Introduction
More than 89 upper extremities and 30 face transplants have been performed worldwide since 1998, proving that vascularized composite allotransplantation (VCA) is technically feasible and guarantees long-term survival under currently available immunosuppressive regimens.
VCA is not a lifesaving procedure, thus minimization or withdrawal of the systemic immunosuppression is imperative to avoid the long-term deleterious complications related to the chronic use of the immunosuppressive treatment. When compared with solid organ transplants, vascularized composite allografts contain a histologically heterogeneous array of different tissue types (skin, muscle, bone, bone marrow, lymph nodes, nerve, and tendon) that present different immunogenicity. Experimental models of limb transplantation showed that skin is one of the most antigenic tissues, and when combined with muscle, subcutaneous tissues, and bone (including bone marrow), can generate a high immunologic response. The vascularized muscle component of the limb allografts was shown to be less antigenic than skin ; whereas bone presents lower immunogenicity; and cartilage, tendon, and vessels are the least antigenic tissues.
In this chapter the authors discuss the basics of transplantation immunology, the mechanisms of allograft acceptance, and rejection as pertinent to vascularized composite allografts. An overview of the tolerance mechanisms and strategies developed to achieve tolerance is presented, as well as a short summary of the novel cell-based therapies, and their role in induction of transplantation tolerance. Considering the increasing number of vascularized composite allografts performed worldwide, as well as their applicability to the complicated cases of reconstructive surgery, it seems imperative that plastic surgeons should become familiar with these techniques and the related immunologic concepts, in order to select the most appropriate candidates and to properly inform and consent patients for these novel procedures of reconstructive transplantation.
Basics of Transplantation Immunology
The Role of the MHC/HLA Antigens
The major histocompatibility complex (MHC) genes code the strongest transplant antigens. Human MHC molecules called human leukocyte antigens (HLAs) are codified by a group of genes located on the short arm of chromosome 6. Each parent provides a haplotype (a linked set of MHC genes) to each offspring in the co-dominant inheritance. There are two classes of the MHC or HLA molecules. MHC class I molecules (HLA-A, HLA-B, and HLA-C) are expressed on all nucleated cells and generally present endogenous antigens, such as viruses and self-protein fragments to CD8 + T cells. Class II molecules (HLA-DP, HLA-DQ, and HLA-DR) are constitutively expressed only on professional antigen-presenting cells (APCs), including dendritic cells, macrophages, and B-cells. Their expression may be upregulated on epithelial and vascular endothelial cells after exposure to proinflammatory cytokines. Class II molecules present antigens derived from the extracellular proteins to the CD4 + T-cells. The degree of HLA mismatch between the donor and the recipient plays a role in determining the risk of chronic rejection and graft loss in solid organ transplantation. HLA-A, -B, and -DR (three pairs, six antigens) are traditionally used for typing and matching before kidney or pancreas transplantation. The HLA-Cw, -DP, and -DQ typing is currently used by many transplant centers. For kidney transplants, the long-term graft survival is best in HLA-identical living related kidney transplants. The major effect is attributed to the match of the DR antigen, and the order of importance for HLA match in kidney transplants is HLA-DR > HLA-B > HLA-A, respectively.
Data from the International Hand and Composite Tissue Transplantation Registry reveal that high mismatch rates for the hand transplantation cases were recorded in 50% of the recipients (for all or five out of six antigens) and in 57% of face transplant recipients.
Non-HLA Antigens
Acute and chronic graft rejection can occur in HLA-identical sibling transplants, indicating presence of the immune response to non-HLA antigens. Several non-HLA related antigens and antibodies derived from either the alloimmune or autoimmune responses have been reported.
ABO Blood Group Antigens
ABO blood group antigens are expressed on the red blood cells, on vascular endothelial cells, and other cells. ABO incompatibility triggers hyperacute rejection of the organ due to the presence of the preformed hemagglutinin A and/or B antibodies. Desensitization protocols to remove the preformed hemagglutinins from the recipient’s circulation have been used for the ABO incompatible transplants. The rhesus factor (Rh) and other red cell antigens are not relevant to the organ transplants, since they are not expressed on the vascular endothelium.
Minor Histocompatibility Antigens
Minor histocompatibility antigens (MiHAs) are small endogenous peptides that occupy the antigen-binding site of the donor MHC molecules. They are generally recognized by CD8 + cytotoxic T-cells, leading to graft rejection. MiHA play an important role in pathogenesis of the graft-versus-host disease in patients who have received an HLA-matched bone marrow transplants. H-Y MiHA is encoded by the Y chromosome in males and can induce alloimmune response when a male organ is transplanted into a female recipient. MHC class I related chain A and B (MICA and MICB) are also expressed on the endothelial cells. Antibodies against MICA and/or MICB can cause antibody-mediated rejection (AMR) and graft loss. Some minor transplant antigens may be derived from the mitochondrial proteins and enzymes.
Allorecognition Pathways
Allorecognition can occur by one of the three pathways: direct, indirect, and semi-direct. In the direct pathway , recipient’s T-cells recognize the intact allogeneic HLAs expressed by the donor cells. In the indirect pathway , T-cells recognize peptides derived from the donor HLAs presented by the recipient APC. In the semi-direct pathway, the recipient dendritic cells or other APCs, acquire the intact HLAs from the donor cells and present them to the recipient T cells. The direct and indirect pathways are well recognized in organ transplantation, whereas the semi-direct pathway is not of the clinical importance.
The direct pathway is very important in the immediate post-transplant period. Without appropriate immunosuppression, a strong and effective alloresponse would be initiated, primarily due to the high number of the recipient T-cells that will recognize the graft antigens and will cause the acute cellular rejection. While the indirect pathway of allorecognition may also participate in the acute rejection, it is predominantly observed in the late onset of rejection, as well as in the chronic rejection. As long as the allograft is present within the host, the recipient’s APCs can pick up the alloantigens, shed them from the graft, and start the alloimmune response. Therefore, lifelong maintenance immunosuppression is required in order to prevent both the acute as well as chronic allograft rejection.
T-Cell Activation
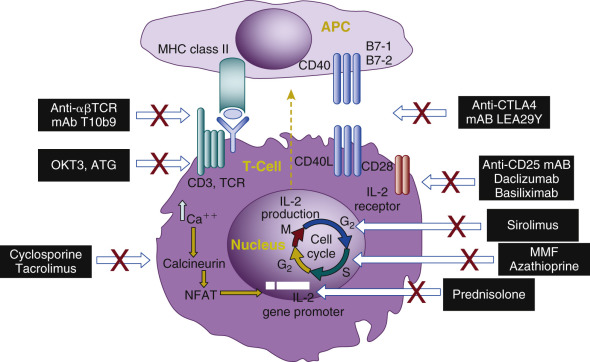
T-cell activation is the key process of allograft rejection ( Fig. 67.1 ). T cells recognize the alloantigens through T-cell receptors (TCR). The initiation of the intracellular signaling requires additional peptides known as CD3 complex, and the antigen-specific signal (signal 1) is transduced through the TCR-CD3 complex. Two signals are needed for complete T-cell activation. The second co-stimulatory signal depends on the receptor-ligand interactions between T cells and the APCs (signal 2). Numerous co-stimulatory pathways have been described, and blockage of these pathways can lead to antigen-specific inactivation or T cell death. The best-studied co-stimulatory signals occur through the CD28-B7 and CD154-CD40 pathways. CD28 and CD154 are expressed on the T cells, and their ligands B7 and CD40 are expressed on the APCs. T cells also express cytotoxic T lymphocyte-associated antigen-4 (CTLA-4), which is homologous to the CD28 and has a higher affinity than CD28 to bind B7. However, when CTLA-4 binds B7, it produces an inhibitory signal to terminate the T-cell response. This unique interaction leads to the clinical development of a fusion protein CTLA-4-Ig (belatacept) as a new immunosuppressive medication.
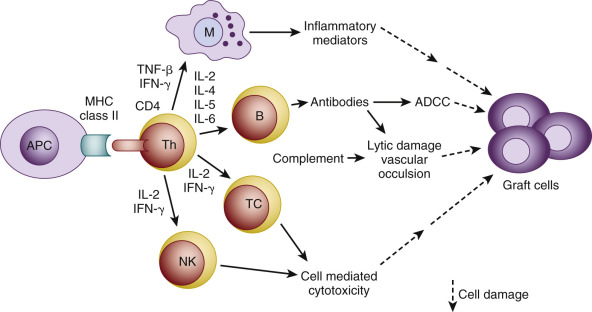
The combination of signals 1 and 2 activates three downstream signal transduction pathways: the calcium-calcineurin pathway, the RAS-mitogen activated protein kinase pathway, and the IKK-nuclear factor κB (NF-κB) pathway. These three pathways further activate transcription factors, including the nuclear factor of the activated T cells, activated protein-1, and the NF-κB, respectively. Several new molecules and cytokines including CD25, CD154, interleukin-2 (IL-2), and IL-15 are subsequently expressed. IL-2 and IL-15 deliver growth signals (signal 3) through the mammalian target of the rapamycin pathway and phosphoinositide-3-kinase pathway, which subsequently trigger the T-cell cycle and proliferation. The fully activated T cells undergo clonal expansion and produce a large number of cytokines and effector T cells, which eventually produce CD8 + T-cell mediated cytotoxicity, help macrophage-induced delayed type hypersensitivity response (CD4 + Th1), and help B cells for antibody production (CD4 + Th2). A subset of activated T cells becomes the alloantigen-specific memory T cells. Current immunosuppressive medications act, blocking different steps of T-cell activation ( Fig. 67.2 ).
B-Cell Activation
B cells express antigen-specific receptors as immunoglobulins on their surfaces. When these receptors bind donor HLA antigens in the presence of helper T cells (CD4 + Th2), B cells are activated. They divide, differentiate, and become plasma cells to secrete antibodies. Some activated B cells become memory B cells. The helper T cells may facilitate B-cell activation either through the intimate membrane contact involving a variety of receptors and ligands (such as CD40:CD154) or through the secreted soluble cytokines (such as IL-4). These HLA antibodies bind antigens and can cause graft injury either by activating the complement cascade (complement-dependent cytotoxicity) or via the Fc receptor on natural killer (NK) cells, neutrophils, and eosinophils (antibody-dependent cellular cytotoxicity). In addition to producing antibodies, B cells are also the APCs. B cells can present allograft-derived antigens to T cells for T-cell activation through the indirect pathway of allorecognition.
Rejection
The rejection process is an inflammatory process developed by a specific immunologic response to the allograft antigens. Within the first post-transplant year, 85% of hand and face recipients experienced at least one episode of acute rejection. Rejection of vascularized composite allografts is assumed to be mediated by mechanisms similar to those observed in solid organ transplantation. Based on our understanding of the rejection process in the solid organ transplants, brief descriptions of the various mechanisms that underlie the rejection process in VCA are presented below.
Hyperacute Rejection
Hyperacute rejection develops very rapidly within a few minutes after transplantation and represents the classic example of the antibody-mediated rejection process associated with the preexisting antibodies against the MHC of donor cells. Anti-MHC antibodies may be present in the recipient’s circulation due to prior blood transfusions, rejection of the previous transplants, or multiple pregnancies. In addition, the presence of antibodies against the ABO blood group system can induce hyperacute rejection. Most of the damage that occurs during the hyperacute rejection process affects the endothelial cells of capillary vessels and small arterioles. Immunologic complexes on the vessel endothelial cells activate the complement and coagulation cascade, which leads to damage of the vascular endothelium and ultimately to the graft dysfunction. Hyperacute rejection has never been observed in VCA.
Accelerated Rejection
Development of accelerated rejection takes place between 24 h and 5 days after transplantation. The increased risk of developing an accelerated phase of rejection arises from sensitization of the allograft recipient, for example, by a previous allograft. Accelerated rejection is associated with the interstitial hemorrhages, vascular thrombosis, and leukocyte infiltrations. Graft destruction is accompanied by extensive endothelial deposition of humoral mediators IgM, IgG, C3, fibrin and leukocyte infiltrations, as well as by alloantibody production. Pro-inflammatory cytokines such as IFN-γ and IL-6 are present within the graft and contribute to the graft damage. Accelerated rejection is considered a classic, humoral mediated response; it precedes T-cell-dependent mechanisms.
Acute Rejection
Acute rejection occurs within a few days to a few weeks after transplantation. Acute humoral rejection is clinically determined by the following factors: morphologic evidence of tissue injury; the presence of complement factor 4 (C4) deposition as the immunopathologic evidence of the antibody-mediated action; and serologic evidence of circulating antibodies to the donor human histocompatibility antigens (HLA) or other endothelial antigens. This process may be related to changes in the graft arteries or parenchymal cells. During acute rejection, fibrinoid necrosis of arteries and aggregation of the platelets and fibrin in the lumen of the affected vessels leads to vascular obstruction. In addition, leakage and focal tissue necrosis occur, and inflammatory cells accumulate in the interstitium of the graft. Acute rejection is primarily a cell-mediated process and arises from the activation of T cells and the subsequent triggering of the various effector mechanisms.
Components of acute composite allograft rejection include vasculitis, dermatitis, myositis, and perineural involvement – confirmed through the skin biopsy specimens taken from the human hand and face transplant recipients and from the skin biopsies provided by the multicenter hand and abdominal wall transplant cases. However, severe acute rejection in solid organ allograft recipients has been attributed to the humoral mechanism of the alloantibody-mediated syndrome, as a consequence of alloantibody production against donor HLA. Presence of the donor-specific antibodies has been documented in recipients of the upper extremity transplants and although their significance has not been completely elucidated, there have been instances where a clear link with the acute rejection has been shown.
Chronic Allograft Vasculopathy (Chronic Rejection)
Chronic allograft destruction is a slow process that occurs over months and/or years and presents common characteristics in all solid organ transplants. The frequency of chronic allograft vasculopathy correlates with the acute rejection episodes, recipient sensitization, insufficient MHC-antigen matching, recipient age and race, inadequate immunosuppression, hypertension, cytomegalovirus (CMV) infection/reactivation, prolonged ischemia time of the graft before transplantation, smoking, and hyperlipidemia.
Kaufman et al. reported graft vasculopathy in two out of nine hand transplant patients, with one case requiring plasmapheresis and one resulting in graft loss. Some level of vasculopathy was present in all of these patients, despite normal conventional vascular imaging and unremarkable histology of the skin biopsies. To date, no evidence of chronic rejection has been reported in face transplant patients.
Based on the similarity of histopathologic lesions observed in the hand transplant recipients when compared with the histologic specimens of the solid organ transplants, the mechanism of chronic rejection in the VCA transplants can be elucidated.
Immunologic Factors of Chronic Rejection
The hallmark of chronic graft rejection is attributed to the endothelial injury of the affected vessels. Initial vascular injury initiates an inflammatory cascade, which in turn leads to the allograft arteriosclerosis and graft fibrosis. The cardinal feature is represented by the luminal obliteration. Blockage occurs due to the proliferation of the smooth muscle cells that have migrated from the vessel wall and deposited matrix proteins. Ultimately, interstitial fibrosis leads to allograft dysfunction ( Fig. 67.3 ). Many studies emphasize that both direct and indirect allorecognition of donor antigens promotes development of transplant vasculopathy.
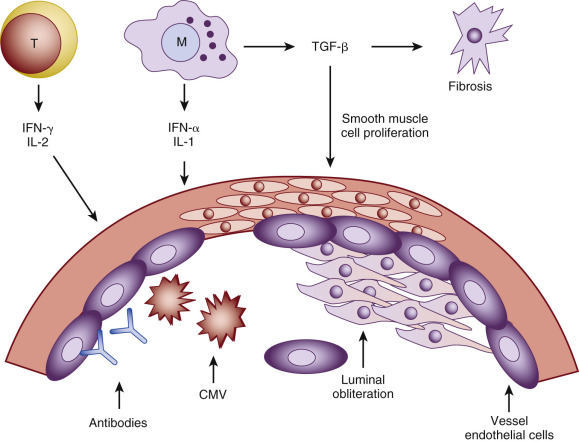
Non-Immunologic Factors of Chronic Rejection
Brain death involves a syndrome that includes rapid swings in blood pressure, hypotension, coagulopathy, pulmonary changes, hypothermia, and electrolyte abnormalities. Organs procured from the living donors are superior to those of brain dead donors, suggesting that brain death can influence graft quality. Animal models reveal that after brain death, the levels of catecholamines and cytokines increase and MHC class I and II antigens are upregulated, implying that the immunogenic response in the peripheral organs is increased.
A second non-immunologic factor that may increase the immunologic response is the ischemia-reperfusion injury. Ischemia-reperfusion may contribute not only to the early delayed graft function but also to the late allograft dysfunction, and it may be a risk factor for chronic allograft vasculopathy. Total allograft ischemia time includes the transient warm ischemia time before or during allograft removal from the donor, the cold ischemia time associated with organ preservation, and ischemia time during the period of organ revascularization. The factors associated with organ removal, storage, and engraftment may increase the immunogenicity of allografts by upregulation of the MHC antigens and activation of the cytokine cascade, which in turn leads to the leukocyte infiltration and ultimately to the obliterative vasculopathy and fibrosis.
Tolerance
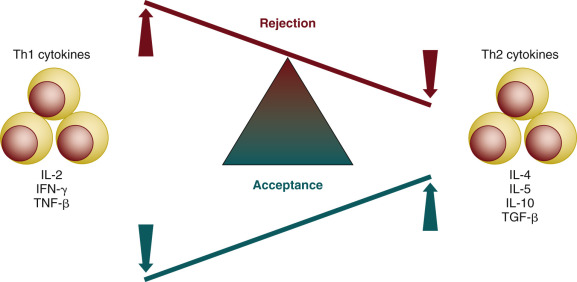
Immunologic self-tolerance is a stable state in which the immune system does not react destructively against self-antigens. To maintain self-tolerance and to defend against pathogens, a highly balanced mechanism has evolved for generation of the antigen-specific effector cell responses and the antigen-specific tolerance induction ( Fig. 67.4 ). Lack or loss of self-tolerance is likely to result in the autoimmune responses and cellular and tissue damage, and in consequence can lead to the clinical onset of the autoimmune disease. Interactions between the antigen-presenting cells and the lymphocytes are critical for self-tolerance, and these are known to take place in the thymus (central tolerance) and peripheral lymphoid tissues (peripheral tolerance).
In the thymus, the developing lymphocytes with no marked reactivity against the self-peptides are positively selected in the thymic cortex and enter circulation as the mature lymphocytes. In contrast, developing lymphocytes with the marked reactivity against self-peptides undergo negative selection (clonal deletion) in the thymic medulla.
Some potentially self-reactive T lymphocytes may escape from the thymus into the periphery. Peripheral tolerance mechanisms are operative in the extra-thymic lymphoid tissues and include elimination of the self-reactive T cells, anergy, antigen ignorance, apoptosis, exhaustion, and active suppression by the regulatory T cells. Anergy is the state in which T cells are alive but unable to respond to the antigen stimulation. Anergy may be induced by the lack of co-stimulatory signals and, in consequence, this process may lead to apoptosis. Ignorance may occur when the donor antigens (e.g., from the graft) are not delivered to the recipient lymphoid system or when the recipient lymphocytes are unable to recognize the donor antigens. For true antigen-specific tolerance, the immune system must not respond to the allograft antigens, but maintain its ability to respond to other antigens, including viruses and bacteria. Although the main goal in organ transplantation is to develop complete stable tolerance to prevent immunosuppression-related side effects, this goal is difficult to achieve in all organ recipients because of the heterogeneity of the donor-recipient combinations, the immune status, and the severity of underlying disease. Currently, the definition of tolerance (normal graft function, normal histology, and withdrawal of immunosuppression) is incomplete and represents a challenge in clinical practice.
A new concept of “prope” (almost) tolerance was first introduced by Calne et al. and is defined as the donor-specific hyporesponsiveness under a low dose of immunosuppressive therapy. A concept of prope tolerance leads to reduced immunosuppression-related side effects and allows for improved allograft survival without episodes of acute or chronic rejection.
Sachs stated that the simple definition of transplantation tolerance – graft acceptance in the absence of exogenous immunosuppression – is not sufficient in the clinical practice because a downregulatory response (not merely the absence of a response) is currently considered a tolerant state, regardless of the mechanism. This partial tolerant state with a minimal baseline nontoxic dose of maintenance therapy is suggested to be clinically more appropriate.
Chimerism and Donor Bone Marrow Transplantation
The persistence of donor-origin cells within the recipient compartment is known as chimerism. Transplanted organs contain large numbers of cells of bone marrow origin known as the passenger leukocytes. These cells may migrate from the transplanted tissues and colonize the lymphoid and non-lymphoid organs of the recipients. The load of the donor leukocytes can be increased in the recipients by infusion of the donor bone marrow cells. Donor hematopoietic cells can develop two types of macrochimerism: full chimerism when the recipient immune system is destroyed by myeloablation and replaced fully (100% donor cells) by the donor hematopoietic cells, or mixed chimerism , which is induced after non-myeloablative host conditioning when donor and recipient hematopoietic cells coexist with the recipient (donor cells >1% and <100%). Macrochimerism can be assessed by flow cytometry and monoclonal antibodies specific for the donor MHC class I and class II. The third type of chimerism is defined as microchimerism (donor-derived cells <1%) and usually occurs spontaneously after organ transplantation.
Early experimental protocols in mice, relied on the myeloablative conditioning and complete reconstitution with donor hematopoietic stem cells in order to achieve engraftment and chimerism ; however, such protocols carry significant morbidity and a risk of impaired immunocompetence, which would not be considered acceptable outside the field of hematological malignancy. The development of non-myeloablative conditioning regimens leading to the engraftment and mixed chimerism with significantly reduced morbidity, represents a key step in the search for clinically relevant tolerance induction regimens and have been demonstrated in the small and large animal models, as well as in the clinical protocols.
Interestingly, the establishment of stable, mixed chimerism may not be required for the rejection-free, long-term survival of various organs and tissues. In both the preclinical and clinical studies, the transient mixed chimerism was found to be sufficient for the induction of renal allograft tolerance. Leventhal et al. reported establishment of durable chimerism and kidney transplant tolerance, without graft versus host disease. Non-myeloablative conditioning and transplantation of the enriched hematopoietic stem cells in combination with infusion of the dendritic cells, resulted in development of chimerism and permitted weaning of immunosuppression in five out of eight patients.
In contrast to the kidney studies and despite the success reported in rodents, the studies in large animal recipients of bone marrow and vascularized composite allografts suggest that these transplants may have more stringent requirements. A series of preclinical studies in the large animal models indicates, that while tolerance of the musculoskeletal components of the vascularized composite allografts may be induced by protocols leading to transient chimerism, induction of tolerance to the skin components is associated with the engraftment of donor hematopoietic stem cells, leading to the development and maintenance of mixed chimerism. In large animal models, the most promising results have been achieved using non-myeloablative conditioning regimens and hematopoietic stem cell transfer in the MHC-matched animals receiving a short course of the immunosuppressive therapy.
In the human hand allograft recipients, despite the transfer of the bone marrow component, peripheral chimerism has not been detected. Only transient microchimerism was reported, and described as presence of the donor antigen-presenting cells in the epidermis only at 77 days after hand transplantation; however, it was not detected thereafter.
Immunosuppressive protocol of the first French face transplant patient included thymoglobulin, tacrolimus, mycophenolate mofetil, prednisone, and two infusions of the donor bone-marrow cells. The patient did not develop graft-versus-host disease and only a very low level of microchimerism (0.1% of the donor cells within the CD34 + enriched bone marrow cell population) was detected in the peripheral blood at 2 months after transplantation. At 5 years after transplantation, the patient is on maintenance immunosuppression consisting of sirolimus, mycophenolate mofetil, and prednisone.
Schneeberger et al. reported early results of a protocol, which combined pre-transplant lymphoid depletion with the delayed post-transplant donor bone marrow cell infusion. This protocol resulted in graft survival under tacrolimus monotherapy in five hand/forearm transplant recipients. Patients demonstrated sustained improvement in the motor function and sensory recovery. Infections were not reported. Post-transplant immunomonitoring revealed transient presence of moderate levels of the donor-specific antibodies and adequate immunocompetence. High-resolution ultrasound biomicroscopy demonstrated the presence of patent vessels with mild luminal narrowing/occlusion reported only in 1 patient. Protocol skin biopsies showed absent or minimal perivascular cellular infiltrates.
Cell-Based Therapies in VCA
Achieving mixed chimerism while minimizing the adverse effects of the conditioning protocols remains a challenging task and a variety of cell-based therapies have been investigated as the potential adjunct therapies in an effort to enhance transplant engraftment and survival and to mitigate complications such as GVHD.
T-Regulatory Cells
T-regulatory cells (Tregs) represent a population of the lymphocytes capable of suppressing the immune responses. CD4 + CD25 + FoxP3 + T regulatory cells were shown to prevent both the acute and chronic rejection of skin and cardiac allografts.
Following radiation-free conditioning and delivery of conventional dose of bone marrow in a fully mismatched murine model, administration of T-regulatory cells of the donor origin, combined with anti-CD40L monoclonal antibody, CTLA4Ig co-stimulatory blockade, and a short course of rapamycin, resulted in cell engraftment, development of stable multilineage chimerism, and subsequent tolerance to skin allograft following radiation-free conditioning and conventional dose bone marrow transplantation in a fully mismatched murine. Rapamycin proved to facilitate selective expansion of T-regulatory cells while inhibiting clonal proliferation of the effector cells.
Issa et al. demonstrated the ability of Tregs to prevent transplant arteriosclerosis (a hallmark of chronic graft rejection), and uniquely, to induce tolerance to human skin allografts. There is also evidence that in the context of bone marrow transplantation, donor CD4 + CD25 + cells may protect against acute graft-versus-host disease (GvHD). The outcomes of these experiments are certainly encouraging, but further work is required to refine the specificity of the Treg markers, and to provide evidence of the efficacy of this protocol in the large animal models prior to considering the therapeutic application of T-regulatory cells in the clinical cases of VCA.
Dendritic Cells
Dendritic cells (DCs) as the specialized antigen presenting cells, are known to facilitate immunologic tolerance. They exert their tolerogenic effects through the interaction with T cells and they have been shown to suppress CD4 + and CD8 + T cell proliferation and to control the activation and function of the CD4 + CD25 + Foxp3 + Tregs.
The immature dendritic cells (characterized by a low expression of the cell surface MHC II and co-stimulatory molecules) induce tolerance on interaction with T cells, while mature dendritic cells (high expressers of the MHC II and co-stimulatory molecules) induce an effector response from the T cells. However, it has been demonstrated that dendritic cells may be directed toward change of the phenotype by ligation of specific cell surface receptors.
Tolerance of cardiac transplants has been achieved in rodent models following administration of the donor allopeptide-pulsed DCs in combination with a short course of antilymphocyte serum. Long-term survival of skin allografts and hind-limb composite tissue allotransplants, have also been demonstrated, in some cases supported by expansion of Tregs. In the context of mixed chimerism protocols, it has been demonstrated in the murine model that co-transplantation of bone marrow cells with the immature dendritic cells leads to engraftment and stable multilineage chimerism without cytoreductive conditioning and with no evidence of GvHD. These chimeras showed long-term acceptance of cardiac allografts, and while skin tolerance was not achieved, the challenged skin grafts showed significantly prolonged survival.
There have been some early studies in non-human primates, which demonstrated the presence of the tolerogenic dendritic cells which modulated T-cell responses to alloantigens. It was also demonstrated that dendritic cells are able to induce tolerance in rhesus macaques and facilitate expansion of T-regulatory cells in human volunteers. These findings point to the possibility of “negative cellular vaccines” as a potential route to tolerance in the composite transplants, but further preclinical work is required.
Mesenchymal Stem Cells
Mesenchymal stem cells (MSCs) are a component of the bone marrow stroma, and play a homeostatic role in hematopoiesis, through synthesis of cytokines and growth factors. MSCs have been shown to lack expression of the co-stimulatory molecules and consequently, have limited capability for stimulating alloreactive T cells. Furthermore, MSCs possess immunological inhibitory potential, suppressing proliferation in the mixed lymphocyte cultures and prolonging skin allograft survival in the rodent transplantation model.
The natural physiologic role of the MSCs within bone marrow stroma, has been found to be successful in treatment of GvHD in patients following bone marrow transplantation. MSCs were proven to facilitate engraftment and induction of tolerance to composite limb allografts. In the rat model of hindlimb allotransplantation, supported with an immunosuppression protocol consisting of antilymphocyte serum, rapamycin, total body irradiation, bone marrow cells transplantation, and MSCs, the animals exhibited stable chimerism, tolerated their transplanted limbs for over 100 days without any exogenous immunosuppression, and showed no evidence of GvHD. Application of the MSCs as the supportive therapy was successful in combination with bone marrow transplantation in tolerance induction in the large animal model. In an outbred miniature swine model, the survival of heterotopic hindlimb allografts was achieved for over 200 days in animals treated with irradiation, bone marrow transplantation, and 28-day protocol of cyclosporine, combined with three doses of donor MSCs. Interestingly, while no signs of GvHD were observed, the same regimen in the absence of MSCs resulted in a maximal allograft survival of 57 days and symptoms of severe GvHD leading ultimately to animals’ death.
The only clinical application of MSCs, in the VCA, was reported by Del Bene et al. where autologous MSCs were infused at 12 h, following double hand transplantation. The immunosuppressive protocol consisted of basiliximab, tacrolimus, mycophenolate mofetil, and methylprednisolone. Prednisone was completely withdrawn at 7 months post-transplantation.
MSC-based protocols alone or in combination with other cell therapies seem promising in VCA; however, their application is still at the experimental stage and further proof of their efficacy is needed.