Introduction
Hand and upper extremity transplantation is the most common form of vascularized composite allotransplantation (VCA) or reconstructive transplantation and has evolved from a procedure only performed at the distal forearm level to a procedure now being performed above the elbow more proximally. At the time of writing this chapter, the authors can count 107 hand and upper limb transplants performed throughout the world. The “modern era” of hand transplantation was ushered in with the first hand transplants performed with modern immunosuppressive medications in 1998 in Lyon, France, and 1999 in Louisville, KY. While both transplants were unilateral distal forearm level transplants performed 4 months apart, the Louisville transplant was the first to demonstrate long-term success due to patient compliance. This emphasized patient-centric factors, which are critical to the success of any kind of transplant. Psychological and social factors are the most important elements in long-term “graft” maintenance. Therefore, all successful upper extremity transplantation begins with screening. Following screening, careful surgical planning must be performed and precise execution of both donor and recipient portions of the transplant operation must be accomplished. Postoperative monitoring and therapy are instituted and therapy may be required for several years to come. Rejection episodes and complications should be expected and anticipated. This chapter discusses the key portions necessary for performing and maintaining a successful upper limb allotransplant.
Immunotherapy
Despite the fact that the clinical results and outcomes with hand and upper extremity transplantation have been highly encouraging, the requirement of life-long, high-dose, multidrug immunosuppression associated with a profound side-effect profile still hampers broader application of these life-changing reconstructive modalities. Currently, there is no standard immunosuppressive regimen established for extremity transplantation; however, most patients have been treated with a “conventional” triple-drug immunosuppressive strategy, largely extrapolated from solid organ transplantation. This includes either polyclonal (antithymocyte globulin, ATG) or monoclonal (alemtuzumab or basiliximab) antibody induction therapy followed by high-dose tacrolimus, mycophenolate mofetil (MMF), and steroids for maintenance immunosuppression.
The adverse effects and toxicities of these immunosuppressive agents include metabolic, infectious, and neoplastic sequelae, and in extreme cases may contribute to patient mortality. Specifically, the triple-drug regimens have resulted in nephrotoxicity, hypertension, new-onset diabetes mellitus, hyperlipidemia, leukopenia, cardiovascular disease, and bilateral hip osteonecrosis. Opportunistic infections have also been observed, most commonly with cytomegalovirus (CMV), which can be potentially life-threatening and can trigger acute rejection. This has led some centers to avoid the high-risk combination of CMV-positive grafts in CMV-negative recipients in upper extremity transplantation. Other encountered opportunistic infections include human papillomavirus, herpes simplex, Staphylococcus aureus osteitis, Clostridium difficile , molluscum contagiosum, and mycoses. Malignancy is another common complication after solid organ transplantation, with an incidence up to five-fold higher in transplant recipients compared with the general population. However, most hand and upper extremity transplant recipients have been followed for <10 years, so it is difficult to assess the true risk and incidence faced by these patients with regard to malignancy.
The risks associated with the long term, potentially lifelong usage of these drugs have prevented the widespread application of VCA as a first-line reconstructive modality in patients with complex tissue defects and limb loss. Therefore, there is a critical need for novel protocols that will allow patients to be weaned off immunosuppression or to induce tolerance to their grafts.
Several centers around the world, including our own, have attempted to develop novel, innovative therapeutic concepts of immunoregulation to reduce systemic immunosuppression to minimal levels. Similarly, recent advances in cell-based therapies and immunosuppressive drug development that incorporate the unique elements and biology of vascularized composite allografts have also shown favorable results at achieving this goal. Clearly, the induction of donor-specific tolerance would obviate the need for long-term maintenance immunosuppression after extremity transplantation, and great strides in this regard have currently been made in translational large animal models. However, until those strategies are implemented in the clinical arena, we advocate for short-course depletional induction followed by low-dose maintenance monotherapy to minimize toxicities of conventional immunosuppression and maximize the risk–benefit ratio for hand and upper extremity transplantation. Our group has recently successfully implemented such a protocol for upper extremity transplantation with favorable functional and immunologic outcomes.
Indications
The authors feel that upper extremity transplant is indicated for individuals of adequate social, psychological, medical, and immunologic health who have unilateral or bilateral hand/upper limb loss that has resulted in significant functional and/or psychological disability, when a substantial enough improvement (based on the level of transplantation and expected functional recovery) can be anticipated that ethically weighing the balance of risks and benefits favors transplantation. We hesitate to create specific indications such as only bilateral amputees or bilateral or dominant hand unilateral amputees only, because this does not incorporate the global assessment of function and potential benefit that a patient may experience through a thoughtful application of reconstructive transplantation. The authors apply this philosophy also through their cell-based, nonsteroid, monotherapy, immunoregulatory protocol, as indicated above. However, many centers worldwide, using traditional immunosuppression do consider only bilateral amputees or unilateral amputees of dominant hands only, and exclude nondominant unilateral amputees and above elbow amputees.
Preoperative Considerations
Patient Screening
The importance of screening cannot be emphasized enough. Screening includes evaluations of the patient’s psychological and social problems, tools, weaknesses, and strengths. Some patients are hand transplant candidates due to “bad luck” or circumstances out of their control. But some are missing limbs due to poor decision-making that may have been a single episode of lack of judgment, or a long-standing pattern of bad choices. One must consider if a pattern of bad choices will change because of a hand transplant. The single best predictor of transplant medication compliance by a patient is the presence of stable and caring family/friends actively engaged within the patient’s life.
Further screening involves evaluation of the patient medically to see if they are healthy enough to undergo a long and physiologically taxing procedure and the immunotherapy regimen to follow without substantially worse risks for problems such as malignancy, renal disease, heart disease, pulmonary disease, infection, ocular disease, dental disease, etc. Many patients may have added risk for infection due to their initial disease process causing limb loss. Our liver transplant anesthesia providers also evaluate the patient for possible line placement and anesthesia risk/concerns.
Immunological screening involves blood and HLA typing, assessment for pre-formed donor-specific (DSA) or HLA antibodies and sensitization, as well as further assessment for infectious diseases known to complicate the immunological management and prognosis for rejection and malignancy in the future such as CMV and EBV infection. It is the authors’ practice that recipients who are seronegative for antibodies, indicating no prior infection with these viruses, should not receive transplants from CMV+ or EBV+ donors. All patients should include an evaluation by a transplant infectious disease physician to screen for these and other problems.
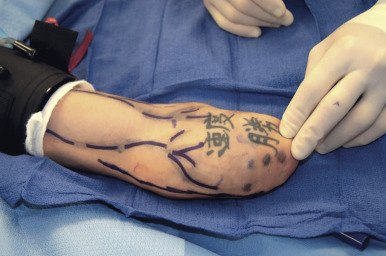
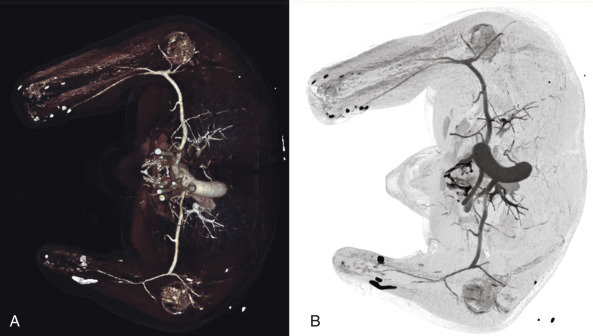
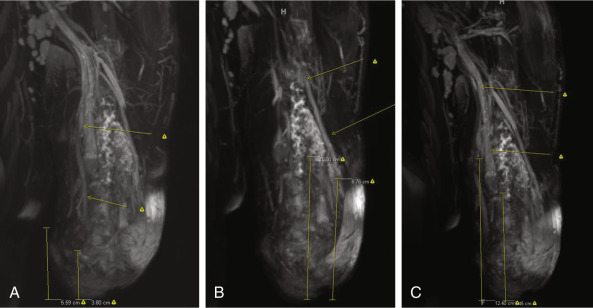
Surgical evaluation focuses on the function of the patient with and without prostheses, and one should also consider their prior participation in rehabilitation. Surgical planning focuses on the level of amputation along with associated hard and soft tissue deficits proximal to the amputation site, which may either complicate a transplant or require reconstruction before or during the transplant. Physical evaluation for skin quality, joint range of motion, remaining motor group strength, location of scars, location of Tinel’s sign over nerve stumps, limb/stump length and circumference, palpable pulses, wounds, heterotopic ossification, etc. should be noted. Radiographic evaluation includes plain films of both upper limbs and either conventional or computed tomographic angiography, along with venous phase reconstructions ( Fig. 65.1 ). Venous mapping is also performed by ultrasound with vein locations and diameters recorded. We physically map the veins with a surgical marking pen and then photograph the limbs for a permanent record of vein location. MR neurography ( Fig. 65.2 ) may be employed when necessary to evaluate for location of nerve stumps or possible traction/avulsion injury of peripheral nerves more proximal than expected by location of amputation. The authors perform this imaging on all patients capable of having MR neurography performed if their mechanism of injury/amputation allows for the possibility of stretch or avulsion injury and/or an obvious Tinel’s sign that is reproducible to the correct nerve sensory distribution cannot be elicited in the vicinity of the amputation stump.
Once patients have been approved following close and detailed screening, they may be enrolled into our IRB-approved hand and upper extremity transplant program. Patients will not be enrolled if they use any form of tobacco, illegal substance, or cannot commit to staying at our institution for at least 3 months postoperatively or treating rehabilitation as their full-time job for the first 1–2 years after transplantation.
Preoperative Planning
Each patient gets a reconstructive transplantation operative plan specifically designed for their particular anatomic needs. While many will have certain similarities, none are exactly the same. Planning incorporates known factors such as soft tissue coverage and skin quality, skeletal factors such as limb length, joint condition, presence of heterotopic ossification, vascular imaging, and possible nerve imaging, as well as unknown factors such as the degree of muscle versus scar present, quality of remaining muscle, potential nerve quality and length, tendon length, potential for indolent infection, etc. All surgical planning incorporates our desire to protect the length of the patient’s limb if at all possible, assuming the transplant were to fail. This means preserving a proximal forearm stump with muscle coverage while still transplanting adequate forearm motors to make a forearm-hand transplant functional, for example. Conversion of a below elbow to above elbow prosthesis user would be a devastating complication for a patient who suffers a transplant loss. However, for a patient with significant elbow pathology already, replacing this joint with a transplant may actually allow for better ultimate function and may be a consideration.
Cadaver Rehearsal
Surgical plans are discussed in classroom format with group input and then any remaining questions are worked out as a group in the cadaver anatomy laboratory. Once the various components of the surgical plan have been discussed and worked through, another cadaver rehearsal is performed with the correct surgical sequence used and the plan is continually refined until all group members are satisfied.
Team Assignments
Surgeons are assigned by name to specific teams and tasks, both incorporating their clinical strengths as well as their ability to work with individual team members. Usually we use a plastic surgery hand surgeon and an orthopedic surgery hand surgeon on each three-member team, with the additional member being either another hand surgeon, a microvascular plastic surgeon, or hand surgery/microsurgery fellow. This balance of team members ensures that appropriate soft tissue and hard tissue techniques and care will be provided. Osteosyntheses are performed by hand surgeons of either specialty, as long as they have the appropriate expertise.
Microsurgery (nerve and vascular) is performed by the plastic surgery hand surgeons, plastic surgery microsurgeons, or orthopedic hand surgeons who have adequate training and experience in microsurgery.
Rotating Surgeons
In an effort to prevent surgeon fatigue, surgeons are able to rotate in and out of the operation in teams once the osteosynthesis begins as the two teams per limb transplanted reduce to one team. Thus, half the previously operating members may break and further team members who may not have yet operated can also be used to help relieve surgeons. Typically, the microsurgery team is a different group than the team performing the osteosynthesis and tendon/muscle repairs. The microsurgery team may also rotate in/out individual members as the hours pass, as every vein capable of coaptation is usually anastomosed resulting in an artery to vein ratio of 2 or 3 : 1.
Use or Replace Existing Motors
For many amputees in the middle third of the forearm and brachium, the decision to use the already present muscles to power the transplant or replace the existing muscles must be made. The authors are unaware of any current guidelines to help direct this decision. The surgeon must decide if the motors present have adequate length, and cross-sectional area, and if they themselves have sustained intrinsic damage that may result in loss of excursion or strength, such as fibrosis, based on their physical assessment of the patient and the mechanism of limb loss. It is likely that blast injuries and ischemic injuries may compromise surviving muscle and limit its ability to provide excursion or power. However, replacing these already innervated motors will result in some loss of motor axon count providing innervation and allows for “mistakes” in reinnervation, such as synkinesis, loss of specificity, or failure to reinnervate. In the authors’ limited experience, we currently prefer to replace questionable forearm motors if half or more of the muscle-tendon unit length is considered compromised or missing. Even less experience is present to guide transhumeral transplants. However, based on our unpublished anatomic research, even up to the junction of the proximal and middle thirds, enough new elbow flexor and extensor motor units are being transferred so that this may be considered an innervated muscle transfer as well. For transplants at the mid-brachium to distal third, our experience is that adequate elbow strength is obtainable using the recipient’s own motors if those muscles are otherwise uncompromised.
Ischemia Time
The authors are unaware of any clinical studies demonstrating the critical tolerable cold ischemia time of preservation solution – infused allograft limbs. Cold ischemia times have approached 11–12 h in our own experience without known sequelae. This is a theoretically larger concern in allografts containing more skeletal muscle such as transhumeral or proximal forearm transplants, than distal forearm transplants. We have performed small incision forearm fasciotomies and carpal tunnel release on a single transhumeral transplant and did not perform these measures on a second transhumeral transplant without attributable difference in outcome. At this time, our algorithm requires maintenance of cold ischemia (ice on the operating table) until the clamps on the reconstructed artery are released for reperfusion. We typically perform as much muscle, tendon, and nerve reconstruction as possible while in a bloodless field due to the preciseness and rapidity with which these can be performed in this manner compared with the bloody field of a reperfused transplant. If ischemia time was approaching 7 or 8 h prior to beginning our transplant, however, we would not hesitate to alter the sequence of events or even temporarily shunt the graft to allow for some perfusion prior to continuing with the transplant.
Surgical Technique
Surgical technique is dependent on the level of transplantation. Technique is essentially the same for bilateral and unilateral transplants except for a need for enhanced communication between both surgical teams and the anesthesiology team in regards to blood loss and reperfusion.
Donor Operation
Procurements are coordinated by the regional organ procurement organization (OPO) and will likely involve teams for procurement of abdominal and thoracic organs. Priority of course, is given to maintaining the viability of life-saving organs during the procurement, so any process that has significant potential to endanger them will be terminated by the OPO coordinator. The upper extremity procurement level depends on the specific donation. For distal forearm and mid-forearm transplants, an elbow disarticulation is usually adequate in providing soft tissue, vessel length, nerve length, and extra skin/bone for grafting as needed ( Fig. 65.3 ). For distal transhumeral transplants, we recommend a proximal humeral procurement at the highest level that tourniquet control can be still be maintained. The authors have used a thin, non-pneumatic tourniquet (HemaClear, OHK Medical Devices, Inc, Grandville, MI) placed as high on the arm as possible over the deltoid with the tourniquet fixed to the skin with penetrating clamps to help prevent slippage ( Fig. 65.4 ). For middle or proximal arm procurements, control may be required via intrathoracic cannulation and control of the subclavian vessels by the thoracic procurement team.
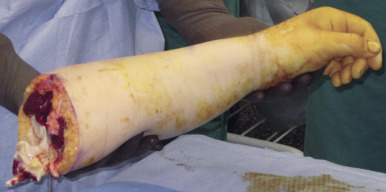
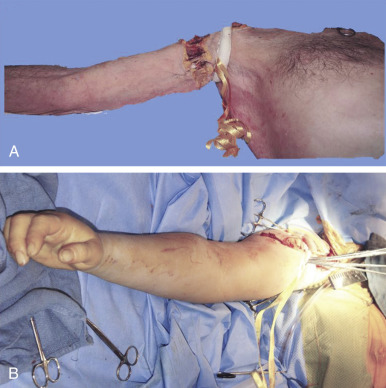
Depending on the level or procurement and ability of all teams to work together, upper extremity procurement may occur concurrent to standard organ procurement, before actual procurement, or after procurement. We recommend procuring prior to or during visceral procurement to keep ischemia times short.
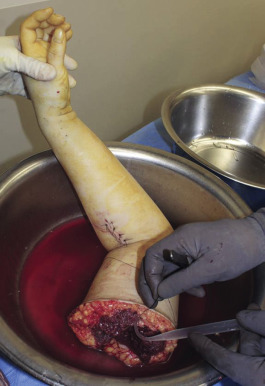
For concurrent procurements, the arm(s) are on a hand surgery table 90° to the operating table. All surgical teams simultaneously prep and drape with full exposure of the arm to the axilla ( Fig. 65.5 ). A tourniquet is inflated after minimal exsanguination, or in the case of proximal upper arm procurement, a non-pneumatic tourniquet is placed, which is self-exsanguinating and secured to the arm in the region overlying the deltoid muscle with penetrating clamps. This tourniquet will be left inflated and/or in place until all organ procurements have been completed. A guillotine incision is outlined and the medial/volar component created first with dissection down through the nerves to the brachial artery/veins. Any divided nerves are tagged if there is potential for any later question of identity. The brachial artery is isolated. It is ligated proximally and distal control is obtained. The largest catheter possible is used to cannulate the artery and this is secured with suture. The veins are clipped or ligated proximally and cut distally. The procedure then proceeds rapidly from medial/anterior to lateral/posterior dividing muscle with cautery and either disarticulating the elbow or performing an osteotomy with a power saw through the humerus. Once the limb is completely detached, it is immediately taken to the back-table, where perfusion solution is instilled ( Fig. 65.6 ). The solution is instilled until at least 500 mL have been infused or all effluent is clear. Typically perfusion volume ranges between 500 mL and 1000 mL. Note is made of which venous system the dominant drainage of infusion is coming from (brachial veins vs superficial veins), as this system will be critical for outflow, though in reality every single vein capable of anastomosis should be anastomosed . The arm is then wrapped in moist gauze and placed in standard sterile triple bag containment identical to that of solid organ transplantation, placed on ice, and secured in a cooler.
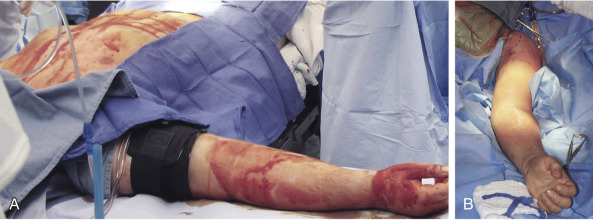
For limb procurements performed prior to organ procurement, the operation will usually begin with all surgeons prepping and draping together with the arms on a hand surgery table identical to the operation described before. The abdominal and thoracic viscera teams will open the chest and abdomen, prepare and cannulate the vessels to the organs for procurement, but then stop. At this point they step back and the upper limb procurement team performs the operation described above. Once completed, all tourniquets are left in place and the chest and abdominal procurement teams then step back in and continue their standard operation.
Once all procurement teams have completed their operations and there is no longer any perfusion within the donor, a remaining party from the procurement team will remove the tourniquets and close the skin of the arm stumps. The donor is then fitted with a prosthetic hand with variable length sleeve, which will attach to the stump of the amputated limb.
Anesthesia Considerations
The authors have found preoperative placement of peripheral nerve catheters for postoperative pain management and theoretical benefit of chemosympathectomy to be helpful. A key point is to not dose the pain catheters with a long-acting agent prior to surgery. Surgical stimulation at the operative site may be necessary to maintain the patients at a normotensive level without undue amounts of crystalloid support, which can contribute toward coagulopathy. Central venous lines and an arterial line are placed prior to beginning surgery. The immunological induction agent is given at this time. The ultrasound machine used by the anesthesiology group for peripheral nerve catheter and vascular access catheter placement is then used by the surgical team to map peripheral veins on the recipient limb. These are marked with a permanent marker (as opposed to a surgical marker, which washes off too easily) to locate veins during the operation ( Fig. 65.7 ).