The advent of acellular dermal matrices (ADMs) has changed the way implant reconstruction is performed and has opened the door to muscle-sparing techniques that can be performed as an NSM (nipple-sparing mastectomy) with DTI (direct to implant) with excellent cosmesis and less patient morbidity. The article discusses differences in types of ADMs and outlines some current strategies and controversies with ADM use.
Key points
- •
A well-performed nipple-sparing, or skin-sparing, mastectomy is the foundation of successful reconstruction.
- •
Techniques are evolving, and standard use of the pectoralis muscle and tissue expanders is in question.
- •
Acellular dermal matrix is both useful and cost-effective in performing immediate one-stage breast reconstruction.
Video content accompanies this article at http://www.plasticsurgery.theclinics.com .
Introduction
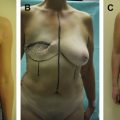
Early attempts at immediate breast reconstruction with immediate placement of breast implants were an exercise in futility. The breast envelope, ischemic after mastectomy, was unable to accommodate a device without subsequent complication and failure of the reconstruction. With the advent of tissue expanders for breast reconstruction, the skin envelope was allowed to slowly accommodate and revascularize, both due to support by healthy muscle and the lack of pressure from a partially deflated device. Tissue expansion over months was effectively a surgical delay and produced a reliable skin envelope that could be used to re-create the breast mound with an implant. Much has changed since those times as the anatomy of the breast skin is now better understood, and breast surgeons have devices to assess blood flow intraoperatively and medical devices like acellular dermal matrix (ADM) that allow one to avoid the use of tissue expanders altogether in primary cases of skin-sparing and nipple-sparing mastectomy (SSM, NSM).
Introduction
Early attempts at immediate breast reconstruction with immediate placement of breast implants were an exercise in futility. The breast envelope, ischemic after mastectomy, was unable to accommodate a device without subsequent complication and failure of the reconstruction. With the advent of tissue expanders for breast reconstruction, the skin envelope was allowed to slowly accommodate and revascularize, both due to support by healthy muscle and the lack of pressure from a partially deflated device. Tissue expansion over months was effectively a surgical delay and produced a reliable skin envelope that could be used to re-create the breast mound with an implant. Much has changed since those times as the anatomy of the breast skin is now better understood, and breast surgeons have devices to assess blood flow intraoperatively and medical devices like acellular dermal matrix (ADM) that allow one to avoid the use of tissue expanders altogether in primary cases of skin-sparing and nipple-sparing mastectomy (SSM, NSM).
Indications for the use of acellular dermal matrix
The clinical use of ADM has been the greatest advancement in breast reconstruction in the last decade. Indications for the use of ADM are expanding ( Box 1 ). ADM addresses directly the main problem that was early faced by early reconstructions, namely, excessive pressure on the overlying mastectomy skin. ADM directly supports the implant so the entire skin envelope with or without the nipple can be used for reconstruction without the negative consequence of internal pressure of the implant on the skin. In the submuscular position, the ADM can be thought of as an extension of the elevated pectoralis major muscle, an idea that resonates with surgeons already placing their devices under the muscle ( Fig. 1 ). Inferior pole support has been used as the main rationale for the use of ADM with tissue expander reconstruction, although this remains controversial. More recently, with improvement in the immediate vascularity of mastectomy flaps, ADM has been used as the sole support of the implant, leaving the pectoralis muscle alone. This “prepectoral” approach, although not the standard approach, resonates with patients and has many surgeon advocates.
- •
Skin-sparing or nipple-sparing mastectomy with immediate direct to implant reconstruction
- •
Treatment of animation deformity
- •
Treatment of malposition or symmastia
- •
Treatment/prevention of capsular contracture
ADM is also used by surgeons for the treatment of secondary problems with breast implant reconstruction, namely, animation deformity, capsular contracture, implant malposition, and symmastia. Although not the standard of care for these problems, ADM provides options in difficult cases, and more long-term studies are needed.
Indications for nipple-sparing mastectomy
ADM is necessary for immediate breast reconstruction with a direct-to-implant (DTI) approach. ADM addresses internal support of the prosthesis but presupposes that there is adequate skin for reconstructing the breast without excessive tension or the need for expansion. In the properly selected patient, the ideal reconstruction would use the existing skin envelope to cover the underlying volume replacement. Indications for an NSM approach continue to evolve and are outlined in Box 2 . Oncologic concerns will dictate the soundness of NSM in any individual patient (see Box 2 ). Large tumors (T3, >4 cm) and proximity of the tumor to the nipple (<2 cm) generally preclude an NSM approach and are good working guidelines until more definitive studies are done. Ultimately, the decision will be made in collaboration with the oncologic surgeon and their comfort level with the procedure. When these criteria are met and the oncologic surgeon is agreeable, there are other characteristics that make patients high risk for NSM ( Box 3 ). Large skin envelopes and excessive ptosis can challenge the limits of the often random and nonaxial blood supply often present after mastectomy. Previous incisions can similarly affect blood flow, and a history of breast reduction, lift, and augmentation all present an increased risk of ischemic complications. Previous breast lumpectomy and breast irradiation can also negatively impact healing as the radiation injury to the skin affects its ability to revascularize and promote healing. Finally, the most common reason that surgeons cite as to why they do not perform NSM with DTI is the inability of their oncologic surgeon to leave an adequate skin envelope that is not excessively thinned and ischemic. Surgeon lack of comfort with NSM is supported by studies that cite a skin necrosis rate with NSM as high as 40%. An NSM with DTI approach can still be considered in these high-risk cases, but a different technique, described in later discussion, should be considered.
- •
BRCA gene– positive or strong family history
- •
Small tumor size (<4 cm)
- •
Proximity to nipple >2 cm
- •
Well-vascularized skin envelope
- •
Large breast size (D cup or larger)
- •
Significant breast ptosis (grade 3)
- •
Previous lumpectomy and breast irradiation
- •
Previous breast reduction or mastopexy
- •
Previous breast augmentation
- •
Inexperienced surgical oncologist
- •
Active smoker
Types of acellular dermal matrix
ADM can have as its source human tissue or animal tissue, such as porcine or bovine origin ( Table 1 ). Within the United States, human products (DermACELL [LifeNet Health, Virgina Beach, VA], Alloderm [LifeCell, Bridgewater, NJ], FlexHD) are favored, and clinical data support its superiority. Outside the United States, due largely to regulations against the use of human tissue, porcine products are used predominantly with good results. Although these products are conceptually similar, decellularized dermis, the details of the decellularization process differ by product and therefore can act differently clinically. There are many head-to-head comparisons in the literature, but none are Level 1 (prospective, randomized, blinded) so they are open to criticism. The main issues when comparing products are the comparison of rates of infection, seroma, “red breast syndrome,” and rates of reconstructive failure. Although the actual process of decellularization for each product is proprietary, differences do exist in the speed of revascularization, the amount of retained proteins, and the degree of sterility of the final product. Some products have a sterility assurance level (SAL) of 10 −3 (Alloderm, FlexHD) or a one-in-a-thousand chance of contamination, while others have an SAL of 10 −6 (DermACELL, Allomax) or a one-in-a-million chance of contamination (see Table 1 ). Nonbiologic implantable medical devices (ie, breast implants, expanders) are required to have an SAL of 10 −6 .
| ADM | Source | Aseptic/Sterile |
|---|---|---|
| AlloDerm (LifeCell Corp, Branchburg, NJ, USA) | Human | Aseptic |
| AlloDerm RTU (LifeCell Corp, Branchburg, NJ, USA) | Human | Sterile (SAL 10 −3 ) |
| AlloMax (Davol Inc, Murray Hill, NJ, USA) | Human | Sterile (SAL 10 −6 ) |
| FlexHD (Ethicon Inc, Somerville, NJ, USA) | Human | Aseptic |
| DermaMatrix (MTF/Synthes CMF, West Chester, PA, USA) | Human | Sterile (SAL 10 −6 ) |
| DermACELL (LifeNet Health, Virginia Beach, VA, USA) | Human | Sterile (SAL 10 −6 ) |
| Neoform (Mentor, Santa Barbara, CA, USA) | Human | Sterile (SAL 10 −6 ) |
| Strattice (LifeCell Corp, Branchburg, NJ, USA) | Porcine | Sterile (SAL 10 −3 ) |
| Permacol (Covidien, Boulder, CO, USA) | Porcine | Sterile (SAL 10 −6 ) |
| SurgiMend PRS (TEI Biosciences Inc, Boston, MA, USA) | Bovine | Sterile (SAL 10 −6 ) |
It is unclear why infection rates of identical product differ so much in the hands of different surgeons in different centers, but it suggests that factors other than the ADM are at play (ischemic skin flaps, long operative times, poor sterile technique, and so forth). Red breast syndrome or RBS appears to be an immune-mediated process that creates a redness overlying the ADM that has no other harbinger of infection (elevated white blood cell count, fever, pain). Accordingly, RBS does not respond to antibiotic therapy, persists for weeks, and eventually resolves without further treatment. Aspiration of seromas associated with RBS is recommended, and cultures should be sent to rule out atypical infection (ie, mycobacteria). RBS is particularly associated with the use of certain ADMs (ie, Alloderm) and is not universally experienced by all surgeons.
Controversies with acellular dermal matrix
Although some surgeons use ADM in all of their procedures, it is still not the standard of care. Without widespread adoption, many controversies can be found in the literature regarding its use, and these are worth discussing. As with any new product or technique, more long-term studies of high quality are awaited to understand their true incidence or causality.
Many earlier studies describing the use of ADM in comparison to the standard approach of no ADM describe an increase in overall complications, especially infection, seroma, and loss of reconstruction. As surgeons have become more experienced, rates of these complications have been minimized, to a point where use of ADM is no longer considered a risk factor in experienced hands. One can certainly postulate that better skin flaps, decreased operative time, and improved sterile operative techniques can be cited for this improvement but this is speculative.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree