Fig. 14.1
Pathomechanisms of atopic dermatitis. Environmental allergens trigger the overproduction of IgE antibody when they come in through barrier-disrupted sensitive, atopic skin, while chemical or bacterial factors induce epidermal keratinocytes and fibroblasts to release several cytokines and predispose the skin to inflammation. Uncontrolled scratching behavior due to aberrant itch perception in the central nervous system is closely related with barrier-disrupted sensitive skin and Th2-skewed allergic inflammation
14.2 Pathogenesis of Atopic Dermatitis
The skin of a patient with AD is often very susceptible to allergy and is called atopic skin [9]. Such vulnerability of the skin may result in a combination of genetic and environmental factors [10]. Environmental allergens trigger the overproduction of IgE antibody when they come in through the barrier-disrupted sensitive, atopic skin, while chemical or bacterial factors induce epidermal keratinocytes and fibroblasts to release several types of cytokines, which predisposes one to Th2-type skin inflammation. External ointments, cosmetics, and shampoos can also cause allergic contact dermatitis in some people, in addition to irritating atopic skin.
Recently, filaggrin (a keratohyalin granule-related small protein controlling skin barrier function) mutations have been reported to be closely associated with the risk of AD development and bronchial asthma in Icelandic and Japanese populations. Since the suggestion by Sulzberger that AD may be associated with the overproduction of IgE antibodies, IgE has been considered to play an important role in the pathogenesis of AD. However, the details of the involvement of IgE antibodies in the onset of AD remain unknown. Elevation of IgE levels in aggravated AD and elevated IgE titers in proportion to the disease duration in AD have been clinically observed. Experimental studies have demonstrated that FcεRl (+) Langerhans cells in the skin of patients with AD are more active in presenting antigens inducing the production of Th2 cytokines from T cells [11, 12]. These findings demonstrate that Th2 immune response with hyperproduction of IgE contributes to the development and progression of AD [13–15].
14.3 Animal Model of Atopic Dermatitis
To clarify this fundamental question, several AD models have been introduced, namely, the IgE transfer model [16]; topical sensitization models of protein antigens, such as ovalbumin [17]; Dermatophagoides-derived antigen (Dfb) [18]; or repeated hapten application models [19] and the spontaneous atopic dermatitis model [20, 21].
After the establishment of transgenic technology, several transgenic or knockout mouse lines expressing atopic dermatitis-like phenotypes have been reported with promising results. These include the Th2 or innate cytokine overexpression model [22–26], IgE transgenic model [27], and skin barrier-related protein-specific knockout mouse (Table 14.1) [28, 29]. We also previously reported a murine atopic eczema/dermatitis (AD)-model, with histopathological eczematous reactions [30–32]. Mast cells and inflammatory cells other than T cells are thought to play an important role in these IgE-mediated biphasic reactions [30]. However, it remains unclear as to whether Th2-type cytokines, IL-4, IL-5, or IL-13, play an essential role in the induction of the IgE-mediated reaction or if impaired skin barrier functions with abnormal itch perception play a more fundamental role in human atopic dermatitis.
Table 14.1
Summary of animal models of atopic dermatitis
Animal model | Authors | Year | Journals | Phenotypes |
|---|---|---|---|---|
Anti-DNP IgE-induced dermatitis | Ray MC et al. [16] | 1983 | J Immunol | Biphasic skin reactions |
Retinoic acid pretreatment and anti-DNP IgE-induced dermatitis | Katayama I et al. [30] | 1990 | Int Arch Allerg appl Immunol | Eczematous biphasic reactions |
IL-4 transgenic mouse model | Tepper RI et al. [22] | 1990 | Cell | Eczematous reaction |
Repeated hapten application model | Kitagaki H et al. [19] | 1995 | J Immunol | Th2-type skin reactions |
Ovalbumin-painted model | Spergel JM et al. [17] | 1998 | J Clin Invest | Eosinophilic dermatitis |
Keratinocyte-specific IL-18 transgenic mouse | Konshi H et al. [23] | 2002 | Proc Nat Acad Sci | Eczematous reaction |
TSLP transgene expression model | Yoo J [24] | 2005 | J Exp Med | Spontaneous atopic dermatitis |
IgE transgenic mouse | Mukai K [27] | 2006 | Immunity | Very-late-phase skin reaction |
Flaky tail mouse model | Moniaga CS [28] | 2010 | Am J Pathol | Human atopic dermatitis-like phenotype |
IL-33 keratinocyte-specific transgenic mouse | Imai Y [25] | 2013 | Proc Nat Acad Sci | Spontaneous itchy dermatitis |
Linoleic acid (LA)-fed mouse | Fuji M [29] | 2015 | Exp Derm | Atopic dermatitis-like pruritic inflammation |
Claudin-1 expression model in keratinocyte | Tokumasu R et al. [34] | 2016 | Proc Nat Acad Sci | Evolutional features of AD |
IL-23-injected model | Ewald DA et al. [26] | 2016 | JACI | Morphological features of AD |
Spontaneous occurrence of dermatitis | ||||
Canine atopic dermatitis | Butler JM et al. [21] | 1983 | Dermatitis, asthma | |
Nc/Nga mouse | Matsuda H et al. [20] | 1997 | Spontaneous pruritic dermatitis |
It has been reported that scratching behavior itself is induced by itch perception and truly represents the behavior triggered by itch-induced brain sensitization [32]. Shimada et al. reported that wiping of the face with the forepaws represents hair grooming and scratching with the hind paws represents behavioral response to itch perception in rodents (Fig. 14.2) [33]. Another point of discussion is whether IgE antibody itself induces spongiotic eczematous reactions occasionally observed in acute phases of human atopic dermatitis. To address this question, we reported the passive transfer model of monoclonal anti-IgE antibody in retinoic acid-treated mice, as described below [30]. It is also an important issue as to whether the evolution of skin manifestations are reproduced in animal models of atopic dermatitis. Recently, we reported that claudin-1 (CLDN1) knockout mice develop evolutional features of human AD (Fig. 14.2) [34]. To establish the evolution model of AD, we established several claudin-1 (CLDN1)-expressing mouse models. The survival rate at 8 weeks was over 80% for Cldn1+/+ and 0% for Cldn1−/− mice. Among the Cldn1 mutant mice, the Cldn1Δ//Δ and Cldn1Δ/- mice exhibited age-dependent changes in their skin appearance. Cldn1Δ/Δ and Cldn1Δ/- mice exhibited wrinkled skin at 1 week, abnormal dry hair at 2 weeks, and were nearly normal at 8 weeks. The Cldn1Δ/- mice, which usually did not survive beyond weaning, demonstrated more severe skin phenotypes, with severe desquamation and wrinkled skin at 2 wks; this phenotype had improved, but was still apparent at 8 wks, and only the Cldn1Δ/- mice, of all the mutant genotypes, still exhibited a different severity of skin lesions from WT at 8 wks by dermatitis score. The levels of CLDN1 were significantly lower in the skin of AD patients compared with controls. In skin samples from the trunk and limbs of human AD patients, after removing the outlier CLDN1 signals, we found that the number of macrophages was significantly inversely correlated with the CLDN1 signal. On the other hand, we found no correlation between the CLDN1 level and the thickness of the epidermis, the severity of AD by Eczema Area and Severity Index, or the serum level of defined AD factors including leukocytes, eosinophils, IgE, or thymus- and activation-regulated chemokine (TARC). Koomen et al. reported that mite antigens were more effectively presented by Langerhans cells that contained IgE antibodies on their surfaces [11]. Recent studies have reported that as FcεRI was not expressed on Langerhans cells of mice [35], their antigen presentation systems may be different from those in humans. Some reports demonstrated that mouse models using mite antigens are not appropriate for analyses of human atopic allergies [36].
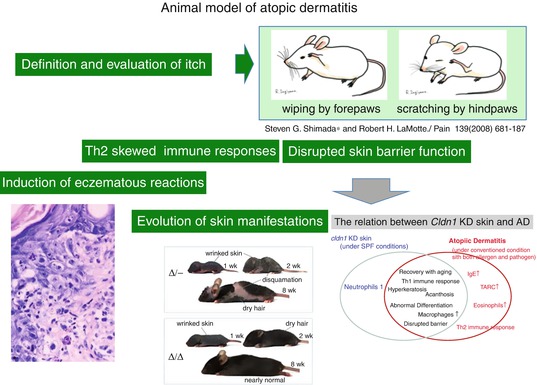

Fig. 14.2
Animal model of atopic dermatitis. Shimada et al. reported that wiping of the face with the forepaws represents hair grooming and scratching with the hind paws represents the behavioral response to itch perception in rodents [33]. Monoclonal anti-IgE antibody in retinoic acid-treated mice, as described below, induced spongiotic dermatitis mimicking human AD [30]. Claudin-1 knockout mice develop evolutional features of human AD [34]. The levels of CLDN1 were significantly lower in the skin of AD patients compared with controls
14.4 Induction of Eczema Reactions by Monoclonal (Anti-DNP) IgE Antibody
14.4.1 Skin Thickening Induced by Retinoic Acid and Eczema Induced by IgE Antibodies
It was reported that eczema reactions could be induced by mite antigen patch tests in human AD patients [37] and that eosinophilic eczema reactions are induced by mite-specific IgE antibodies. For the purpose of analyzing these reactions, the following experiment was performed: monoclonal (anti-DNP) IgE antibody was intravenously administered to BALB/c mice and then DNCB was applied on their auricles and skin reactions were observed. In this case, bimodal reactions were observed: one early reaction after 30 min, and a late reaction after 24 h. Histologically, only moderate edema and mild invasion of mononuclear cells were observed, which were thought to be a kind of LPR reaction. Epidermal reactions were mild as well. As the epidermal layers of mouse skin are only 1 or 2 layers, histopathological findings observed in contact dermatitis, such as spongiosis of the epidermis or small round cell infiltrations, are not observed in mice. Therefore, retinoic acid (vitamin A) was applied five times to make the mouse skin more similar to human skin. After 10 days from the final application, inflammation reactions had disappeared and only thickening of the epidermis was observed. At this time, epidermis layers had thickened to 4–5 layers from 1–2 layers, and tissue mast cells had increased to almost twice as many as usual. In this condition, it was possible to observe eczema reactions, such as spongiosis of the epidermis or small round cell infiltrations, and even degranulation of tissue mast cells and invasion of eosinophils were observed. These results demonstrated that IgE antibodies could induce edema reactions by themselves. From another viewpoint, it could also be interpreted that in cases of plaques only exhibiting skin thickening without inflammation, which is often observed in AD patients, edema reactions could easily be induced by the activation of mast cells.
It is generally thought that in seemingly normal skin tissues of atopic dermatitis patients, cell adhesion molecules, such as intercellular adhesion molecule-1 (ICAM-1), which are expressed in keratinocytes of the epidermis or vessel endothelial cells, or vascular cell adhesion molecule-1 (VCAM-1), which is generated in vessel endothelial cells, are easily induced by scratching or other causes to make the preliminary conditions for inflammation [38].
As expression of ICAM-1 was observed in keratinocytes of the epidermis for a relatively longer time during the pretreatment with retinoic acid, certain reactions that are generally observed in atopic dermatitis patients may have occurred on the skin of the retinoic acid pretreated mice.
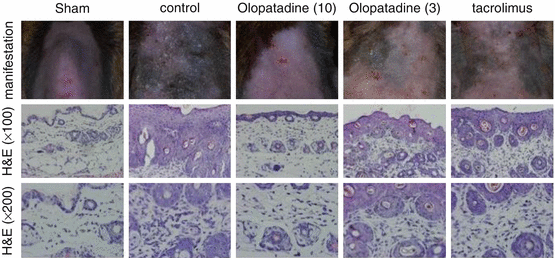
14.5 NC/Nga Mice Model Induced by Topical Dermatophagoides farinae Body (Dfb) Extract Application
After introduction of the spontaneous AD mouse model induced at conventional but not SPF conditions by Matsuda [20], the Nc/Nga mouse has been widely used for the analysis of pathomechanisms of AD or as a human AD model for the evaluation of newly developed anti-AD drugs. More recently, a mite antigen-induced AD model in Nc/Nga was reported at SPF conditions which enabled us to analyze more precise DP antigen-dependent skin reactions and to perform more accurate and convincing studies of the clinical effects of new drugs. For the application of this mouse model, we evaluated the effects of the anti-allergic drug, olopatadine, on scratching behavior induced by topical application of the mite allergen [39]. In daily practice, control of itch is an important issue in the treatment of AD. Itch is mediated by a variety of pruritogens, including histamine, and promoted by neurite outgrowth in the epidermis of AD patients, probably due to the release of nerve growth factor. Effects on mite antigen-induced skin manifestations in Nc/Nga mice are shown in Fig. 14.3. As shown, olopatadine markedly improved skin reactions due to decreased scratching behaviors (Fig. 14.4). Furthermore, olopatadine inhibited neurite infiltration into the epidermis, which may enhance itch perception (Fig. 14.5c, d) [39]. In contrast, topical tacrolimus markedly suppressed scratching behavior in this system, although neurite infiltration into the skin was observed (Fig. 14.5e). Recently, decreased production of the axonal guidance molecule semaphorin 3A in the lesional skin of AD resulting in increased epidermal innervation and itch perception was reported [40, 41]. Our study demonstrated that olopatadine robustly enhanced semaphorin 3A (Table 14.1). We also demonstrated that olopatadine decreased the pruritogenic cytokine [42], IL-31, expression in this Nc/Nga mouse [43]. IL-31 levels were significantly increased in mice that received Dfb application (n = 10) compared with sham-treated mice (n = 6). Olopatadine at 3 and 10 mg/kg/day (n = 10 each) significantly suppressed this increase in IL-31 levels by 88.1% and 94.5%, respectively. Tacrolimus ointment also significantly suppressed the increase in IL-31 production by 94.3%. In the sham-treated control and olopatadine-treated groups, IL-31 correlated positively with the tissue concentrations of several inflammatory and pruritus mediators including NGF, IL-1β, E-selectin, and amphiregulin (r = 0.7574, r = 0.7324, r = 0.8368, and r = 0.6970, respectively) [43] (Fig. 14.6, Table 14.2).