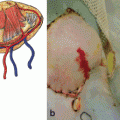
Fig. 8.1
Episodes of acute rejection within the first year in upper extremity transplantation
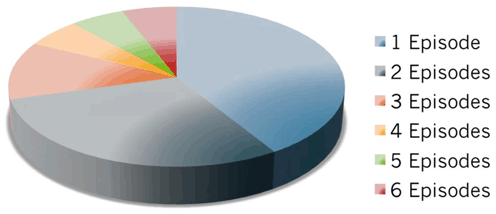
Fig. 8.2
Episodes of acute rejection within the first year in face transplantation
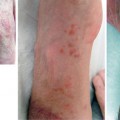
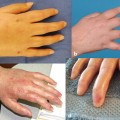
In extremities, as well as in face transplantations , AR reactions manifest clinically as erythematous macules, diffuse redness, or asymptomatic papules over the allografted skin [7] . Microscopically, they show characteristic, although nonspecific, changes involving mainly the dermis and the epidermis that may even extend to the hypodermis in cases of severe rejection. These changes consist of a perivascular lymphocytic infiltrate in the superficial and mid-dermis predominantly made of CD3 + /CD4 + T cells, with smaller proportions of CD8 + and TIA-1 + cytotoxic T cells, FoxP3 + T regulatory cells, and occasional CD68 + histiomonocytic cells of recipient’s origin. The epidermis can show exocytosis and keratinocyte necrosis or apoptosis associated with basal keratinocyte vacuolization. More rarely, the epidermis shows spongiosis (intercellular edema) or lichenoid changes (orthokeratotic hyperkeratosis, hypergranulosis, acanthosis, band-like subepidermal infiltrate) similar to those observed in (lichenoid) graft-versus-host disease (GvHD). In the case of very severe rejection, the epidermis (and its appendages, hair follicles, and sweat glands) may show extensive necrosis. In those severe cases, the infiltrate may extend to the hypodermis and also contain eosinophils [5]. A specific score (Banff score 2007) consisting of five severity grades has been established in order to assess AR in hand and face transplantations [8, 9] .
In the case of facial allotransplantation, biopsies obtained from the allografted oral mucosa show qualitatively similar changes, which are as a rule more pronounced than those found concomitantly on the skin [10–12]. The explanations for this discrepancy are unclear; however, one possible explanation could be the existence of a higher density of vessels and antigen-presenting cells (dendritic and endothelial cells) in mucosa versus skin .
The presence of C4d deposits in the skin and their significance is somewhat controversial. In our experience, such deposits are rarely (if ever) detected in the allografted hand and face skin and mucosae [13]. In some studies, such deposits have been reported in the skin of hand transplants with and without signs of AR, albeit in the absence of concomitant donor-specific antibodies [14, 15]. The role of humoral rejection in VCA is not clear. It has yet to be demonstrated [16] in experimental limb transplantation, although in clinical hand and face transplantations alloantibody production against donor human leukocyte antigen (HLA) has been reported. In the unique case of lower extremity transplantation [17], the patient presented two AR episodes that were graded on the basis of Banff score; C4d deposits were detected without donor-specific HLA antibodies, but they were considered to be “artifacts” by the authors. It is interesting to note that in the first posttransplant year the patient presented two episodes of AR, grade II and III, respectively . These findings showed that the number and severity of AR episodes are not correlated to the mass of the transplanted tissues, which primarily consists of muscle. Indeed, several studies [18, 19] suggested that muscles are the most sensible tissue to the ischemia/reperfusion injury and, consequently, to rejection.
The immune response in hand and face transplantations is essentially T-cell mediated, and the cytotoxic activity of these T cells is donor specific, as shown in the first face transplantation recipient [20]. It has also been shown that the rejection of a VCA in sensitized recipients is mainly cell mediated and differs mechanistically from that of solid organ transplantation [21]. The predominance of the T-cell response during VCA rejection probably explains the efficacy of the traditional triple therapy approach used in the majority of VCAs (steroid, tacrolimus, mycophenolate mofetil (MMF), which primarily targets the T-cell response .
Chronic Rejection
In solid organ transplantation , high rates of acute rejection are often associated with high incidence of chronic rejection and low organ survival rates. However, despite the high incidence of AR episodes to date (76.7 % of the hand recipients and 54.5 % of the face recipients presented at least one episode of AR in the first posttransplant year), no clear evidence of chronic rejection has been found in compliant patients on long-term follow-up [7] and the graft survival rate was 90.5 and 95 % at 1 year for upper extremity and face transplants, respectively.
Insufficient data are available to define specific changes of chronic rejection in VCA. The Banff 2007 classification, while useful in assessing acute rejection, has not yet included features of chronic rejection [8]. Clinicopathologic features suggestive of chronic rejection could include myointimal proliferation of arterioles, loss of adnexa, nail changes, skin and muscular atrophy, and fibrosis of deep tissues.
The small number of extremity and face allotransplantations performed to date, the relatively short follow-up, and the limited data on deep tissue biopsies make it difficult to evaluate the real incidence of chronic dysfunction of VCA .
The hallmark feature of chronic rejection is graft vasculopathy, which initiates with injury of vessels’ endothelium, proliferation and migration of smooth muscle cells, deposited matrix protein, and finally concludes with arterial luminal narrowing.
An experimental murine study employing a rat hind limb transplantation model showed that graft vasculopathy was determined by multiple AR episodes and that it was the last lesion to occur after skin and muscular atrophy [22].
Another experimental model of face transplantation in nonhuman primates [23] showed graft vasculopathy involving both the external carotids as well as smaller vessels when immunosuppression was withdrawn. All grafts developed arteritis, intimal hyperplasia progressing to vessel occlusion, and tertiary lymphoid follicles.
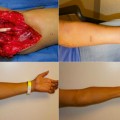
The first clinical case of graft vasculopathy [24] occurred 275 days after transplantation and was characterized by acute ischemia of a grafted hand. On the basis of the pathological findings, which showed intimal hyperplasia and vessel occlusion similar to that reported in heart transplantation, the Louisville team introduced a sophisticated ultrasound technique for monitoring transplant vasculopathy. Although this technology remains experimental and not yet ready for clinical application, varying degrees of neointimal hyperplasia in other hand-grafted recipients were reported without correlation with the number of AR episodes, follow-up time, and the presence of acute skin rejection.
Additionally, one case of vasculopathy in a vascularized knee allograft has been reported [25]. In this case, necrosis of the skin sentinel flap occurred 36 months after transplantation, and the knee was removed 50 months posttransplantation after showing intimal hyperplasia and occlusion of the small vessels .
Although currently there are no reports of face allograft vasculopathy, careful evaluation of skin lesions and the treatment of all AR episodes are imperative for proper graft management.
A face loss was reported due to patient noncompliance with his immunosuppressive regimen. The patient died afterwards, but the circumstances concerning his death are not clear.
It is of paramount importance, particularly in facial transplant, to detect the early signs of graft vasculopathy due to the tremendous psychological impact of chronic rejection and graft loss on the recipients .
Graft-Versus-Host Disease, Chimerism, and Tolerance
Although bone marrow was present in the transplanted upper extremity bones and a substantial amount of active hematopoietic cells was in the bone marrow of the bilateral femoral transplantation, GvHD did not occur in any of these cases. Similarly, GvHD also failed to develop in partial and total face transplantations including the mandible, a bone that contains a rich marrow compartment.
It is even more remarkable that no GvHD occurred in the recipients of face or upper extremity transplantations who also received bone marrow infusions.
GvHD has never been observed in VCA to date; however, this possibility has to be considered prior to expanding immunosuppressive protocols including bone marrow infusion [26].
Peripheral blood microchimerism was detected for a very short period in two hand recipients in Louisville [27]. Although the long bones transferred as part of arm transplantation contained a substantial amount of active bone marrow, it was neither sufficient to induce chimerism nor allow for a reduction in immunosuppressive treatment .
Chimerism was not assessed in the case of bilateral femoral transplantation.
The team from Pittsburgh University and Johns Hopkins University [28] used a new protocol of “immunomodulation” or “minimizing immunosuppression” in upper extremity transplantation based on a regimen of donor bone marrow infusion 10–14 days posttransplantation and low-dose tacrolimus monotherapy after an induction with Campath-1H. Although they have obtained interesting and highly encouraging results, neither chimerism nor tolerance has been detected in these patients to date.
Similarly, face transplant recipients, including those receiving bone marrow infusions, did not show chimerism or tolerance. The only exception to this observation is first face transplant patient, who showed a transient microchimerism in a CD34 +-enriched cell population [20]. Despite this anomaly, there was no evidence for reduction in immunosuppressive requirements. In this case, the absence of durable donor chimerism and tolerance may be explained by an insufficient hematopoietic engraftment due to poor hematopoietic stem cell quality or an insufficient conditioning regimen.
At present, no VCA recipient has proved to be spontaneously tolerant. Indeed, it was noted in the first hand allotransplantation and in all recipients who discontinued immunosuppressive therapy that consequent rejection of the graft inevitably occurs [5, 7].
It is evident that the results obtained in rodent vascularized bone marrow transplant could not be transferred to the clinical practice in extremity as well as in face transplantation.
Immunosuppression and Complications
The large majority of upper extremity transplants [7] have been maintained on immunosuppression therapy similar to that used in solid organ transplantation, consisting of tacrolimus, steroids, and MMF. The induction therapy included antithymocyte globulin, basiliximab, and more recently, Campath-1H. All recipients received tacrolimus in the early postoperative period because of the stimulatory effect of this drug on the synthesis of axotomy-induced growth-associated protein (GAP-43) that seems to promote nerve regeneration [29]. Over the years, various modifications have been made to the initial maintenance treatment in order to decrease the risk for opportunistic infections, metabolic disorders, and malignancies. Such modifications include steroid-sparing maintenance, MMF-free treatment, and replacing tacrolimus with sirolimus.
The use of conventional immunosuppression in VCA is associated with the same complications commonly reported in solid organ transplantation [7




Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree