Steven Dayan, David Ellis, and Mary Lynn Moran address questions about Facial Fillers for discussion and debate:
- 1.
Are there different indications for the different fillers?
- 2.
In your opinion, what are the durations of the various fillers?
- 3.
Is there still a role for the use of collagen injections?
- 4.
What complications concern you and what do you do in your practice to attempt to avoid or minimize these?
- 5.
What type of anesthesia do you use, when, and why?
- 6.
Analysis: over the past 5 years, how have you modified your techniques or approach or what is the most important thing you have learned/observed in working with injectables and fillers?
Steven H. Dayan, David Ellis, and Mary Lynn Moran address questions about facial fillers for discussion and debate:
- 1.
Are there different indications for the different fillers?
- 2.
In your opinion, what are the durations of the various fillers?
- 3.
Is there still a role for the use of collagen injections?
- 4.
- 5.
What type of anesthesia do you use, when, and why?
- 6.
Dr Dayan presents a video of his technique for facial fillers using a blunt tip cannula. Available at : http://www.facialplastic.theclinics.com/
Are there different indications for the different fillers?
Dayan
It has been my impression that, as we gain more experience, the greater are the apparent differences between the various fillers approved by the Food and Drug Administration (FDA). Once recognized, each filler’s unique physical properties can be relied on to achieve specific outcomes in individual patients. Furthermore, the fillers are priced differently, have different durations, and are marketed differently. All of these factors ought to be taken into consideration before deciding which filler is best for a patient.
Hyaluronic acid fillers
The most popular fillers are the hyaluronic acids (HAs), as they comprised 85% (1.3 million of the 1.5 million) of the filler treatments in 2010. The 2 leading brands Restylane/Perlane (Medicis Aesthetics, Scottsdale, AZ, USA) and Juvéderm Ultra/Ultraplus (Allergan, Irvine, CA, USA) make up most of the market share, and although they are often thought of interchangeably they have very different physical properties. Juvéderm is positioned as the “smoother filler.” Its gel-like 24 mg/mL concentration with hydrophilic properties 6 times that of Restylane is likely the reason behind its smoother effects. Restylane, a firmer product defined by its rheological properties G′, is more than 6 times more resistant than Juvéderm to deformation.
It is these precise differences between Juvéderm and Restylane that I frequently rely on for specific indications. Juvéderm-corrected areas tend to lead to a fuller and more diffuse augmentation as water is absorbed. I also have noticed that the Juvéderm augmentation may be dynamically related to the body’s current levels of hydration. Anecdotally I have a young staff member who, after receiving Juvéderm into her lips, has a noticeably variable augmentation of her lips the day after she has a meal of salty food. I have not witnessed this with the less hydrophilic Restylane. Consequently, knowledge of its greater hydrophilicity can be used to an advantage, especially in the lips ( Fig. 1 ). For the younger patient who is seeking enlarged lips and already has a full body lip, I will place 0.4 mL of Juvéderm deep into each lip body. For the more mature patient with natively thinner lips and/or who is overly concerned about a recognizable difference, I will place a fine whisper (0.3–0.5 mL) of Restylane within either vermilion border. This procedure has the effect of obviating fine vertical rhytids of the lip and nicely defining the lip border without extending into the cutaneous lip or body. Lipstick can now be placed without concerns of migration, and the lips appear more youthful but not enlarged.
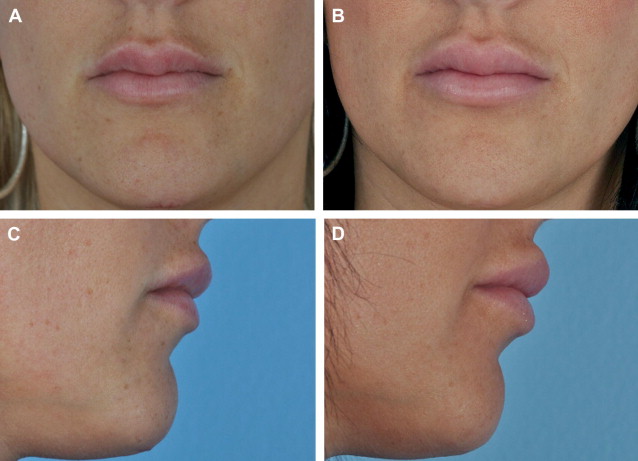
However, because of its firmer nature and less hydrophilic properties, Restylane can lead to lumps and irregularities if not placed evenly. Juvéderm, with its highly hydrophilic nature, tends to attract more fluid and, therefore, results in a diffuse filling effect, which I believe with its nonparticulate gel consistency work to deliver its marketed “smoother” effect. However, if a larger quantity of Juvéderm (more than 1.0 mL approximately) is placed into a lip, it can rather quickly lead to an unnatural, large shelf-like appearance of the cutaneous white lip.
Beyond the lips, the differences in hydrophilic nature of the HAs can also be relied on when correcting or augmenting the nasolabial folds (NLF) and cheeks. For the buccal cheek space, I find Juvéderm’s superior hydrophilic and diffuse filling properties more beneficial, whereas in the infraorbital tear trough I prefer Restylane. Nowhere on the face are the differences between Juvéderm and Restylane more apparent than when placed into the loose areolar tissues surrounding the orbital region. I find Juvéderm to be contraindicated because it will reliably swell, resulting in dark puffy undereye circles representative of a tired unflattering appearance. Although it can occur with both products under the eyes, the Tyndall effect, whereby optical chamber of HA and fluid reflects blue light, is more commonly associated with Juvéderm. In those with thin, translucent skin, the blueness is distractingly obvious. Nowadays I find myself removing more misplaced Juvéderm from under the eyes than insertion of Restylane.
Both products are also manufactured in a more robust version that is intended for deeper placement into the soft tissues with a potential for greater duration. Perlane, at the same concentration (20 mg/mL), is a version of Restylane with larger particle size, and Juvéderm Ultraplus, at the same concentration as Juvéderm, has greater viscosity. However, I remain unconvinced that a clinically recognizable difference is appreciable between the more robust version and its less expensive version by the patient or me. More recently, I have been limiting my use to the Restylane or Juvéderm Ultra versions. However, I eagerly anticipate the arrival of the highly regarded large-particle Voluma and Restylane SubQ for facial shaping. In addition, Belotero (Merz Aesthetics, San Mateo, CA, USA), a smooth-consistency monophasic HA, seems likely to gain FDA approval soon, and is reported to provide correction without risk for a Tyndall effect because of its trademark cohesive polydensified matrix properties. This product may prove to be superior for treating the tear trough.
Calcium hydroxyapatite fillers
Radiesse (Merz Aesthetics, Inc, San Mateo, CA, USA), a biostimulating filler approved by the FDA in 2006, comprises calcium hydroxyapatite (CaHA) microspheres (30%) surrounded by a carboxy methylcellulose resorbable aqueous gel carrier (70%), leaving behind a matrix scaffolding of CaHA beads and setting up a potential framework for neocollagen deposition. Although I do not find the collagen inducting property to be clinically significant, I find CaHA to be a very reliable and safe product. Radiesse’s high G′, indicating the product’s firmness and ability to project tissues, is a major advantage for correcting deeper wrinkles and folds. I find it particularly helpful for treating those with thicker skin and those who desire highlighting or emphasis of bony prominences such as malar, chin, and jawline areas. Its insignificant hydrophilic properties mean “what you see is what you get” after placing it, and any further augmentation is negligible. Therefore, it is important to realize that if not placed thinly or diffusely, it can result in hard streaks and nodules. I prefer to premix 0.3 mL of 1% lidocaine with epinephrine into the product just before treatment. This FDA-approved process not only makes the product more malleable but also reduces discomfort during placement.
In economic terms, most of the fillers are priced competitively; however, Radiesse contains 1.5 mL of product per syringe, compared with 0.8 mL and 1.0 mL for Juvéderm and Restylane, respectively. Although variations in retail pricing to the patient exist across the country, for most practices, Radiesse with the additional product per syringe may be a more economical option. It goes further for the dollar and this may be a deciding factor for some patients. Radiesse is not immediately reversible, whereas the effects from misplaced or excess HA can be reversed within 12 to 24 hours following an injection of hyaluronidase (Vitrase; ISTA Pharmaceuticals, Irvine, CA, USA). This factor may be an influence on the inexperienced filler patient or doctor.
Poly -l -lactic acid
Poly -l -lactic acid (Sculptra; Sanofi Aventis, Bridgewater, NJ, USA) received FDA approval for cosmetic use in 2009. It is a product that relies on its controlled foreign-body biostimulatory properties, and is preferred by many physicians and patients for its subtle and progressive augmentation. However, too robust of a response can be problematic, resulting in nodules and, rarely, granulomas. I target this product for a small niche of patients requesting global facial volumization. I often prefer fat in such situations, but in thin individuals lacking adequate fat stores for harvesting or in human immunodeficiency virus patients with lipodystrophy, I find Sculptra to be the best and most appropriate filler. Although its augmenting benefits are slowly realized, they can be very impressive; but as the results are not immediate, patients’ expectations have to be well managed. In an era of immediate gratification, the necessity to reconstitute with water days in advance and having to do a series of treatments to recognize the benefits make this product less user-friendly than other off-the-shelf fillers.
Silicone and polymethylmethacrylate
Other fillers that I use less often include the permanent silicone (Silikon 1000 purified polydimethylsiloxane; Alcon Laboratories, Fort Worth, TX, USA) and polymethylmethacrylate (PMMA) (Artefill; Suneva, San Diego, CA, USA). I find the hardness properties of these products very advantageous for filling in the fine rhytids of the upper lip, scars, and deep etched-in dermal rhytids. However, silicone’s long-term questionable side effects prevent me from selecting it in younger patients, the lip bodies, or those with thicker skin. Artefill comprises PMMA beads surrounded by a bovine collagen, and in 2006 became the only permanent FDA-approved filler product in the United States. However, it has had difficulty gaining popularity. Its properties are impressive, as is its 5-year safety data. I use this product for deeper etched-in wrinkles of the face in those insistent on a permanent solution.
Ellis
At present, there is no universally accepted classification system for injectable fillers but they can be categorized into the following parameters: type of filler, method of action, and duration of action. Fillers can be either biodegradable or nonbiodegradable. The source of the filler may be natural/animal, synthetic, or natural synthetic. Method of action can be based on replacement of volume or stimulator of fibroplasia and collagen, or both. The duration of action may be temporary, semipermanent, or permanent. Patients are more interested in their duration of the filler in the specific areas they wish to be augmented.
Indications can be viewed as filler-based and patient-based depending on the characteristics of each filler and where the filler is to be injected. For instance, in the tear troughs, I like to use a minimal amount of filler that is not stiff. HA-based fillers work well in this area. Several pitfalls, however, may occur in this area:
- 1.
Too superficial an injection can result in a bluish hue under thin skin, known as the Tyndall effect.
- 2.
Too much volume can result in lumpiness and exacerbation of bags under the eyes, especially when the patient is smiling.
I have used Juvéderm Refine, Juvéderm Ultra Plus with Lidocaine, and Juvéderm Ultra with Lidocaine for this area. Juvéderm Voluma (20 mg/mL total HA concentration) is too viscous and thick to inject in the tear trough, periorbital areas, or lips, but is an ideal product for the cheek and chin. Juvéderm Voluma has been approved for use in Canada but is still under clinical investigation in the United States. As one of the initial clinical investigators of this product in Canada, we have seen tremendous results in patients requiring volume replacement for facial rejuvenation. I find the product easy to inject (although it is noticeably firmer and requires more force to inject than traditional high molecular weight [MW] HAs), to sculpt, and to mold. It is well tolerated by patientsl as evidenced by their satisfaction with the product, likelihood of returning for additional treatment, and their willingness to recommend the product to others. However, I have injected into the lips and the patients have not felt it to be too stiff or firm. In previous years, Gore-Tex is been used to insert the lips for a permanent filler, and Gore-Tex is much firmer than Voluma. Patients enjoy the length of time Voluma lasts.
The added benefit of using an HA-based filler is that it can be dissolved out with hyaluronidase if the patient is unhappy with the product. In areas where inadvertent skin necrosis is a concern, hyaluronidase is essential in one’s armamentarium to treat such a disastrous complication.
Defects to be corrected by fillers as a first choice include the NLF, marionette and oral commissural lines, labiomental crease, mid-cheeks/zygoma, chin, lips, philtrum, ear lobe, tear trough, infrabrow, temporal depressions, and hands. In young Asian patients, we have used fillers to augment the dorsum of the nose, project the nasal tip, and occasionally have placed filler in the nasal spine to allow increased tip rotation. Fillers can also be used for dynamic wrinkles of the upper face and vertical wrinkles of the white lip, but are less effective that other rejuvenation techniques available such as Botox or laser resurfacing. Sites where fillers are not indicated would include nonextensible scars, neck and necklines, and sites of previously implanted permanent or unknown fillers. One exception to this would be our clinical experience of HA fillers in patients with previously implanted Bio-Alcamid (Polymekon, Brindisi, Italy), a nonabsorbable hydropolymer used as injectable permanent filler for cosmetic treatment and soft-tissue reconstructive defects.
Moran
With our enhanced understanding of the role of fillers in panfacial rejuvenation, the indication for fillers has expanded. Even without any significant changes in technology of the fillers themselves, we have a greater appreciation of their varied applications.
I use lighter, less viscous HA products near the surface of the skin and in tear troughs. I use heavier, more viscous products in the deeper subcutaneous layers and to augment fat and bony deficiencies in the face. I often layer them when appropriate.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree