Keystone and Related Perforator Flaps
Michael W. Findlay
DEFINITION
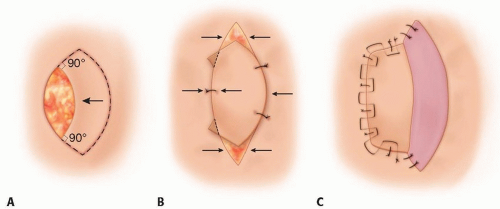
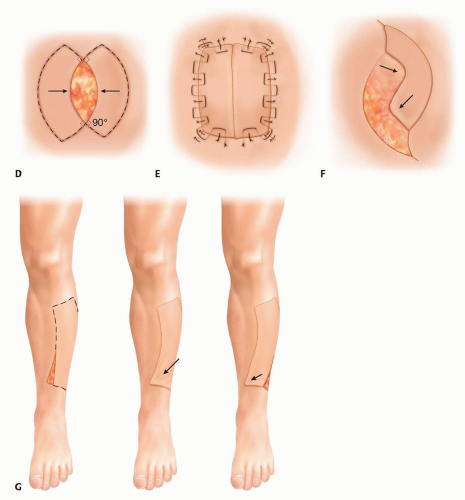
Keystone flaps encompass a group of related fasciocutaneous flaps first devised by Behan that share a common geometric design (a keystone shape similar to the keystone of stone arches) but can vary in the following ways:1
Blood supply (perforator vs direct vs neurovascular vs composite)
Degree of islandization (complete vs incomplete, cutaneous or fasciocutaneous)
Method of transfer into the defect (advancement, transposition, combination)
Whether used alone or along with another keystone flap on the opposite side of the defect (double opposing keystone flaps).
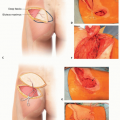
The classification of keystone flaps is based on the method of transfer and the number of keystone flaps involved (FIG 1).
ANATOMY
Keystone flaps close defects by transferring fasciocutaneous tissue from an adjacent donor site to the defect while maximizing use of cutaneous arteries, superficial and deep venous drainage, and lymphatics without being defined by a specific form of the transfer (eg, transposition vs rotation).
Human skin is well supplied with perforators and direct vessels for supply of keystone flaps with established patterns of cutaneous supply in the limbs (longitudinally oriented septocutaneous and musculocutaneous perforators), the trunk (segmental perforators with intercommunications in welldefined intermuscular planes), and the head and neck (profuse cutaneous supply dominated by direct cutaneous vessels in the head and perforators and direct vessels in the neck).2
Conjoint supply to flaps refers to the incorporation of both cutaneous vessels and neurovascular supply within the flap (eg, incorporation of saphenous or sural nerves and their vasa nervorum) so as to augment blood supply. Codevelopment of arteries and nerves during development result in conjoint flaps that have a very strong axial blood supply that improves their robustness. An additional benefit is that blood supply from the vasa nervorum can be significant in some areas (eg, posterior thigh), further contributing to flap vascularity.
Certain sites do not contain cutaneous perforators (eg, pretibial border, scalp) and hence represent problem areas for keystone flap elevation. Keystone flaps can be raised either side of the pretibial border for mobilization of tissue over a pretibial defect, but cannot reliably be designed directly over the pretibial skin as the basis for its blood supply due to the lack of direct cutaneous perforators.
Superficial and deep venous drainage can be incorporated in most keystone perforator flaps through preservation of deep venous system along with arterial perforators and the subcutaneous veins by blunt dissection of subcutaneous tissues and islandization of the underlying fascia (if necessary) via the use of scissors through small windows in the subcutaneous tissue. Superficial veins are often numerous, and some may need to be divided to permit sufficient mobilization, but their division is performed sequentially, stopping once sufficient mobilization is achieved.
The longitudinal orientation of fascial planes in many areas of the body (eg, limbs, paraspinous region) predispose to a longitudinal orientation for keystone flaps (slightly oblique orientations may help in limbs; see below), with the most appropriate mobilization of longitudinal keystone flaps
being in a transverse direction. That is, the longitudinal orientation of the paraspinous musculature makes medial and lateral movement of keystone flaps relatively easy to cover central back defects, whereas the fibrous attachments between the skin and underlying spinous processes make superior or inferior movement problematic.
Where there is circumferential limitation for flap mobilization and direct closure of the secondary defect (eg, distal limbs), orienting keystone flaps oblique to the long-axis provides an oblique (longer) circumference, thereby maximizing available donor tissue for closure.
Placement of incisions in relaxed skin tension lines where possible is very helpful. The keystone shape is one that naturally hides well. Caution should be exercised in selection of any island flap in patients prone to abnormal scarring (eg, hypertrophic/keloid scarring). Partial flap islandization is a good option where feasible in such individuals or in patients very sensitive about the scar burden.
Undermining and skeletonization of the perforator/direct vessel pedicle to the flap can be undertaken but is unnecessary in the vast majority of cases, and therefore, only the division of structures felt absolutely necessary for elevation, mobilization, and inset of the specific keystone flap are undertaken, thereby ensuring both surgical efficiency and safety.
The design and flexibility in the blood supply, venous drainage, and mobilization of the keystone flap make it readily suitable for application in most areas of the body with local variation to optimize the outcome in specific areas.
PATIENT HISTORY AND PHYSICAL FINDINGS
An understanding of the etiology of the defect to be reconstructed and relevant patient factors (health, comorbidities, activity levels, mobility, nutrition) help direct the most appropriate form of reconstruction in any potential flap case.
As with all forms of flap reconstruction, particular interest should be focused on removal of the causative factor (eg, cancer, infection), identification and optimization of locoregional blood flow, selection of the best possible flap based on the needs of the defect and locoregionally available tissue, and optimization of the patient in the perioperative period.
Any past history of conditions that affect local blood flow and wound healing should be elicited and mitigated where possible (eg, irradiation, peripheral vascular disease (micro
and/or macroangiopathy), diabetes mellitus, smoking, and previous surgery/trauma.
Examination findings of note include quality of surrounding donor sites for skin and tissue quality (including presence of radiotherapy tattoos, scars, fibrosis).
IMAGING
The vast majority of patients do not require preoperative imaging or the use of intraoperative Doppler ultrasound to achieve reliable and timely reconstruction using the keystone flap or its variants as long as normal design principles are followed (see FIG 1).
Computed tomography, angiography, and similar imaging modalities are the most useful to answer what direct or perforating vessels are available to the locoregional tissues, and delayed views can show the venous drainage to the area of interest.
Fluorescence microscopy is invaluable for pre- and intraoperative imaging of the perforators and direct vessels to the skin but is rarely indicated for keystone flap elevation and has anaphylaxis as a risk factor.
Imaging can be of use where previous surgery or trauma may have damaged the normal blood supply of the donor skin, or if there is significant arterial disease, and therefore, the selection of the most appropriate local vessels is necessary to ensure success.
NONOPERATIVE MANAGEMENT
As with any fasciocutaneous defect, careful consideration should be given to the potential of the defect to be closed directly (under physiologic tension and utilizing biological and mechanical creep as tolerated) or healed by secondary intention.
The progression to flap reconstruction is considered where these options are either not possible or lead to unacceptable risks/complications.
SURGICAL MANAGEMENT
Keystone flap reconstruction is reliable as long as a sequence of steps is undertaken in which the primary goal is sufficient mobilization of the flap to close the recipient and donor sites, while preserving as much arterial input, venous output, and lymphatic drainage as possible.
No additional maneuvers are undertaken once recipient and donor-site closure can be achieved.
If further mobilization of tissues is needed, then the flap can be fully islandized where the cutaneous vessels have been followed back through deep fascia and or freed where they exit muscles to facilitate mobilization; however, one must be cautious not to damaging perforating vessels as this can lead to tissue ischemia and necrosis.
Preoperative Planning
The defect is assessed for its suitability for fasciocutaneous reconstruction, with a consideration of locally available tissue, including potential vascular pedicles, as the basis for the flap.
Keystone flaps can be performed in irradiated tissue1 or even in the setting of lymphedema,2 but caution should be exercised with flap planning in these settings so as to ensure adequate perforators/direct vessels and avoid unnecessary tension across suture lines.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree