Note: Page numbers of article titles are in boldface type.
A
Acne scars, injectable filler techniques for, 488
LIS for, 434, 436
PMMA for, 437, 439–440, 444
atrophic, 437, 442–443
injection procedure and technique for, 443
needle advancement for, 443
patient selection and consultation for, 442
photography for, 442
pivotal study of, 441–442
skin testing for, 442–443
ADATO Sil-Ol-5000 silicone oil, 434, 437
Adverse effects/events, of collagen stimulators, 465, 467–468
of fillers, 447–458
for jawline augmentation, 493–494
for midface and malar augmentation, 492
for temporal augmentation and rejuvenation, 490–491
granulomas as, 450–452
infection as, 449–450
introduction to, 447–448
key points of, 447
management of common, 451
poor cosmetic results as, 448–450
presentation of common, 451
prevention of, 425, 453–454
vascular occlusion as, 451–454
with HA filler, 430
with illegal fillers, 454–456
with synthetic injectables, 436–437, 440–443
Aesthetic outcomes, of filler injections, for jawline augmentation, planned, 493
for lower face enhancement, 474
for midface and malar augmentation, 493
planned, 492
for temporal augmentation and rejuvenation, 491–492
planned, 489
of fillers, facial feature changes as, 481
poor, 448–450
responsibility for, 476
Aesthetics companies, battle for provider attention, 419–420
Age/aging, facial, as three-dimensional process, 459, 461–463, 471, 479
cheek complex in, 484–485
hyaluronic acid distribution and, 424, 427
lip restoration for, 483
skin laxity and, 481
Allergic response, to HA fillers, 424–426, 430
American Society of Plastic Surgeons (ASPS), soft tissue filler statistics of, 459, 463
Angular artery, as vascular occlusion danger zone, with fillers, 452, 454
Animal sources, of hyaluronic acid, 425–426
Anterograde arterial occlusion, as filler complication, 451–454
Anteromedial cheek region, in midface and malar augmentation, 491–492
Anticoagulants, filler complications related to, 448–449
Antioxidants, for HA filler degradation prevention, 425
Antiviral activity, of hyaluronic acid, 424, 430
Areas of face. See Facial areas.
Arnica montana, for minimizing bruising, with fillers, 449
Artefill, as filler, 418, 433, 437. See also Bellafill .
Arterial occlusions, as filler complication, 451–454
Asymmetries, of NLF, injectable filler technique for, 481–482
Atrophic acne scars, PMMA for, 437, 442–443
Augmentation, facial. See Soft tissue augmentation; specific anatomy or procedure.
hand, collagen stimulators for, 460, 463–465
injectable filler techniques for, 487–488
Autoimmune diseases, hyaluronic acid production and, 424
B
Bacterial infections, fillers and, 449–450
Bellafill, as injectable filler, 433, 437–443, 448
discussion on, 443
for acne scars, 437, 439–442
atrophic, 437, 442–443
for nasolabial folds, 437–441
mechanism of action, 437
overview of, 437, 443–444
product information on, 437–440
Belotero, as HA filler, 425–426, 451
Biochemistry, of HA fillers, 424
Biocompatibility, bioengineering definition of, 460–461, 480
Bioengineering, with collagen stimulators, 459–461
Biofilms, filler complications related to, 450, 454, 480
BioForm, in filler market, injector confidence with, 420
regulation of, 418
Biomechanical properties, of HA fillers, 429–430
Biomedical applications, early, of HA fillers, 423–424
Blindness, as filler complication, 451–453
Bone remodeling/resorption, facial, with aging, 461, 463, 471, 483
Botulinum toxin type A, for forehead and brow enhancement, 472–473
Brow enhancement, customized, 471–473
injectable filler techniques for, 487
Bruising. See Ecchymosis .
B
Bacterial infections, fillers and, 449–450
Bellafill, as injectable filler, 433, 437–443, 448
discussion on, 443
for acne scars, 437, 439–442
atrophic, 437, 442–443
for nasolabial folds, 437–441
mechanism of action, 437
overview of, 437, 443–444
product information on, 437–440
Belotero, as HA filler, 425–426, 451
Biochemistry, of HA fillers, 424
Biocompatibility, bioengineering definition of, 460–461, 480
Bioengineering, with collagen stimulators, 459–461
Biofilms, filler complications related to, 450, 454, 480
BioForm, in filler market, injector confidence with, 420
regulation of, 418
Biomechanical properties, of HA fillers, 429–430
Biomedical applications, early, of HA fillers, 423–424
Blindness, as filler complication, 451–453
Bone remodeling/resorption, facial, with aging, 461, 463, 471, 483
Botulinum toxin type A, for forehead and brow enhancement, 472–473
Brow enhancement, customized, 471–473
injectable filler techniques for, 487
Bruising. See Ecchymosis .
C
Calcium hydroxyl apatite (CaHA), as collagen stimulator, 459–469 . See also Radiesse .
biocompatibility components of, 460–461
choice factors of, 480
for neck, chest, and hand rejuvenation, 487–488
for NLF correction, 482
introduction to, 459–461
key points of, 459
mechanism of action, 460–461, 480
patient positioning for, 461–462
placement precautions for, 461
postprocedural care for, 468
potential complications of, 465, 467–468
preoperative planning and preparation for, 461–463
procedural approach to, 462–468
for cheek and chin augmentation, 462–465, 476, 485
PLLA vs., 465–468
rehabilitation and recovery for, 468
summary overview of, 469
Cannulas, for collagen stimulators placement, 463–464, 468
for filler placement, 454
in forehead and brow enhancement, 472, 486, 488
in nasolabial folds, 449
Captique, as HA filler, 426–427
Carcinomas, hyaluronic acid production and, 424
Cheek augmentation, collagen stimulators for, caHA as, 462–465
PLLA vs., 465–468
customized, 476
fat compartment aging and, 461–462
injectable filler techniques for, 484–485
skeleton aging and, 461, 463, 471
skin redundancy and, 482
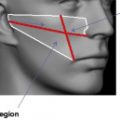
Cheek subregions, in midface and malar augmentation, 485, 491–492
Chest rejuvenation, collagen stimulators for, 465
injectable filler techniques for, 487–488
Chin augmentation, collagen stimulators for, caHA as, 462–465
PLLA vs., 465–468
customized, 473–474, 476
fat compartment aging and, 461–462
skeleton aging and, 461, 463, 471
Classification, of injectable fillers, 434
Clinical data, on LIS, for facial soft tissue augmentation, 435–436
on PMMA, for acne scars, 441–442
for NLFs, 439–441
Clinical outcomes. See Aesthetic outcomes.
Clinical trials, of fillers, 418
synthetic injectables as, 433–434, 437, 441–442
Collagen, HA fillers effect on, 424, 427–429
in filler market, evolution of, 417
HA fillers vs., 425
regulation of, 418
Collagen stimulators, 459–469
biocompatibility components of, 460–461
introduction to, 459–461
key points of, 459
mechanism of action, 460–461
patient positioning for, 461–462
placement precautions for, 461
postprocedural care for, 468
potential complications of, 465, 467–468
preoperative planning and preparation for, 461–463
procedural approach to, 462–468
PLLA in, 465–468
Radiesse for cheek and chin augmentation, 462–465
rehabilitation and recovery for, 468
summary overview of, 469
Complications, of fillers, 447–458 . See also Adverse effects/events.
Confidence, of physicians, with filler injections, 420, 487
Consolidation, of filler market, 420
Consultations, on fillers, 419–420
for acne scars, 442
Contour depressions, traumatic, injectable filler techniques for, 488
Cosmederm, in filler market evolution, 417
Cosmetic fillers. See Fillers.
Cosmoplast, in filler market evolution, 417
Cost of research, on fillers, 418
Cross-hatching technique, for décolleté rejuvenation, 465
for jawline correction, 486
for lower eyelid correction, 487
for marionette line correction, 482–483
for NLF correction, 482
for prejowl sulcus correction, 484
for submalar hollowing, 485
Cross-linking agents, for HA fillers, 425–426, 480
Cross-linking density, of HA fillers, 425–427
Cupid’s bow, in lip restoration, 483
in lower face enhancement, 474–476
D
Décolleté rejuvenation, collagen stimulators for, 465
injectable filler techniques for, 487–488
Degradation, of HA filler, prevention of, 425
Depot injections, of collagen stimulators, 463–465
Dermal fillers, other, HA fillers vs., 425, 427
Dermal Gel Extra, as HA filler, 425–426
Differential effect claims, of fillers, 418
Dorsal nasal artery, as vascular occlusion danger zone, with fillers, 452, 454
E
Ecchymosis, as filler complication, 448–449
HA fillers and, 430
in eyelid correction, 487
in temporal augmentation and rejuvenation, 491
LIS and, 436–437
livido vs., 451, 453
minimizing, 449
Edema, as filler complication, 448–449
Elastic storage shear modulus (G’), collagen stimulators and, 460
HA fillers and, 425–426, 429–430, 473
Elevess, as HA filler, 426
Embolia cutis medicamentosa, 451
Embolization, as filler complication, 451–455, 486
Energy devices, fillers combined with, 421, 479
Enhancement, facial, customized approach to, 471–477 . See also Facial enhancement.
Enhancement claims, of fillers, 418–419
European Union (EU), filler market regulation in, 418
Excision, surgical, of silicone nodules, 455–456
Extracellular matrix, HA naturally found in, 424
Eyelid correction, injectable filler techniques for, lower, 486–487
upper, 487
F
Facelift, liquid, 481
Facial areas, new, for injectable fillers, 421
subregions of, in cheek and chin augmentation, 461–463, 465, 484
in midface and malar augmentation, 484–485, 491–492
thirds, enhancement approaches to, 471
lower, 473–476
midface, 471
top, 471–473
Facial enhancement, customized approach to, 471–477 . See also specific anatomy or procedure.
introduction to, 471
key points of, 471
of lower face, 473–476
of midface, 473
of top third of face, 471–473
summary overview of, 476
Facial fat, aging changes of, 461–462
customized enhancement of, 472–473, 481
Facial features, changing with fillers, 481
Facial fillers. See Fillers.
Facial muscles, toxin injection points and, 471–473
Facial rejuvenation, collagen stimulators for, 459–469 . See also Collagen stimulators.
customized approach to, 471–477 . See also Facial enhancement.
filler injections for, 481. See also Injectable fillers.
temporal, 489–492. See also Temporal rejuvenation.
Facial skeleton, aging changes of, 461, 463, 471
Fanning technique, for marionette line correction, 482–483
for NLF correction, 482
for prejowl sulcus correction, 484
for submalar hollowing, 485
Fat compartments/pads, facial, aging changes of, 461–462
cheek complex in, 484–485
customized enhancement of, 472–473, 481
skin laxity and, 481
Fat injections, as filler, 453
FDA. See Food and Drug Administration (FDA), US.
Fibroblasts, granulomas and, 450
HA fillers effect on, 424, 427–429
Fibroplasia, PLLA and, 460
Filler injection. See also Injectable fillers.
case for, 433–445
current concepts in, 489–494
industry perspectives on, 420–421
Filler market, evolution of, 417–419
in US vs.. EU, 418–419
injectables in. See Injectable fillers.
Fillers, choice of, 419–420, 480–481
collagen stimulators vs., 459–469 . See also Collagen stimulators.
complications of, 447–458
granulomas as, 450–452
infection as, 449–450
introduction to, 447–448
key points of, 447
poor cosmetic results as, 448–450
prevention of, 425, 453–454
vascular occlusion as, 451–454
with HA filler, 430
with illegal fillers, 454–456
with synthetic injectables, 436–437, 440–443
hyaluronic acid as, 423–432 . See also Hyaluronic acid (HA) fillers.
ideal characteristics of, 424, 429, 437
illegal, 434
complications of, 454–456
industry perspectives on, 417–421
battle for attention to, office staff touch points in, 419–420
physician touch points in, 419–420
reception area as first impression, 419
consolidation of, 420
future developments in, 420–421
injectors/injection in, new areas for, 421
new materials for, 421
new ways of, 420–421
provider confidence for, 420, 487
key points of, 417
market evolution of, 417–419, 433
patient selection influences in, 420
office staff as, 419–420
perspectives of, 480–481
regulatory drivers of, 418–419
summary overview of, 421
US vs.. EU markets in, 418–419
injectables as. See also Injectable fillers.
case for, 433–445
current concepts in, 489–494
industry perspectives on, 420–421
techniques for, 479–488
mechanical properties of, testing for, 429
reversible, 421, 427, 447
Focal points, midface, in midface and malar augmentation, with filler injection, 491–492
Food and Drug Administration (FDA), US, approval of fillers, 447, 480–481
collagen stimulators as, 459–460
facial subregions defined by, 491–492, 494
HA as, 423–424, 430
synthetic injectables as, 433–434, 437, 439
filler market regulation by, 418, 433
Forehead enhancement, customized, 471–473
Foreign body granuloma, as filler complication, 450–451
Free radicals, HA filler degradation by, 425
Freudenthal-Nicolau syndrome, 451
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree