(6.1)
where t #.max.drug; z drug and F are the maximum transport number achievable for the drug, the valence of the drug and the Faraday constant, respectively. Alternatively, the input rate can be approximated from the daily dose. Please note that experimentally measured maximum transport numbers will include both the electromigration and electro-osmotic contributions for a charged drug and are more correctly termed transference numbers. In the case of an uncharged drug, only transported by electro-osmosis, Eq. 6.2 can be modified to:

(6.2)
Once the intensity of current required is known, the current density (CD) resulting from such an administration can be determined using Eq. 6.3:
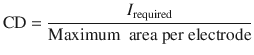
The maximum area per electrode will be that considered acceptable for the specific iontophoretic application and body site envisaged. Note that the total iontophoretic patch area would be, at least, double this value. Traditionally, the value of 0.4–0.5 mA.cm−2 has been considered as an upper limit for current density above in which sensations of tickling, warmth or discomfort experienced by the user become less tolerable (Ledger 1992). Figure 6.1 shows some simulations illustrating the discussion above.
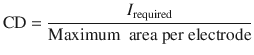
(6.3)

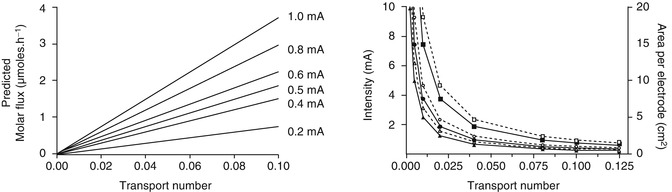
Fig. 6.1
Feasibility simulations in iontophoresis. Left panel: molar flux (μmol.h−1) achievable for model monovalent drugs for different transport numbers and intensity of current applied. Right panel: area per electrode (dashed lines, open symbols) and current intensity (solid lines, filled circles) required to deliver 10 mg of hypothetical monovalent drugs of 150 (squares), 300 (circles) and 450 (triangles) molecular weight in 24 h. The current density was fixed at 0.4 mA.cm−2 (The data was generated using Faraday’s law (Eq. 6.1))
The previous method allows a quick estimation of feasibility, that is, whether an iontophoretic administration merits further development. However, this method is primarily a steady-state calculation. Subsequent stages of the feasibility assessment should look into the kinetics of the process and the fact that iontophoretic fluxes require some time to reach steady values. The length of this time varies for different drugs and seems to be related to both the physicochemical properties of the drug and the presence of endogenous ions within the skin (Phipps and Gyory 1992). Thus, a more realistic approach should consider the magnitude of fluxes achieved during the length of current passage envisaged. Some of these concerns have been addressed by Nugroho et al. (2004).
Finally, the dose delivered in an iontophoretic administration can be estimated through Eq. 6.4:

Thus, at least theoretically, the same dose can be delivered by judicious manipulation of the application time and the iontophoretic fluxes (the latter being typically modified by changing the intensity of current applied for the same iontophoretic vehicle). This dosing flexibility is indeed an advantage of iontophoresis over other forms of controlled drug delivery. All the same, the concept of bioavailability encompasses both the extent and rate of drug absorption, and therefore, to achieve therapeutic efficacy, both the amount of drug absorbed and the rate with which it is absorbed must be appropriate to the pharmacodynamics and pharmacological properties of the drug.

(6.4)
6.2 Iontophoresis Drug Delivery for Local and Topical Therapies
The most widespread applications of iontophoresis are the treatment of palmoplantar hyperhidrosis and the diagnosis of cystic fibrosis. Other popular applications are the iontophoretic delivery of lidocaine, acyclovir and dexamethasone phosphate.
The treatment of palmoplantar hyperhidrosis is one of the oldest applications of iontophoresis (Kreyden 2004). Hyperhidrosis, an excessive rate of sweat secretion from the eccrine glands, is a disabling condition that affects both children and adults. Iontophoresis is one of the most effective, safest and inexpensive treatment options available. Although it has been applied for a long time in the treatment of this pathology, the mechanism of action of iontophoresis is unknown. Different options have been considered in the treatment of hyperhidrosis with iontophoresis, including the application of tap water, saline, botulinum toxin or anticholinergics. Anodal iontophoresis is more efficient than cathodal iontophoresis, and tap water is more effective than saline (Sato et al. 1993). Iontophoresis with anticholinergics is more effective than tap water iontophoresis, but may induce systemic side effects (Dolianitis et al. 2004). The administration of botulinum toxin by iontophoresis (the dose was administered in one 30-min session with a current intensity of 3 mA) has shown to reduce sweating more quickly and for a longer period than saline, without side effects (Davarian et al. 2008). Potentially, the iontophoretic administration of this toxin would avoid the painful injections required to this day even though the duration of effect observed is shorter. The apparent success in delivering this large toxin could be related to the high density of sweat glands in the palms and the very high potency of this active.
Iontophoresis is an integral part of the method used to diagnose cystic fibrosis. The disease results from a mutation in the gene encoding the cystic fibrosis transmembrane conductance regulator. Among other functional problems, this causes extensive dysfunction of the exocrine glands which constitutes the basis for the diagnosis of cystic fibrosis, basically a sweat test. First, pilocarpine is iontophoretically (5 min × 2.5–3.0 mA) delivered to a small area of the arm or leg to stimulate sweating. The sweat is then collected and analysed for volume and chloride content (Beauchamp and Lands 2005).
Delivery of topical anaesthetics prior to dermal surgery remains one of the most common applications of iontophoresis. The iontophoresis of hydrochloride salts of anaesthetics of the amide type, such as lidocaine (Maloney et al. 1992), bupivacaine, etidocaine, mepivacaine, prilocaine and ropivacaine, has been frequently reported (Brouneus et al. 2001). Several studies demonstrated that lidocaine iontophoresis and the eutectic mixture of lidocaine and prilocaine (EMLA®, AstraZeneca PLC, UK) were of a similar efficacy in providing the analgesia necessary for cannulation and CO2 laser surgery of superficial skin lesions (Phahonthep et al. 2004; Galinkin et al. 2002). Purpose designed, disposable electrodes (Numby Stuff® from Iomed Clinical Systems, Salt Lake City, USA) containing lidocaine HCl and epinephrine were the first available to be used in combination with a Phoresor power supply. The Phoresor II Auto PM850 is a programmable, direct current delivery device which can apply up to 4 mA and uses a 9-volt battery. Later on, the LidoSite® Topical System (lidocaine topical anaesthetic system) was developed by Vyteris, Inc. (Fair Lawn, NJ), and approved by the US FDA in 2004. The LidoSite® was a small, easy-to-use, preprogrammed iontophoretic lidocaine delivery system composed of a drug-filled patch connected to a controller (Kalia et al. 2004). Several trials have investigated the safety and effectiveness of lidocaine iontophoresis and compared it with either placebo or with EMLA cream in adults and children (Galinkin et al. 2002; Kearns et al. 2003; Rose et al. 2002; Squire et al. 2000). Briefly, the results indicate that both techniques provide similar pain relief; however, the application time is greatly reduced for iontophoresis (5–15 min) in comparison to the 50–90 min required by the EMLA cream. Therefore, the faster delivery of lidocaine via iontophoresis provides a faster onset of analgesia. Regrettably, the LidoSite® System was later discontinued. Despite this, lidocaine iontophoresis still attracts interest and is object of ongoing clinical research according to ClinicalTrials.gov. For example, clinical trials sponsored by Dharma Therapeutics, Inc., USA, currently look into the same application (lidocaine/epinephrine to provide local anaesthesia for venepuncture), whereas other trials sponsored by Acclarent, Inc., USA, investigate the anaesthetic effect of lidocaine/epinephrine iontophoretically delivered to the external auditory apparatus to treat otitis. This new application can be implemented using the Tula® Iontophoresis System also by Acclarent, Inc., USA, which is object of another clinical trial. Unfortunately, no results have been posted in any case.
The efficacy of topical creams and ointments in the treatment of herpes labialis remains a controversial issue, more precisely their relative benefit with respect to spontaneous healing. This poor outcome has been attributed to the low penetration of the drug across intact skin and its insufficient delivery to the basal epidermis, the targeted site of infection. The possibility of enhancing the skin permeability of acyclovir via an electric current was investigated in several occasions. Morrel et al. demonstrated that a single dose of a conventional formulation of acyclovir (ACV) via iontophoresis was a convenient and effective treatment for cold sores (Morrel et al. 2006). ACV is an ampholyte drug with two pKa values (2.27 and 9.25); thus, it is primarily cationic below pH 2, neutral between pH 3 and 8.5 and anionic at pH greater 9.5. Thus, depending on the pH of the vehicle, ionized ACV can be delivered both by anodal or cathodal iontophoresis and neutral ACV by electro-osmotic flow (Padula et al. 2005). Shukla et al. (2009) tested the iontophoretic delivery of ACV from different formulations (neutral creams and pH 11 gels); according to these authors, only the neutral drug solubilized in the water phase of the creams was available for transport. On the other hand, delivery of the negatively charged ACV from the pH 11 gels increased the dermis exposure to the drug. For example, the AUC0–60 (area under the curve ACV interstitial fluid concentration versus time) measured by microdialysis were 8.8 ± 1.5 mg.min.L−1, 174 ± 078 mg.min.L−1 and 13.3 ± 4.8 mg.min.L−1 for the neutral cream, pH 11 gel iontophoresis and pH 11 gel passive, respectively. Apparently, the high pH did not cause skin irritation. At the time of this review, two phase II clinical trials sponsored by Transport Pharmaceuticals, Inc. (USA), on the effectiveness of iontophoretic delivery of ACV to treat herpes labialis are listed in ClinicalTrials.gov, unfortunately with no results posted yet.
The efficacy of iontophoresis to deliver other antiviral agents used against herpes labialis has also been reported. Iontophoresis was used to deliver the antiviral drug idoxuridine to 14 recurrent herpes labialis lesions in a clinical trial involving six patients. Results were characterized by immediate relief of discomfort and swelling, rapid appearance and coalescence of vesicles, minimal or no spread of the lesions and accelerated healing with minimal or no scab (Gangarosa et al. 1979). Gangarosa et al. (1986) compared iontophoresis of vidarabine monophosphate (ara-AMP) and acyclovir for efficacy against herpes orolabialis. Patients with vesicular orolabial herpes participated in a double-blind, placebo-controlled clinical study comparing iontophoresis of vidarabine monophosphate (ara-AMP, n = 9), of acyclovir (ACV, n = 9) and of NaCl (placebo group, n = 9). Ara-AMP-treated lesions yielded lower titres of virus after 24 h compared with lesions treated with NaCl or ACV. Ara-AMP significantly decreased the duration of shedding of virus compared to the placebo group (Gangarosa et al. 1986).
The iontophoretic administration of dexamethasone sodium phosphate (DP) to treat local pain and soft tissue inflammation in sports medicine remains a popular application. The disodium salt DP is water-soluble and, at a physiological pH, is present mainly in its dianionic form (pKa, 1.9 and 6.4). DP is relatively well delivered to the skin by cathodal iontophoresis, and, in this case, the challenge is to ensure that a sufficient DP reaches the target tissue beneath the skin despite of the drug’s systemic clearance. Iontophoretic treatment of various musculoskeletal problems with DP has been the subject of clinical studies employing a great variety of protocols, many of which have reported beneficial effects (Nirschl et al. 2003; Li et al. 1996), though improvement in the patient’s condition has not been always observed (Reid et al. 1994; Runeson and Haker 2002). Again, it has been demonstrated that, to achieve optimal iontophoretic delivery, DP must be delivered from the cathode and formulated rationally, excluding mobile competing co-ions, such as chloride ions (Fig. 6.2) (Sylvestre et al. 2008a,b). When properly implemented, iontophoresis enhances significantly the delivery of DP; for example, the combined amounts of dexamethasone sodium phosphate and dexamethasone in the treated SC (0.78 cm2) were estimated via tape stripping to be 9.9 ± 3.3 nanomoles and 0.69 ± 0.29 nanomoles after 3 h of iontophoresis and 3 h of passive diffusion, respectively (Sylvestre et al. 2008a).
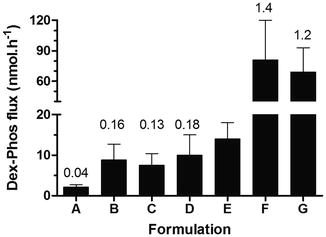

Fig. 6.2
Donor composition, primarily the presence of competing co-ions, has a significant impact on the cathodal iontophoretic flux of dexamethasone phosphate (DP). The formulations tested were A (1:2 injectable DP 0.4 %, Faulding (Mayne Pharma PLC, UK) + lidocaine 4 % (AstraZeneca, UK)), B (injectable DP 0.4 % (American Regent, USA)), C (injectable DP 0.4 %, Faulding), D (DP 0.4 % in potassium citrate), E (DP 0.4 % in 0.9 % NaCl), F (injectable DP 0.5 %, Organon, UK) and G (DP 0.4 % in water). The current intensity was 0.3 mA (0.38 mA.cm−2). Numbers on top of the columns indicate the average transport number of the drug expressed as percentage (Data taken and redrawn from Sylvestre et al. 2008b)
The topical iontophoresis of non–steroidal anti–inflammatory drugs (NSAIDs) is attractive in that this alternative, local delivery method would reduce the risk of GI-related side effects commonly encountered after oral administration. The cathodal iontophoresis (0.625 mA.cm−2 for 90 min) of a series of NSAIDs (salicylic acid, ketoprofen, naproxen and indomethacin) suggested an important effect of the drug lipophilicity on their in vivo delivery in rats (Tashiro et al. 2001). NSAID skin concentrations were higher for the most lipophilic drugs, whereas the cutaneous plasma concentrations decreased for the most lipophilic actives. According to the authors, the rate of transfer of NSAIDs from the skin to the cutaneous vein decreased with increasing lipophilicity. These results underline the key role which drug partitioning between the stratum corneum (SC) and the viable epidermis may have in determining the local bioavailability of drugs (Tashiro et al. 2001).
A double-blind study compared the efficacy and tolerability of pirprofen and lysine soluble aspirin (Flectadol®, Maggioni-Winthrop S.p.A., Milan, Italy) administered by iontophoresis to 80 patients with various painful rheumatic diseases. The treatment lasted 2 weeks and consisted of five administrations a week, each lasting 20 min. After five administrations, patients showed significant improvement in pain at rest, and on movement, no significant differences were observed between the pirprofen and aspirin groups. The final outcome was described as excellent or good for ~75 % of the treated patients with a satisfactory functional improvement being reported for ~80 % (Garagiola et al. 1988).
A commercially available gel containing piroxicam was used to administer this drug both passively and with iontophoresis to human volunteers (Curdy et al. 2001). The total amount of drug recovered from the SC via tape stripping was significantly higher post-iontophoresis (48.6 ± 18.8 μg.cm−2) than after passive diffusion (5.4 ± 2.0 μg.cm−2).
The iontophoresis of ketoprofen was efficacious and safe in the treatment of acute osteoarthritic diseases also in high-risk patients (Salli 1993). Three hundred and twelve patients with osteoarthritic diseases were admitted to this multicentre clinical trial and received ketoprofen by iontophoresis twice a day for ten consecutive days. Pain relief was almost complete in 94.6 % of the patients, improvement of active motility in 83.6 %, disappearance of swelling in more than half of the patients and functional improvement in about all the cases. Two allergic skin reactions of a mild degree (in 0.6 % of patients) were registered.
Ketorolac has also been successfully delivered by iontophoresis to human volunteers. The drug was delivered using silver electrodes with a current of 2 mA for five treatment sessions for 20 min every day (Saggini et al. 1996).
A double-blind randomized study compared ionization with diclofenac sodium and ionization with saline solution in two groups of patients with scapulohumeral periarthritis or elbow epicondylitis. Both groups were treated with 20 ionization sessions each lasting 30 min during a 1-month period. There was a significantly greater improvement in pain at rest, pain on pressure, pain on movement and joint swelling in the 11 patients treated with diclofenac sodium compared with the 13 placebo-treated patients, but no significant differences between the two treatments as regards functional impairment were observed (Vecchini and Grossi 1984). However, other studies have reported side effects including a systemic adverse reaction (Macchia et al. 2004) and allergic contact dermatitis (Foti et al. 2004).
The iontophoretic administration of antineoplastic agents to treat skin cancer has been proposed to avoid the scarring associated with surgery, the long-term complications of radiation therapy and the adverse effects associated with the systemic administration of these actives. Chang et al. (1993) investigated whether iontophoretic delivery of cisplatin could be used to treat basal and squamous cell carcinomas in a group of patients who refused surgery. Cisplatin was delivered from the anode together with the vasoconstrictor epinephrine hydrochloride; the dose of drug and current applied depended on the size of the lesion. Eleven of the 15 patients showed either a partial reduction in the lesion area or a complete response. There were no incidences of systemic side effects (nausea or vomiting) although a minor burning sensation at the cathode was reported. The authors concluded that small lesions responded better and suggested a treatment schedule involving a daily iontophoretic therapy of 20–30 min for 5 days followed by a 2-week recovery period.
Smith et al. (1992) investigated the iontophoresis of vinblastine sulphate as a possible treatment for the cutaneous lesions associated with Kaposi sarcoma in human immunodeficiency virus-positive patients. The first step involved the iontophoretic delivery (4 mA, 10 min) of both lidocaine and epinephrine; after which vinblastine sulphate was delivered (4 mA, 10–90 min) on the same skin site. A non-HIV-infected group showed signs of local erythema which cleared up within 2 weeks. However, less inflammation was observed in the HIV-1 patients treated over a period of 6 months. This difference could be explained by the immunosuppression at advanced stages of the illness experienced by the patient group. All patients showed a significant clearing of the lesions.
Cathodal iontophoresis enhanced the transdermal flux of methotrexate as compared to passive diffusion (Alvarez-Figueroa et al. 2001); it was not clear whether therapeutic local concentrations could be achieved given that target values are unknown. A topical dosage form of this drug for the treatment of psoriasis would be of great interest, as the systemic administration use of this drug results in numerous side effects.
Iontophoretic delivery of 5-fluorouracil (5-FU) appears to be a safe, effective and well-tolerated therapy for Bowen’s disease. Twenty-six patients with biopsy-proven Bowen’s disease received eight 5-FU iontophoretic treatments in 4 weeks (Welch et al. 1997). A local excision procedure took place 3 months after the last treatment, and the specimens were step-sectioned and evaluated for any histologic evidence of bowenoid changes. Only one of the 26 patients showed histologic evidence of Bowen’s disease 3 months after treatment. A case report described a cutaneous allergic reaction in one patient after 5-FU iontophoretic treatment (Anderson et al. 1997).
Photodynamic therapy (PDT) is now considered as a valuable alternative treatment of cancer. PDT involves the administration of a tumour-localizing photosensitizer or photosensitizer prodrug (5-aminolevulinic acid [ALA], a precursor in the heme biosynthetic pathway) and the subsequent activation of the photosensitizer by light (Peng et al. 1997). Using iontophoresis, a rapid and quantifiable system for topical ALA delivery, with measurement of subsequent protoporphyrin IX (PpIX) fluorescence and phototoxicity, has been reported (Rhodes et al. 1997). ALA was iontophoretically delivered from a 2 % solution into the upper inner arm skin of 13 healthy volunteers with the delivery being sufficient to induce tumour necrosis. Iontophoretic transport of ALA across human SC was studied quantitatively in vitro by Bodde et al. (2002). The results showed that the amount of ALA that passively diffuses through the SC in several hours, leading to therapeutic levels of PpIX in the epidermis, can be delivered by iontophoresis in 10 min or less. However, because the formation of sufficient PpIX also requires several hours and also because the SC overlying skin lesions such as basal cell carcinoma (BCC) is not intact, the clinical benefit of topical ALA delivery by iontophoresis for PDT of BCC is yet to be established.
Finally, the delivery of calcitonin gene-related peptide (CGRP) and vasoactive intestinal polypeptide (VIP) has shown potential to treat venous stasis ulcers (Gherardini et al. 1998). The anodal electrodes (40 cm2) contained drug reservoirs filled with 1 mL of a 3-nM CGRP or of a 0.3-μM VIP; the cathodal electrode reservoirs contained sodium phosphate buffer. This iontophoretic set-up was applied to intact skin in the proximity of the ulcer in 66 patients, and pulsed electric current was delivered for 20 min. The clinical results suggested that both peptides’ delivery was enhanced and that the electric current passage close to the ulcer area had a positive influence on the healing process.
6.3 Iontophoretic Transdermal Drug Delivery
Until very recently, only the fentanyl iontophoretic system (Ionsys®, Alza Corporation, USA) had been marketed as a transdermal drug delivery system. In January 2013, the sumatriptan iontophoretic delivery system, ZecuityTM by NuPathe Inc., USA, was approved by the FDA for the acute treatment of migraine.
The Ionsys® System was indicated for the short-term management of acute postoperative pain in adult patients requiring opioid analgesia during hospitalization. Fentanyl physicochemical and pharmacological properties together with its pharmacokinetics – short half-life and high first-pass effect – make it an ideal candidate for transdermal administration (Chelly et al. 2004). Passive transdermal fentanyl patches for the treatment of chronic pain have been marketed for nearly 20 years but cannot provide the rapid bolus of drug input required to treat acute pain. The feasibility of fentanyl iontophoresis was demonstrated in vitro and in vivo (see Fig. 6.3) (Thysman and Preat 1993; Ashburn et al. 1995) and led to the development of the Ionsys® System (using E-TRANS® electrotransport technology, Alza Corporation, USA) – a preprogrammed, self-contained, on-demand drug delivery system activated by the patient which can deliver 80 doses of 40 μg of fentanyl in a 24-h period. The Ionsys® was designed to operate for 24 h after the first activation and allowed 6 doses per hour up to a maximum of 80 doses, after which the system shut off. Subsequently, a new transdermal system is required if the fentanyl administration is to be continued. The transdermal system used a 10-min transdermal infusion for each 40-μg dose (Gupta et al. 1999; Gupta et al. 1998). Clinical studies showed that the Ionsys® System was well tolerated by patients suffering postoperative pain and was equivalent to a standard intravenous morphine pump (Viscusi et al. 2006; Minkowitz et al. 2007; Ahmad et al. 2007; Hartrick et al. 2006). Ionsys was marketed in Europe by Janssen-Cilag, an affiliate of Alza; however, a defect involving corrosion of a component within the system was found by Janssen-Cilag in 2008. There had been neither complains linked to the defect nor evidence of patient harm; nevertheless the device was recalled as a precautionary measure. Because the root cause of the defect was not identified, the marketing authorization was finally suspended by the European Medicines Agency (EMA) (EMA 2008). The current marketing status of the device at the Food and Drug Administration (FDA) website is also discontinued. In January 2013, The Medicines Company (Parsippany, NJ, USA) acquired Incline Therapeutics, Inc., which had previously acquired the rights to Ionsys from Alza, with intention to resubmit the product marketing approval in the USA and Europe.
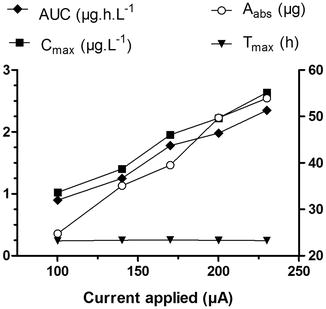

Fig. 6.3
Current intensity controlled the extent of fentanyl absorbed as shown by changes in AUC (area under the curve plasma concentration vs. time, filled diamonds), C max (maximum plasma concentration, filled squares) and the amount of fentanyl absorbed per 10-min dose, A abs (open circles), but did not modified t max (time to reach C max, filled triangles). Fentanyl HCl was delivered using a patient-controlled transdermal system, 16–35 volunteers received each treatment (Data taken and redrawn from Sathyan et al. 2005)
Other opioid analgesics are good candidates for iontophoresis from both physicochemical and pharmacological standpoints. A prospective, randomized, single-blind study investigated the effectiveness of iontophoretically delivered morphine hydrochloride to control postoperative pain in 38 patients who underwent total knee or hip replacement. Postoperative pain was initially controlled with IV meperidine and, thereafter, with a patient-controlled analgesia (PCA) device also administering meperidine. The following morning, either morphine hydrochloride or lactate ringer’s solutions (control) were delivered iontophoretically for 6 h. Because of the red wheal and flare observed under the anodal compartment, apparently due to the local histamine release provoked by morphine, the investigators were aware of the patients who received morphine, and only the patients were blinded. PCA analgesia remained available to patients during this period and for 12 h after iontophoresis. The morphine group usesd significantly less PCA meperidine both during iontophoresis and for the following 12-h post-iontophoretic treatment as compared to the lactate group (Ashburn et al. 1992).
The iontophoretic delivery of apomorphine for the treatment of idiopathic Parkinson’s disease was evaluated in human subjects. Although some encouraging results were obtained, they will require further confirmation, especially as the skin required pretreatment with surfactants (Bodde et al. 1998; Li et al. 2005) to further improve delivery of the drug. In vivo studies in rats suggest that therapeutic levels of ropinirole hydrochloride could be achieved via iontophoresis (Luzardo-Alvarez et al. 2003). However, the results will need further confirmation in humans and after formulation of a suitable electrode patch.
The iontophoretic delivery of tacrine, the first centrally acting cholinesterase inhibitor approved for the treatment of Alzheimer’s disease, in healthy adult volunteers was investigated in two occasions. One study used Iogel® silver/silver chloride electrodes (Chattanooga Group, Inc., USA), whereas the second one employed a novel two-compartment electrode system in which the drug reservoir was separated from the electrode by a membrane; this arrangement maximized the drug transport efficiency by avoiding competition with co-ions. A 0.4-mA.cm−2 current was applied for 3 h using patches with an active surface area of 10 cm2. Apparently, both types of electrodes would be able to provide tacrine blood levels similar to those observed following oral administration of the drugs (Kankkunen et al. 2002).
The treatment of migraine also represents an interesting application for iontophoresis. Several in vitro (Femenia-Font et al. 2005) and in vivo studies carried out with sumatriptan in human volunteers (Siegel et al. 2007) suggested that iontophoresis could provide adequate drug plasma levels for migraine treatment. Similar results have been obtained with zolmitriptan (Patel et al. 2009). A phase III randomized, double-blind, placebo-controlled trial evaluated the efficacy and tolerability of a sumatriptan iontophoretic transdermal system for the acute treatment of migraine in 469 patients. Patients on sumatriptan iontophoresis experienced less nausea, photophobia and phonophobia than those on placebo iontophoresis; further they had a rapid and sustained headache pain relief and used less rescue medication than the placebo group. About 50 and 44 % of patients treated with the sumatriptan iontophoresis and placebo iontophoresis experienced treatment-related adverse reactions, most of which were transient and mild-to-moderate application side effects. Therefore, sumatriptan iontophoresis was effective and well tolerated and particularly advantageous in migraine patients with nausea (Goldstein et al. 2012). In January 2013, the FDA (Drugs@FDA 2013) cleared the Zecuity iontophoretic transdermal delivery system by NuPathe Inc., USA, which delivers 6.5 mg of sumatriptan over 4 h.
The following paragraphs summarize the key progress in peptide and protein delivery by iontophoresis. Comprehensive and recent reviews on this topic field are available elsewhere (Gratieri et al. 2011). Early work demonstrated that iontophoresis could deliver peptides systemically, more specifically the tripeptide, threonine-lysine-proline (Thr-Lys-Pro) after topical application onto hairless rat skin (Green et al. 1992). In vivo results were consistent with the measured flux of the peptide in vitro. Iontophoretic pretreatment of rats resulted in enhanced delivery of the peptide subsequently applied passively. No important changes in skin morphology were observed following the current passage.
Currently, there is wide evidence supporting that iontophoresis can successfully deliver peptides and proteins in their active form. However, the therapeutic and commercial usefulness of the technique will depend on the dose and input profiles required for each application and on which alternative routes of delivery are available for the peptide considered. An increase in delivery rates of the somatostatin analogue, octreotide, was obtained in vivo in rabbits when mild current densities (50–150 μA.cm−2) were applied (Lau et al. 1994). Peptide plasma levels increased proportionally with the intensity of the current applied and drug input declined quickly upon current cessation. Several iontophoretical studies have investigated calcitonin, a 32-amino acid peptide with a molecular weight of ~3500 Da which, under physiological conditions, is positively charged. For example, human calcitonin was delivered into hairless rats, although lowering of serum calcium was not linearly dependent upon either the current density or time of current application (Thysman et al. 1994). The possibility of delivering calcitonin topically to the dentin in order to treat invasive cervical resorption (Kitchens et al. 2007) and systemically to treat osteoporosis and Paget’s disease has been reported as well. Nevertheless, no studies have been carried so far in humans. Salmon calcitonin, which is more potent than the human form, has been iontophoretically delivered in animal skin models with the blood levels attained being comparable to those achieved by intravenous infusion (Santi et al. 1997; Chaturvedula et al. 2005). Iontophoresis of LHRH has been investigated in vivo in pigs (Heit et al. 1993). Elevated LHRH concentrations were measured in the blood and concomitant increases in LH (luteinizing hormone), and FSH (follicle-stimulating hormone) levels were observed, demonstrating that the hormone had been delivered as a pharmacologically active species. Further, the circulating levels of LHRH fell rapidly upon the termination of iontophoresis. A larger peptide, growth hormone-releasing factor, GRF (1–44) (MW 5040), was delivered by iontophoresis into hairless guinea pigs, resulting in steady-state plasma levels of ~0.2 ng.mL−1 which were associated to a ~3.16-μg.h−1 transdermal flux (Kumar et al. 1992).
The iontophoretic delivery of the LHRH analogue leuprolide has been investigated in humans (Meyer et al. 1990; Lu et al. 1993). The application of a low current intensity (0.2 mA) over 70 cm2 resulted in LH levels comparable to those obtained after subcutaneous injection (Meyer et al. 1990). These results are remarkable, considering the low current density (~3.1 μA.cm−2) employed. Only 15 % of the subjects reported a tingling sensation during current passage, and 46 % observed at the electrode sites some erythema which resolved quickly after current termination. The effect of formulation variables on the iontophoretic delivery of leuprolide was also studied in humans (Lu et al. 1993). The lowest leuprolide concentration provided the highest transport. Vehicles formulated with different ionic strengths were evaluated. Systemic leuprolide levels differed, but there was little effect on the LH and testosterone concentration. This observation was attributed to differences in the pharmacological response of the subjects included in the study.
Despite the many in vitro and in vivo studies performed on peptide iontophoresis (Kochhar and Imanidis 2004; Nair and Panchagnula 2004; Chaturvedula et al. 2005), the prediction of iontophoretic fluxes and blood levels attainable for a given compound remains difficult. Computational studies of 3D quantitative structure-permeation relationships suggest that iontophoresis is favoured by peptide hydrophilicity and hindered by voluminous, localized hydrophobicity (Schuetz et al. 2005, 2006). An especially interesting situation is presented when the bulky lipophilic moiety is directly adjacent to a positively charged residue as it is the case, for example, of nafarelin and leuprolide. It has been demonstrated that these two peptides reduce the magnitude of the electro-osmotic solvent flow typically observed in the anode-to-cathode direction at physiological pH and, in doing so, dramatically affect their own iontophoretic transport (Schuetz et al. 2006; Lau et al. 1994).
Insulin has attracted significant attention from researchers in the field of iontophoresis. It is generally accepted that hexameric insulin is too large for being delivered via iontophoresis; however, the flux of monomeric human insulin (mean MW ~6000 Da, negatively charged) was increased significantly by current application as compared to passive diffusion. Nevertheless, the iontophoretic administration of insulin is hindered by numerous challenges including the instability of the peptide. A considerable number of in vitro studies have investigated the effect of iontophoretic parameters on insulin delivery and have demonstrated the physiological effect of iontophoretically delivered insulin on blood glucose levels in different small animal models, particularly mice, rats and rabbits (Pillai et al. 2003, 2004a; b; Pillai and Panchagnula 2003a; b; Kari 1986; Liu et al. 1988; Rastogi and Singh 2005; Langkjaer et al. 1998). More recently, different approaches, combining iontophoresis with other techniques such as chemical enhancers, ultrasound, liposomes or microneedles, have been tested (Singh et al. 2012). While some of these studies have successfully delivered insulin in these small animal models, a delivery method which involves compromising the SC may not be appropriate for chronic use in humans. Further, it is uncertain whether the results can be extrapolated to humans, who will require significantly greater doses of the hormone to be delivered and provide the pharmacologic effect.
6.4 Iontophoresis Applications in Ocular Drug Delivery
The treatment of ocular diseases, particularly those afflicting the anterior and posterior camera of the eye, represents an important challenge. Drug delivery by topical formulations is typically ineffective due to both precorneal obstacles (drainage, blinking, tear film and induced lacrimation) and anatomical barriers (lipophilic corneal epithelium, hydrophilic corneal and scleral stroma and tight junctions) presented by the eye. It has been suggested that ocular iontophoresis may overcome some of the above-mentioned challenges, and therefore, the technique has been tested for the delivery of antibacterials, antivirals, antifungals, steroids, antimetabolites and genes.
Ocular iontophoresis can be classified into transcorneal and transscleral iontophoresis, according to treatment location. Transcorneal iontophoresis aims to deliver the drug to the anterior segment of the eye (cornea, aqueous humour, ciliary body, iris and lens) for the potential treatment of keratitis, glaucoma, dry eyes, corneal ulcers and ocular inflammation (Eljarrat-Binstock and Domb 2006).
Transscleral iontophoresis delivers the drug directly into the vitreous and retina. For this purpose, the iontophoretic device is placed on the conjunctiva, over the pars plana area, to avoid potential damage to the retina by current passage. Transscleral iontophoresis represents a potential alternative to intravitreal injections and to systemic administration in the treatment of endophthalmitis, uveitis, retinitis, optic nerve atrophy, paediatric retinoblastoma and age-related macular degeneration (Eljarrat-Binstock and Domb 2006).
Like the skin, the sclera has an isoelectric point between 3.5 and 4 and therefore a net negative charge at physiological pH; thus, transport by electro-osmosis will be possible depending on the pH. The key parameters controlling the iontophoretic transport of low molecular weight compounds are very similar to those controlling transdermal iontophoresis, i.e. the current intensity, duration of the application and composition of the iontophoretic vehicle (buffers and competing ions) (Gungor et al. 2010). On the other hand, the iontophoretic flux of some high molecular weight cationic compounds was not proportional to their concentration in the donor (Gungor et al. 2010). Similar observations were made in an in vitro study which delivered a marker, a hydrophilic 40-kDa dextran, across the isolated porcine sclera and across the trilayer sclera-choroid-Bruch’s membrane. It was observed that drug depots built up inside the sclera during in vitro iontophoresis and that this reservoir provides for a sustained transscleral flux for up to 3 h after iontophoresis (Pescina et al. 2011) (see Fig. 6.4).
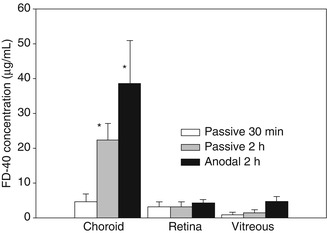

Fig. 6.4
Concentration of a dextran of 40 kDa (FD-40) in the choroid, retina and vitreous after its application onto excised eye bulb (no dynamic barrier). *Significantly different to the passive 30-min application (Redrawn from Pescina et al. 2011)
Although generally considered as a safe technique if properly implemented, some complications including epithelial oedema, decrease in endothelial cell inflammatory infiltration and burns can occur depending on the site of application, current density and duration. Because the corneal tissue and sclera present important differences, the passage of current affects them differently. For example, damage to the cornea surface affects vision, whereas the sclera is very sensitive to pain and hypoxia. In the case of humans, a transscleral exposure to 0–3.0 mA for 20 min and to 1.5 mA for 40 min was well tolerated, while passage of 4.0 mA for 20 min produced a burning sensation in half of the subjects treated (Parkinson et al. 2003).
Several studies have been conducted to demonstrate that iontophoresis can efficiently enhance the passage of various molecules across the eye membranes. Most of these studies have been performed in vivo in rabbits, while fewer in vitro studies and human trial are being reported.
The transcorneal and transscleral penetration of dexamethasone after short-term iontophoresis was investigated in rabbits, using drug-loaded disposable HEMA hydrogel sponges and a portable iontophoretic device. The drug levels measured in the cornea after a single transcorneal iontophoresis (1 mA for 1 min) were up to 30-fold higher than those obtained after repeated eye drop instillation. Further, high drug concentrations were measured in the retina and sclera 4 h post transscleral iontophoresis (Eljarrat-Binstock et al. 2005).
Antibiotics such as gentamicin, ciprofloxacin and vancomycin have been delivered with iontophoresis either transcorneally or transsclerally. A study in rabbits compared transcorneal iontophoresis (1 min, 1 mA) of gentamicin sulphate with topical eye drops of fortified gentamicin (1.4 %) applied every 5 min for 1 h and with subconjunctival injection of 0.25 mL of a 40-mg.mL-1 gentamicin solution. The peak gentamicin concentrations after a single iontophoresis treatment were 12–15 times higher than those obtained after gentamicin injection or after topical eye drop instillation; moreover, iontophoresis maintained the therapeutic drug levels in the cornea for more than 8 h (Eljarrat-Binstock et al. 2004). In another study, the concentration of ciprofloxacin in the aqueous humour just after 5 min of transcorneal iontophoresis was not significantly higher than in the no-current control. However, 6 and 12 h post-iontophoresis, the concentrations of drug in the aqueous humour were approximately six- and fivefold higher than in the control group, respectively (Vaka et al. 2008).
Methotrexate iontophoresis was investigated in rabbits using drug-loaded hydrogels mounted on a portable iontophoretic device. Following (1.6 and 5 mA.cm−2 for 4 min) the iontophoretic applications, the therapeutic drug levels were maintained for at least 8 h at the sclera and retina and for 2 h at the aqueous humour. Increasing the current density from 1.6 to 5 mA.cm−2 led to a twice-higher concentration at the vitreous and to 8 and 20 times higher concentrations at the retina and sclera, respectively (Eljarrat-Binstock et al. 2007).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree