Magnet characteristics
Experimental model
Compound applied
Passive permeation
μg/cm2/h
Magnetophoretic permeation
μg/cm2/h
Enhancement ratio
References
Permanent magnets (0.01–0.05 mT)
Excised abdominal skin of albino rats
Benzoic acid (saturated solution)
≈1.8–2.6 (with increasing field strength)
Murthy (1999)
Permanent magnet (0.01 mT)
Excised abdominal skin of albino rats
Salbutamol sulphate
Terbutaline sulphate (saturated solutions)
53.12 ± 1.2
61.33 ± 2.8
130.31 ± 4.4
141.10 ± 3.0
2.5
2.3
Murthy and Hiremath (1999)
Static neodymium magnets (30, 150, 300 mT)
Excised porcine epidermis
10 mg/mL lidocaine hydrochloride
0.18 ± 0.06
0.53 ± 0.09 at 30mT
1.01 ± 0.17 at 150 mT
1.61 ± 0.12 at 300mT
2.9
5.6
8.9
Murthy et al. (2010)
Static neodymium magnets (450 mT)
Excised abdominal skin of Sprague–Dawley rats
20 mg/mL lidocaine hydrochloride in aqueous HPMC gel
0.94 ± 0.13
3.07 ± 0.43
≈3
Sammeta et al. (2011)
Static neodymium magnets (450 mT)
Sprague–Dawley rats with cutaneous microdialysis
20 mg/mL lidocaine hydrochloride in aqueous HPMC gel
10.57 ± 2.05 μg.h/mL (AUC0–6 h)
23.02 ± 2.96 μg.h/mL (AUC0–6 h)
≈2
Murthy et al. (2010)
PEMF – Dermaportation
(0.25 mT average, 5 mT peak)
Excised human epidermis
20 mg/mL 5-aminolevulinic acid in PBS solution
0.12 μg/cm2/h (estimated 0–2 h)
50.79 μg/cm2/h (estimated 0–2 h)
≈400
Namjoshi et al. (2007)
PEMF – Dermaportation
(0.25 mT average, 5 mT peak)
Excised human epidermis
25 mg/mL lidocaine hydrochloride and 25 mg/mL prilocaine hydrochloride in PBS solution
LH: 4.5 μg/cm2/h
PH: 4.3 μg/cm2/h
LH: 8.9 μg/cm2/h
PH: 8.5 μg/cm2/h
≈2
Cacetta et al. (2007)
PEMF – Dermaportation
(0.25 mT average, 5 mT peak)
Excised human epidermis
11.6 mg/mL diclofenac diethylammonium salt (Voltaren Emulgel)
1.58 μg/cm2/h
2.97 μg/cm2/h
≈2
Benson et al. (2007)
PEMF – Dermaportation (0.25 mT average, 5 mT peak)
Excised human epidermis
Dipeptide: 1 mg/mL Ala-Trp in PBS solution
0.78 μg/cm2/h (0.33–2 h)
19.43 μg/cm2/h (0.33–2 h)
≈25
Namjoshi et al. (2008)
PEMF – Dermaportation (0.25 mT average, 5 mT peak)
Excised human epidermis
Naltrexone hydrochloride
131.7 μg.h/cm2 (AUC0–4h)
859.5 μg.h/cm2 (AUC0–4h)
≈6.5
Krishnan et al. (2010)
Magnetic film array – ETP (40 mT)
Excised human epidermis
50 mg/mL urea
14.4 μg/cm2/h
45.0 μg/cm2/h
≈3
Benson et al. (2010)
12.2 Static Magnetic Fields
Murthy reported enhanced skin permeation of benzoic acid, salbutamol sulphate and terbutaline sulphate by magnetophoresis (Table 12.1) (Murthy 1999; Murthy and Hiremath 1999; Murthy and Hiremath 2001). These studies involved the use of stationary permanent magnets in close proximity to the donor formulation. Their initial study reported magnetophoresis enhanced the delivery of benzoic acid across excised rat abdominal skin (Murthy 1999). The static magnetic field was generated by permanent magnets of field strength 1 to 5 × 10−2 mT. The enhancement ratio for magnetophoretically delivered benzoic acid ranged from just less than 2 to approximately 2.5 with increasing magnetic field strength. Murthy concluded that the enhanced transdermal permeation was primarily due to the increased diamagnetic flow as benzoic acid has a high diamagnetic susceptibility (−70.3 × 10−6 cgs units) and is thus likely to be repelled away from the external magnet and driven into the skin along the direction of the magnetic field gradient. Diamagnetic susceptibility increases with magnetic field strength; thus the increase in enhancement ratio with magnetic field strength might suggest that diamagnetic flow is the likely mechanism for magnetophoresis. Murthy did not rule out the possibility of other mechanisms such as magnetohydrokinesis (movement of water) or altered barrier function due to the magnetic field.
Murthy’s group conducted a series of more extensive studies on lidocaine to further elucidate the mechanism of enhancement from static magnet fields (Murthy et al. 2010). In all in vitro and in vivo studies, skin permeability of lidocaine was greater under the influence of the static magnetic fields. In vitro studies were conducted across heat-separated porcine epidermis in Franz-type diffusion cells with magnetic field strengths of 30, 150 and 300 mT generated by placing two neodymium magnets on either side of the donor compartment at a distance of 2 mm from the epidermis. They confirmed their earlier reports that the penetration enhancement ratio increased with increasing magnetic field strength, with a range of 2.9–8.9 for lidocaine hydrochloride and 1.3–3.9 for lidocaine base (30–300 mT magnetic field strength).
Exposure of the porcine epidermis to the static magnetic field for 24 h prior to conducting a passive in vitro diffusion study showed no significant difference in lidocaine permeation compared to no prior exposure to the magnetic field (Murthy et al. 2010). In addition, there were no significant changes in electrical resistance and transepidermal water loss (TEWL) or shift in the peaks of the lipid or amide regions of the FTIR spectra of the epidermis in the presence of the static magnetic field. Thus no evidence of alteration to the epidermal barrier was detected. The authors suggested that the mechanism is likely to be similar to the mechanisms of electrorepulsion and electroosmosis that provide enhanced skin permeation in iontophoresis. In order to eliminate any skin barrier effects, they assessed the effect of the 300 mT static magnetic field on diffusion of lidocaine hydrochloride across a 1000 Da MWCO dialysis membrane, reporting a fourfold increase over passive diffusion. This confirms that magnetokinesis (combination of diamagnetic repulsion and magnetohydrokinesis) contributes to the mechanism of enhanced permeation by the static magnetic field. The magnetic field also generated a 2.7-fold increase in permeation of 3H-water across the porcine epidermis, suggesting that there is flow of water molecules with the magnetic field gradient (Murthy et al. 2010).
A 450 mT magnetophoretic patch was fabricated (Fig. 12.1), which was tested on rats with the permeation of both lidocaine base and hydrochloride monitored by cutaneous microdialysis (Murthy et al. 2010). An approximate two- to threefold increase in permeation was achieved with the magnetophoretic patch.
12.3 Pulsed Electromagnetic Fields (PEMF)
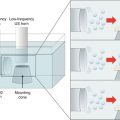
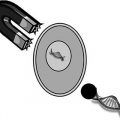
In contrast to the studies undertaken by Murthy, Benson’s group utilized pulsed electromagnetic fields (PEMF) generated by the Dermaportation technology that was designed and manufactured by OBJ Ltd, a Perth-based biotechnology company (www.obj.com.au). The Dermaportation system uses a low voltage (3 V) and does not require direct physical contact with the skin. The technology used in these experiments generates an asymmetrical pulse packet-type electromagnetic field comprised of a series of repeating quasi-rectangular waves of electromagnetic energy of 400 μs duration, with peak maximum field strength of 5 mT (Fig. 12.2). The electromagnetic pulse is propagated through the energizing of a small spirally wound monofilament air-filled coil which is placed externally to the donor compartment of a Franz-type diffusion cell so that the energizing coil is 7 mm above the skin surface (Fig. 12.3). The Dermaportation system utilizes a secure microprocessor smart card technology with automatic CRC data integrity testing and system integrity testing to ensure the quality and repeatability of the field characteristics between experiments. Whilst the momentary peak magnetic field is consistent with that used by Murthy’s group, the average field of 0.25 mT was substantially less.
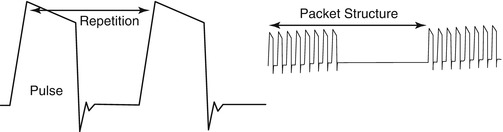


Fig. 12.2
Dermaportation waveform: time-varying magnetic fields use a complex ‘packet’ format to avoid increased entropy

Fig. 12.3
Dermaportation coil mounted on a Franz diffusion cell
PEMF by the Dermaportation system has demonstrated enhanced skin penetration of small molecules and peptides (Table 12.1) including 5-aminolevulinic acid (5-ALA) (Namjoshi et al. 2007), naltrexone hydrochloride (Krishnan et al. 2010), diclofenac (Benson et al. 2007), lidocaine hydrochloride (Cacetta et al. 2007) and dipeptide alanine-tryptophan (Ala-Trp) (Namjoshi et al. 2008). All experiments were conducted using excised human epidermis mounted in Franz-type diffusion cells, with the Dermaportation coil mounted on the exterior of the donor compartment of the cell (Fig. 12.2). Thus the magnet did not come into direct contact with the skin or the donor formulation.
A small preliminary investigation of the effect of PEMF on epidermal penetration of 5-ALA showed a 400-fold enhancement of epidermal flux over the first 2 h of administration (Table 12.1) (Namjoshi et al. 2007). 5-ALA is a very poor candidate for transdermal delivery and showed very low passive permeation (0.12 μg/cm2/h). In contrast, when applied with PEMF, the permeation rate (50.79 μg/cm2/h) was so high that donor depletion occurred within the first 2 h. Thus the flux values are estimated over a small number of data points and with a small number of replicates (n = 4). Consequently, whilst this preliminary study confirms the enhancement ability of PEMF, the extent of enhancement effect, which is much greater than reported in other studies (Table 12.1), should be treated with caution.
The applied PEMF significantly increased the skin permeation of the dipeptide Ala-Trp (Table 12.1) (Namjoshi et al. 2008). The enhancement appeared to be greatest in the initial 20 min suggesting an initial “push” of drug permeation by the magnetic field, which was then followed by a continued significantly enhanced permeation for the duration of magnetic field administration. Like 5-ALA, Ala-Trp is a poor candidate for transdermal delivery and shows low passive permeation (0.78 μg/cm2/h), thus allowing for a substantial enhancement ratio with an effective enhancement technology (19.43 μg/cm2/h with PEMF).
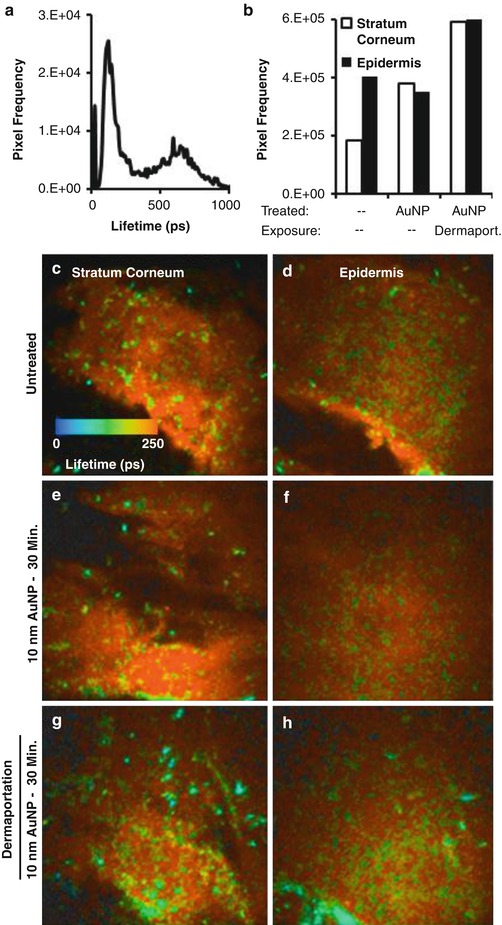
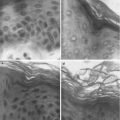
An enhancement ratio of approximately 5.7 was reported for PEMF administered naltrexone hydrochloride across human epidermis (Krishnan et al. 2010). A higher enhancement ratio of 6.5 was recorded during PEMF administration (0–4 h) followed by a lower but still enhanced permeation (5.3) for the following no PEMF phase (4–8 h). This shows a residual effect of the electromagnetic field on permeation, which the authors suggested could be due to an alteration of the skin barrier due to the applied electromagnetic field. Whilst there was a slight enhancement of naltrexone diffusion across silicone membrane with PEMF, this was not significantly different to passively applied naltrexone. This supports the view that the mechanism of enhancement for the PEMF is not predominately magnetokinesis, as Murthy has suggested for the static magnetic fields, but may be due to alteration of the stratum corneum barrier. To further investigate this theory, the authors applied 10 nm diameter gold nanoparticles to human skin with and without the presence of a PEMF. The distribution of the nanoparticles within the skin was monitored after 30 min by multiphoton microscopy–fluorescence lifetime imaging microscopy (MPM-FLIM). Gold nanoparticle-treated human skin exposed to the PEMF had 200 times more gold nanoparticle positive pixels than the skin exposed to gold nanoparticles without PEMF (Fig. 12.4). This suggests that the PEMF facilitates penetration of the gold nanoparticles through the stratum corneum and that the channels through which the nanoparticles move must be larger than their 10 nm diameter. There were no major differences in the microanatomy of the stratum corneum or epidermis between the treatment groups, indicating no obvious tissue damage occurred due to the PEMF.