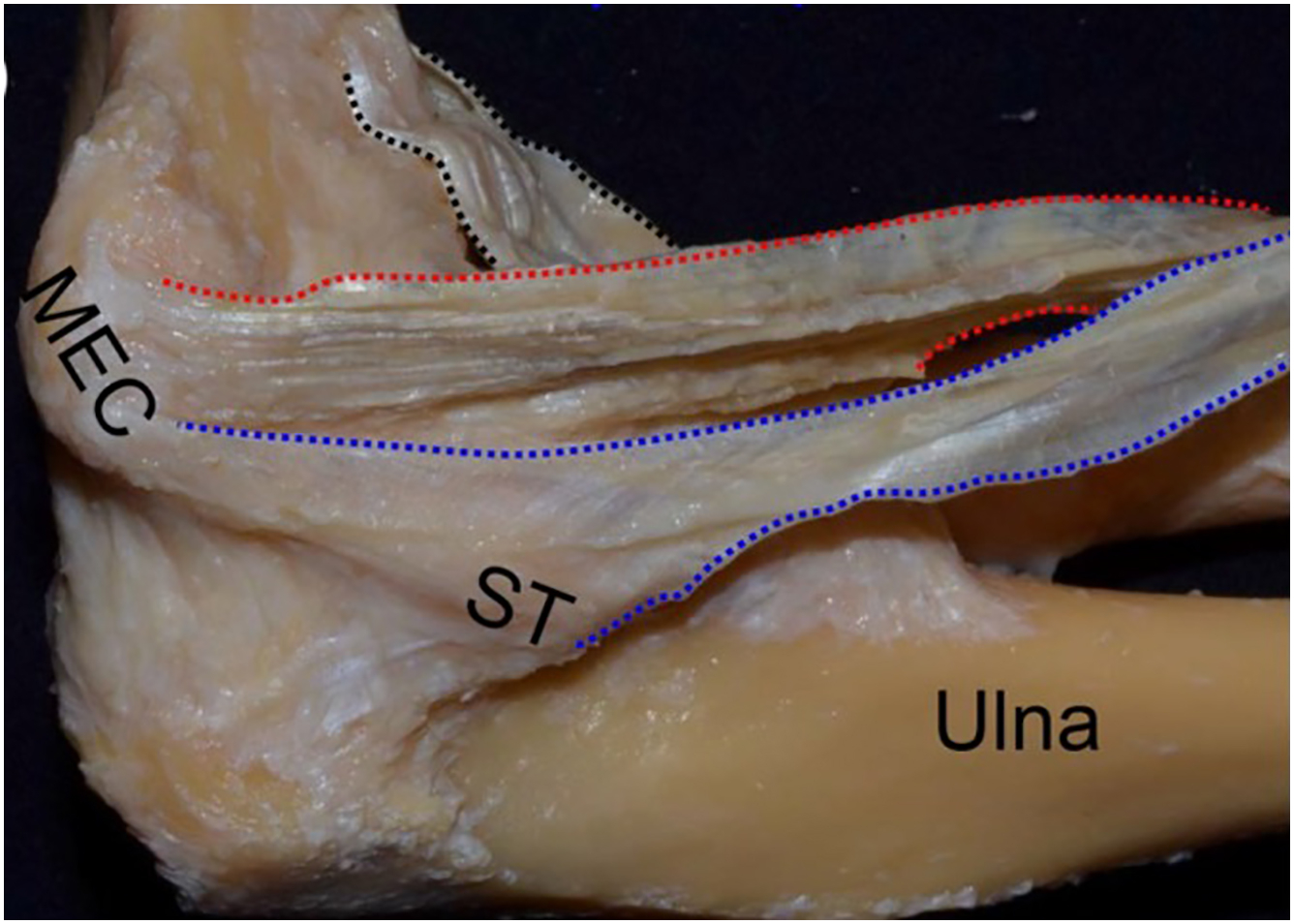
The vascular anatomy of the UCL also has clinical relevance. The proximal UCL is well-vascularized compared with the hypovascular distal portion. The proximal half of the UCL has a rich vascular plexus with contributions from the medial epicondyle and the FP muscle that significantly diminishes distal to the midpoint of the ligament. The regional differences in vascularity may explain the improved healing potential and success with nonoperative treatment with proximal tears. , Overall, a thorough understanding of the detailed anatomy, biomechanics, and vascularity of the UCL and its relationship with the other static and dynamic stabilizers is crucial in managing the overhead athlete.
Evaluation and diagnostic imaging
History and Physical Examination
The evaluation of any overhead-throwing athlete with medial elbow pain should start with a detailed history and physical examination. The chronicity of the symptoms, location, timing of pain during the throwing cycle, and the presence of ulnar sided and nerve symptoms should be elicited. Athletes should also be asked about their level of competition, goals, maximum velocity, and recent changes in velocity, control, workload, or mechanics. Prior shoulder and elbow injuries and treatments should be reviewed.
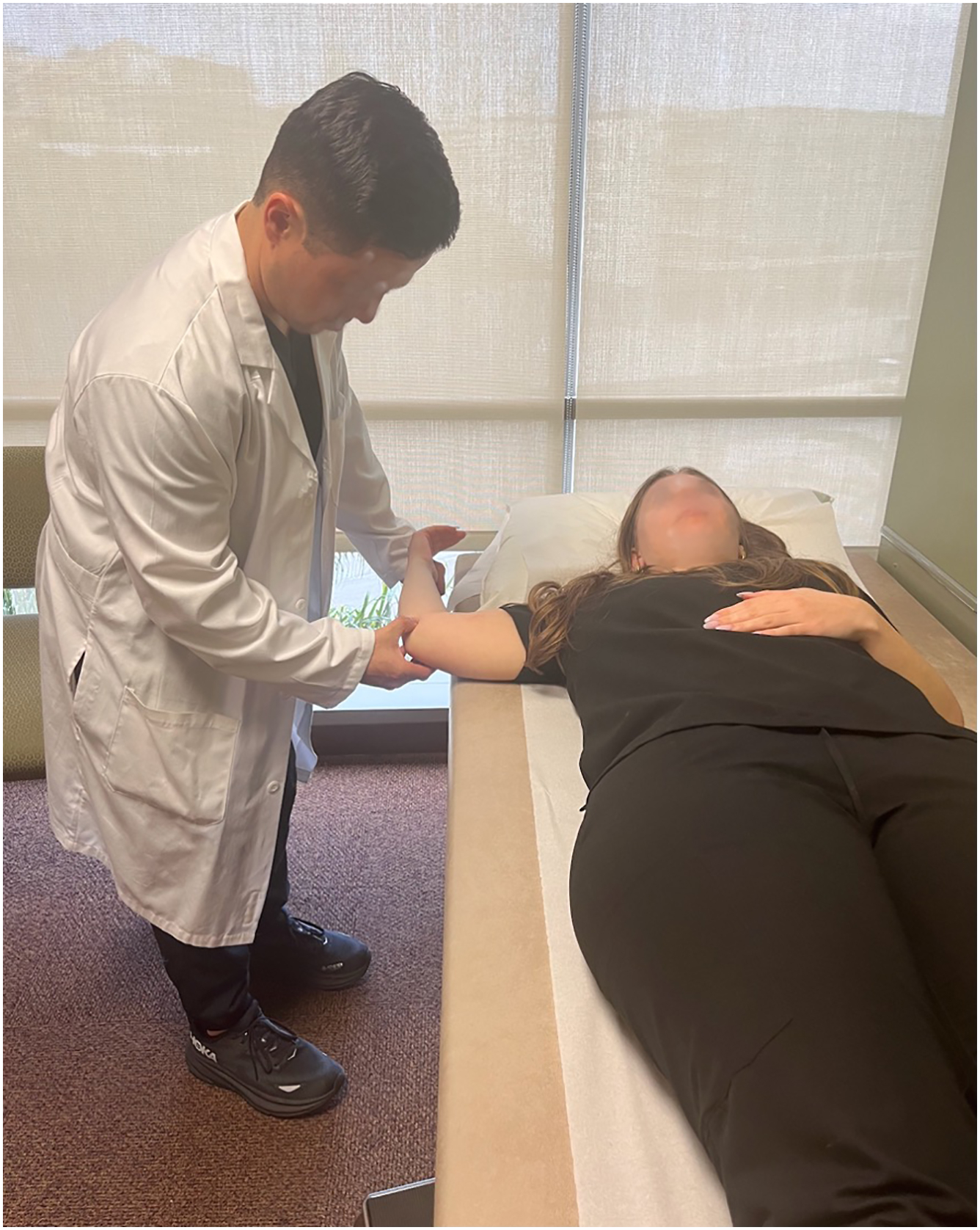
On examination, key landmarks to palpate include the medial epicondyle, UCL, sublime tubercle, FP, ulnar nerve, and olecranon. Both passive and active range of motion of the elbow should be evaluated, specifically for full extension and the presence of posterior pain with terminal extension as this is associated with posteromedial impingement. The shoulder should be assessed for range of motion (ROM) deficits compared with the contralateral shoulder and for scapular dyskinesis. A neurovascular assessment should include sensory and motor testing of the ulnar nerve, Tinel’s sign, ulnar nerve compression test, and stability of the ulnar nerve with elbow ROM. The presence of a palmaris longus should also be noted. Special maneuvers to evaluate the integrity of the UCL include the milking maneuver and moving valgus stress test. The milking maneuver test is performed by placing the shoulder at 90° of abduction, the elbow at 90° with the forearm in full supination followed by pulling on the thumb to create a valgus force. The moving valgus stress test is performed in the same position as the milking maneuver while flexing and extending the elbow ( Fig. 2 ). Pain or apprehension between 70 and 120° of flexion is considered a positive test. Flexion and extension outside of this range will engage the coronoid and olecranon, respectively, and provide stability, resulting in a reduction of symptoms. Prior studies have demonstrated a sensitivity of 100% and specificity of 75% with the moving valgus stress test. Additional tests to evaluate for a FP strain include resisted forearm pronation with the elbow extended and temple pressure with the thumb, index, and middle finger.
Radiographs
Anterior posterior, lateral, and oblique radiographs of the elbow can evaluate calcifications within the ligament, bony avulsions, enthesophytes extending from the ulna, capitellar osteochondral defects, loose bodies, and if the physes remain open. Contralateral views are useful in the skeletally immature athlete. An axial view can help identify posteromedial osteophytes. Valgus stress radiographs are performed with the elbow in 30 of flexion and the forearm supinated. Complete UCL tears gap on average 0.6 mm more than the contralateral elbow.
Advanced Imaging
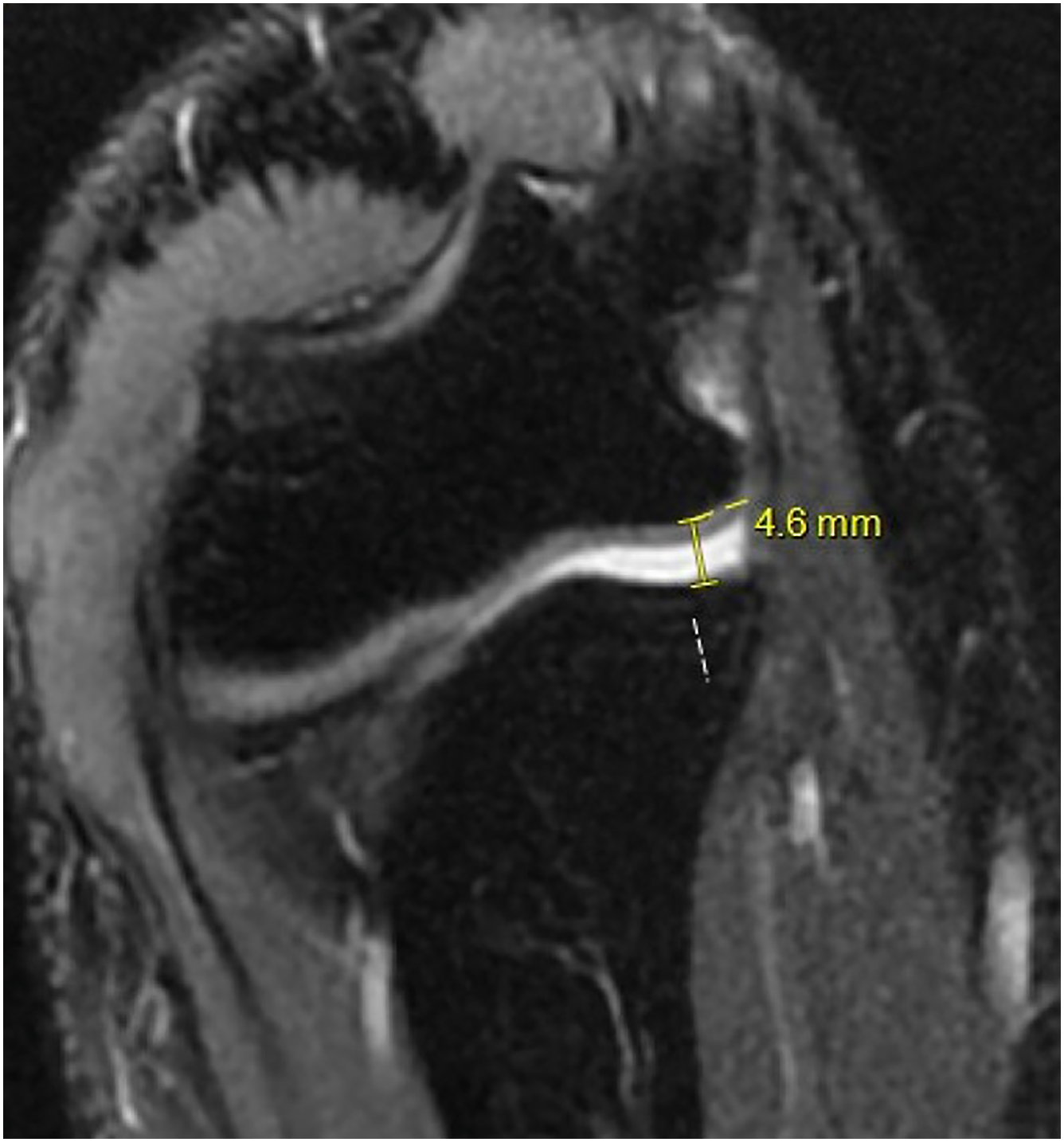
Conventional 3 Tesla (3T) MRI and MR Arthrograms (MRA) are the study of choice for diagnosing UCL injuries. MRA has slightly better diagnostic accuracy than conventional 3T MRI. MRA is 100% sensitive and specific and 3T MRI is 100% sensitive and 89% specific for detecting full-thickness UCL tears. The improved diagnostic capabilities should be weighed against the invasiveness, added patient discomfort, and increased cost. A flexed elbow valgus external rotation (FEVER) MRI view was recently described as a dynamic assessment of the competency of the UCL that may improve diagnostic accuracy. A FEVER view is obtained using a 30° coronal oblique MRI view to be parallel with the long axis of the UCL while placing the elbow at 90° with valgus stress applied and subsequent measurement of the ulnohumeral gapping from the subchondral edges ( Fig. 3 ); computed tomography scan is reserved for surgical planning, particularly revision cases to assess the amount of tunnel widening or bone loss, as well as for large posteromedial osteophytes.
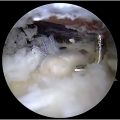
Ultrasound
The utility of ultrasound as a diagnostic tool has emerged as a valuable resource. Ultrasound allows for point-of-care assessment of the UCL, which can be coupled with a dynamic stress examination to further assess the competency of the ligament. Dynamic ultrasound has a sensitivity of 96%, specificity of 81%, and accuracy of 87% in diagnosing a UCL tear when there is joint gapping greater than 1 mm compared with the contralateral side. Ultrasound has the capability of detecting partial- and full-thickness tears, edema, quality of the ligament, medial joint space gapping with stress test, and concomitant FP injuries. While ultrasound is operator-dependent, it can be a quick, safe, and non-invasive adjunct when evaluating for UCL injuries.
Nonoperative management
Nonoperative treatment of UCL injuries includes careful consideration of various factors, which can be summarized using “the Five P’s” approach: (1) personality of the tear (severity, location, and ossifications); (2) player (level of play); (3) position (pitcher vs position player); (4) potential (realistic upside of the player); and (5) period (time frame relative to the season). Seasonal timing of an injury plays a particularly important role that is often under-recognized. For example, if a player suffers a UCL injury at the beginning of the season, fails 3 months of nonoperative treatment, and ultimately requires surgery, they could miss two full seasons instead of potentially just one had they undergone early surgical intervention. In contrast, if the same injury occurs near that later part of the season and they fail nonoperative treatment, they will likely only miss the next with the potential of avoiding surgery.
Some relative indications for nonoperative treatment include partial-thickness tears, proximal tears, low career aspirations, unwillingness to participate in prolonged rehabilitation, and no prior formal trial of conservative treatment. The goals of nonoperative treatment initially are to decrease pain, inflammation, and restore motion. This is typically achieved through a combination of throwing cessation, avoidance of valgus stress, ice, non-steroidal anti-inflammatory drugs, and physical therapy. A structured rehabilitation program should be implemented early on and focus on strengthening and addressing kinetic chain deficiencies. Specifically, therapy should focus on optimizing elbow, shoulder, and hip ROM, scapular mechanics, pitching mechanics, core and lower body strength and flexibility, and FP strength. FP strengthening can start once the patient is pain-free. Kinetic chain deficiencies including shoulder ROM deficits, scapular dyskinesis, contralateral trunk tilt, poor lumbopelvic control, and imbalance in lower body strength and balance have been linked to increased elbow torque and UCL injuries in overhead athletes and should be addressed before implementing a throwing program. , , Finally, a progressive throwing program over 2 to 3 months is started once the patient no longer has pain and kinetic chain deficiencies have been addressed.
In recent years, there has been more interest in the use of biologic adjuncts. Corticosteroid injections are generally avoided as they can further attenuate the ligament. In contrast, platelet-rich plasma (PRP) has drawn interest recently given its ability to stimulate angiogenesis and cell proliferation. Some studies have shown high RTP rates approaching 88% to 96% with leukocyte rich-PRP, however, most studies are limited by several confounding factors and lack of control groups. , In contrast, Chauhan and colleagues retrospectively evaluated a matched cohort from a database of professional baseball players and found a 46% RTP with PRP versus 57% RTP with no PRP, as well as a longer delay to throwing following PRP. However, there was significant variability in the preparation of the PRP, injection protocol, and rehabilitation. While PRP has shown promise, more research is needed to fully understand the ideal preparation and true efficacy when managing UCL injuries.
Outcomes
The outcomes with nonoperative treatment are variable in large part because of the influence of factors such as injury pattern, patient characteristics, sport, position, and variability in rehabilitation protocols. Rettig and colleagues first reported on the results of nonoperative treatment in throwing athletes and found a 42% RTP rate at or more than their pre-injury level after 24.5 weeks. Furushima and colleagues similarly showed an overall 49% RTP with 3 months of therapy, while partial tears had an 82% RTP rate. They also noted predictors of failure included complete tears, longer duration of symptoms, ulnar nerve symptoms, and the presence of an ossicle. Frangiamore and colleagues also found a 12.4 times higher likelihood of failing nonoperative treatment with a distal tear. They also found that 82% of failures occurred with distal tears and 81% of those treated successfully with nonoperative care had proximal tears. At the professional level, Walker and colleagues found an 85% RTP with no players requiring UCL reconstruction in a small cohort. Additional studies are needed to assess the effectiveness of nonoperative treatment as the authors’ understanding of the rehabilitation principles have improved but also the demands on the modern days pitcher have changed with a steep rise in velocity and spin rates coupled with decreased time in between pitches. Ultimately, the keys to favorable outcomes with nonoperative treatment are patient selection, throwing cessation, correction of kinetic chain deficiencies, and reconditioning the shoulder and elbow with a structured throwing program.
Primary surgical management
Surgical techniques have evolved significantly since the first UCL reconstruction was first performed by Frank Jobe in 1974. His initial technique described detaching the FP, creating a V-shaped ulnar bone tunnel and Y-shaped humeral bone tunnel at the medial epicondyle, creating a figure-of-8 construct by tying the graft over itself and performing a submuscular ulnar nerve transposition. Subsequent modifications aimed at minimizing surgical morbidity by utilizing a muscle-splitting approach, adjusting the proximal humeral tunnels to exit more anteriorly, and selectively performing ulnar nerve transposition. , The docking technique was later developed in an attempt to decrease bone removal from the medial epicondyle, improve graft tensioning, and enhance graft fixation by tying suture over a humeral bone bridge, ultimately creating a triangular graft construct. Linear constructs, such as the dual interference screw technique, David Altcheck and Neal ElAttrache for Tommy John technique or DANE TJ, and anatomic techniques, were introduced to mitigate the risk of bone bridge fracture as seen with bone tunnels, avoid posteriorly directed drilling near the ulnar nerve, and to better recreate the central fibers of the anterior UCL bundle.
The first reports of direct suture repair for UCL injuries were by Norwood and colleagues in 1981 and later by Conway and colleagues in 1992. , Felix Savoie was the first to report on UCL repair using suture anchors in 2006 and 2008. , It was not until 2016 when Dugas and colleagues introduced the modern-day UCL repair augmented with an internal brace.
Current surgical options for primary UCL tears include a standard UCL reconstruction, most commonly with a docking or modified-Jobe technique, augmented primary repair, and a hybrid reconstruction, which includes a standard reconstruction augmented with an internal brace or suture.
Reconstruction
General indications for UCL reconstruction over repair include poor ligament quality with attritional changes, soft-tissue loss, pathology at both the humeral and ulnar attachments, and complete mid-substance tears. Some also advocate for UCL reconstruction over repair in high-demand throwing athletes, however, there is no clear consensus. It is the senior authors’ preference to perform a UCL reconstruction with a docking technique using an ipsilateral palmaris longus graft as described as follows.
Surgical Approach
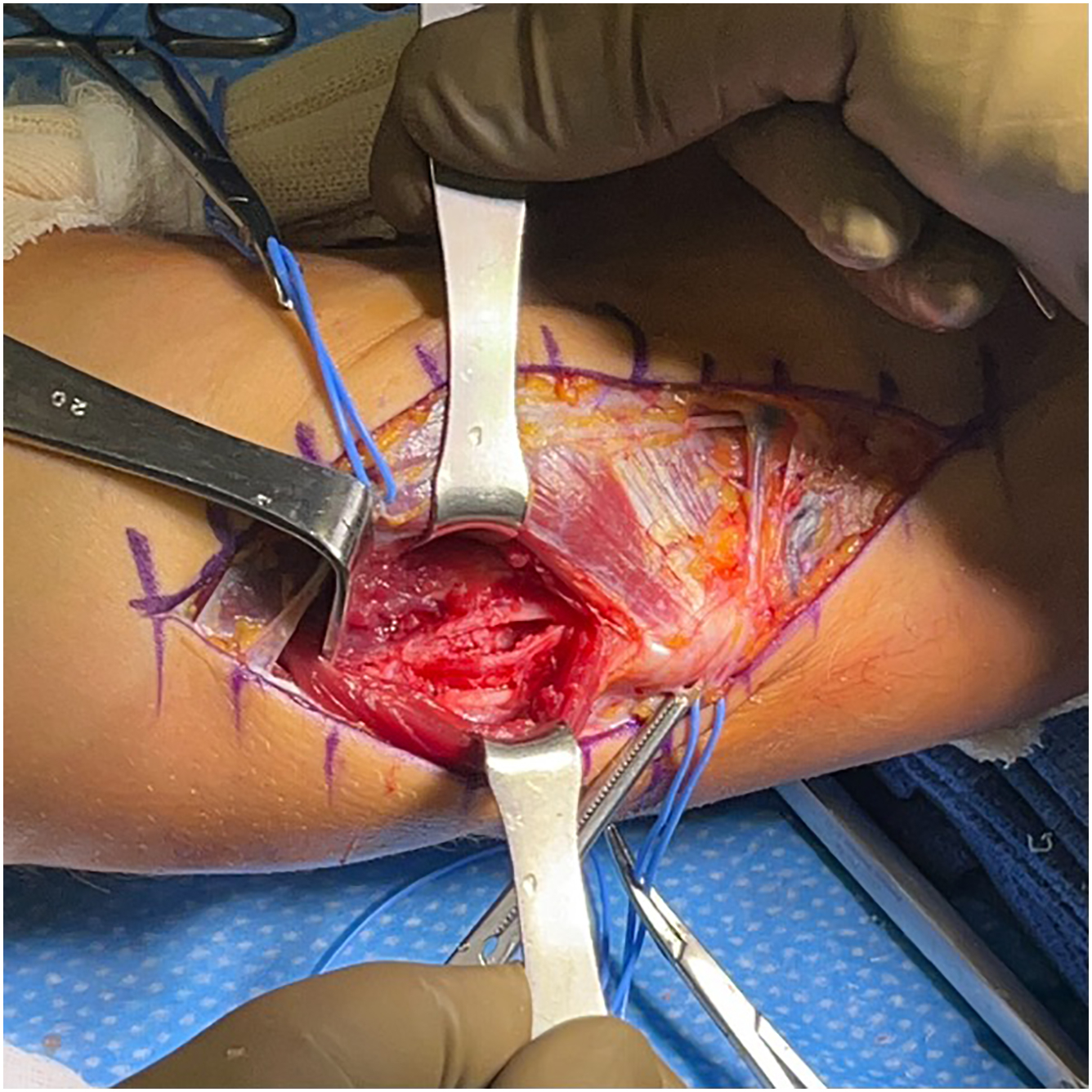
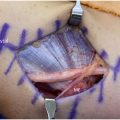
The patient is placed supine with the arm on a hand table. After administration of general anesthesia, a nonsterile tourniquet is applied. A curvilinear skin incision is made centered over the medial epicondyle. Subcutaneous dissection is carried down to the fascia taking care to preserve the medial antebrachial cutaneous nerve. A periosteal elevator is used to expose the anterosuperior surface of the supracondylar ridge to prepare for the exiting humeral bone tunnels. A muscle-splitting approach is performed by making a longitudinal incision over the tendinous raphe of the FCU. The underlying ligament is exposed with a periosteal elevator and a longitudinal split is made in the anterior bundle of the UCL ( Fig. 4 ).
Preparation of Bone Tunnels
Converging ulnar bone tunnels are made at the sublime tubercle centered over the ulnar ridge using a 3.5 mm drill and ensuring there is at least a 5 mm bone bridge ( Fig. 5 ). The ulnar tunnels are connected with a curved curette and a passing suture is placed for later graft passage. A 4.5 mm docking tunnel is made at the footprint of the UCL ensuring to lateralize the footprint and on the anteroinferior surface of the medial epicondyle while aiming proximally toward the insertion of the intermuscular septum ( Fig. 6 ). Precise humeral tunnel placement is critical, particularly for achieving isometry as the several of reconstructed grafts fail proximally. Lateralization of the humeral tunnel also allows for better bone quality and blood supply, decreases the risk of medial epicondylar fracture, and preserves the native ligament proximally for closure over top of the graft. Two converging 2 mm drill holes are then directed toward the docking tunnel, 1 just anterior to the medial intermuscular septum, and a second one on the anterosuperior surface of the medial epicondyle. Two passing sutures are placed through the docking tunnel and out both superior drill holes.