Key Points
- ▪
The surgical treatment of lymphedema has not been evaluated in randomized clinical trials; therefore, the level of current evidence does not justify the widespread adoption of surgery as the standard of care for lymphedema.
- ▪
Complete decongestive therapy remains the only lymphedema treatment that is supported at the highest level of evidence.
- ▪
Health-related quality of life is a multidimensional construct that encompasses physical, functional, emotional, and social well-being parameters.
- ▪
A variety of quality of life instruments have been used to capture outcomes in lymphedema patients, but some of these instruments were not sensitive to lymphedema-specific outcomes.
- ▪
Lymphedema-specific symptom assessment tools may be more sensitive than other instruments to clinically meaningful changes in patients with lymphedema.
Introduction
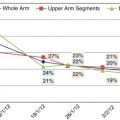
Lymphedema continues to be a significant health issue for cancer survivors, as shown by the 2006 and 2010 LiveStrong Cancer Survivor Surveys. Among the 6593 survivors of various types of cancer who completed the questionnaires, lymphedema was the sixth most-frequently reported physical issue; lymphedema was an increasing concern for 21% of respondents in 2006 and for 25% of respondents in 2010. Of the respondents, 53% had their cancer treated with surgery or radiation, with or without adjuvant systemic therapy.
Standard-of-care lymphedema treatment is a management-based program providing symptomatic relief but requiring lifelong compression, self-massage, and in some cases regular visits to a lymphedema therapist for bandaging, education, and manual lymphatic drainage. Although some surgical management techniques for the treatment of lymphedema offer patients an opportunity to forgo lifelong treatment and in some cases to forgo ongoing compression, little evidence exists for the long-term efficacy of these relatively novel procedures.
Because quality of life (QOL) outcomes have become a subject of increasing focus as primary or secondary outcomes in most clinical trials, a variety of general, malignancy-specific, and symptom-specific self-report instruments have been developed and used to assess QOL and symptoms in patients who have or are at risk for lymphedema. This chapter will provide an overview of the levels of evidence used in plastic surgery research, the published evidence for the surgical treatment of lymphedema, the assessment of patient-centered outcomes, and specific QOL and symptom assessment tools that have been used to evaluate patients with lymphedema.
Level of Evidence
Definitions
The Center for Evidence-Based Medicine has created a document that categorizes the levels of evidence available for current clinical practices. The highest level of evidence is level 1A, which is evidence derived from a systematic review of randomized controlled trials, and the lowest level of evidence is level 5, which is expert opinion without clinical basis. Each level has clearly defined criteria ( Table 22.1 ).
| Level | Evidence for Therapy/Treatment/Prevention |
|---|---|
| 1a | Systematic review with homogeneity of randomized controlled trials |
| 1b | Individual randomized controlled trials with narrow confidence intervals |
| 1c | All-or-none case series |
| 2a | Systematic review with homogeneity of cohort studies |
| 2b | Individual cohort study |
| 2c | Outcomes research; ecological studies |
| 3a | Systematic review with homogeneity of case studies |
| 3b | Individual case-control study |
| 4 | Case-series and poor-quality cohort or case-control studies |
| 5 | Expert opinion without explicit critical appraisal |
These levels of evidence have been modified to specifically address the levels of evidence used in plastic and reconstructive surgery articles. This modified schema first classifies articles as diagnostic, therapeutic, or prognostic and then further classifies them using the Center for Evidence-Based Medicine levels 1 through 5. The highest-level evidence is defined as evidence arising from a high-quality cohort study with adequate power or from a systematic review of multiple studies, and the lowest-level evidence is defined as expert opinion. The levels vary between the diagnostic, therapeutic, and prognostic-associated sub-classifications but share general characteristics. For therapeutic studies, the highest level of evidence comes from high-quality single- or multicenter randomized controlled trials with appropriate power; for diagnostic studies, the highest level of evidence comes from cohort studies validating a diagnostic test using a gold-standard test as a comparison; and for prognostic/risk studies, the highest level of evidence comes from prospective cohort or comparative analyses. In each category, a systematic review of the study types at the highest level for that category can also be classified as the highest level of evidence. The lowest level of evidence in each category comes from expert opinions or consensus documents based on the evaluation of case studies ( Table 22.2 ).
| Level | Therapeutic Studies | Diagnostic Studies | Prognostic/Risk Studies |
|---|---|---|---|
| I | High-quality, multicenter or single-center randomized controlled trial with adequate power | High-quality, multicenter or single-center cohort study validating a diagnostic test with a ‘gold’ standard as a reference in a series of consecutive patients | High-quality, multicenter prospective cohort or comparative study with adequate power |
| Systematic Review of the Above Studies Qualifies as Level I Evidence | |||
| II | Lesser-quality randomized controlled trial, prospective cohort or comparative study | Exploratory cohort study developing diagnostic criteria (with a ‘gold’ standard as a reference) in a series of consecutive patients | Lesser-quality prospective cohort or comparative study; retrospective cohort or comparative study; untreated controls from a randomized controlled trial |
| Systematic Review of the Above Studies Qualifies as Level II Evidence | |||
| III | Retrospective cohort or comparative study | Diagnostic study in non-consecutive patients without consistently applied ‘gold’ standard | Case-control study |
| Systematic Review of the Above Studies Qualifies as Level III Evidence | |||
| IV | Case series with pre/post-test or post-test only | Case-control study or any above-mentioned study without a ‘gold’ standard | Case series with pre/post-test or post-test only |
| V | Expert opinion developed via consensus; case report or clinical example; evidence based on physiology, bench research, or ‘first principles’ | Expert opinion developed via consensus; case report or clinical example; evidence based on physiology, bench research, or ‘first principles’ | Expert opinion developed via consensus; case report or clinical example; evidence based on physiology, bench research, or ‘first principles’ |
NonSurgical Treatment of Lymphedema
The traditional, gold-standard treatment for lymphedema remains complete decongestive therapy (CDT). In the treatment phase of CDT, trained lymphedema therapists apply multiple modalities including manual lymph drainage, multilayer short-stretch compression bandaging, therapeutic exercises, skin care, elastic compression, and education in self-maintenance. After the treatment phase, which usually lasts three to eight weeks, the patient is expected to continue into the maintenance phase, which includes self-administered lymphatic drainage, exercise, skin care, and compression garments.
A recent systematic review by Lasinksi et al. includes eight randomized controlled trials and 10 prospective cohort studies that examined the outcomes associated with CDT, placing this review at the highest level of evidence. This study utilized both data collected from studies and data described in previously published reviews. The authors concluded that those studies were moderately strong but lacked consistent, objectively collected data and that several of the studies that did describe consistent interventions often bundled CDT with other interventions. Findings from these data indicate that CDT is effective in reducing limb volume. However, many treatments are only evaluated as a bundled therapy (e.g., manual lymph drainage and bandaging) which makes it difficult to determine the overall efficacy of a single component of the CDT treatment schema.
Nonsurgical treatments other than CDT can be considered for the treatment of lymphedema. A systematic review of complementary, alternative, and other non-CDT lymphedema treatments found that treatments like these were widespread but poorly supported by evidence. In this review, the lymphedema therapies were categorized as either physical agents or modalities of contemporary value. The physical agent modalities included ultrasound therapy, electrically stimulated lymphatic drainage, high-voltage electrical stimulation, diathermy, low-level laser therapy, hyperbaric oxygen therapy, elastic taping, and acupuncture. Among the modalities of contemporary value were internationally designed at-home units devised to complete manual lymph drainage through circular massage motions, aqualymphatic therapy, and extracorporeal shockwave therapy. The evidence supporting the use of any of these therapies remains limited. Most of these studies were completed with a small convenience sample (level 2) or a case series (level 4) and have not been replicated. Also, very few of these studies used completely objective measures of limb volume reduction, such as circumference, perometry, water displacement, or bioimpedance; subjective measures including symptom assessment and clinical examination were used to identify the pre- and post-treatment changes which limit the validity of the data.
Surgical Treatment of Lymphedema
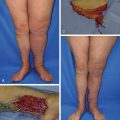
Surgical treatment of lymphedema has become more widely available and is used primarily in the treatment of chronic, progressive lymphedema that remains refractory to standard treatment protocols. Because of the paucity of published high-level studies evaluating the long-term outcomes following surgical treatment of lymphedema, this type of treatment remains controversial and is considered experimental in many countries, including the US.
In 2011, a systematic review on the surgical treatment of lymphedema cited a lack of evidence due primarily to small patient populations, failure to use objective measurement criteria for limb volume change, and a lack of long-term follow-up. Summaries of the literature included in the systematic review have been updated through February 2014 and are presented in Tables 22.3–22.6 . Subsequent systematic reviews have been published evaluating the efficacy of microsurgery and tissue transplantation in treating lymphedema; these reviews have found mixed outcome results.
| Author (Year) | Study Design | Number of Patients | Lymphedema Site | Specific Surgical Procedure | Follow-Up Time, Months | Measurement Technique | Volume Reduction, % | QOL Tool Used |
|---|---|---|---|---|---|---|---|---|
| Kim et al. (2004) | Retrospective | 20 | Lower extremity | Excision | 17.8 | Volumeter | 16% | None |
| Modolin et al. (2006) | Prospective | 17 | Penis/scrotum | Excision | 72 | NR | NR | None |
| Lee et al. (2008) | Retrospective | 22 | Lower extremity | Excision | 48 | Infrared optometric volumetry and circumference | NR | Mentions QOL but no instrument |
| Salgado et al. (2009) | Prospective | 11 | Upper extremity | Excision with preservation of perforators | 17.8 | Circumference | 21 | None |
| van der Walt et al. (2009) | Retrospective | 8 | Lower extremity | Modified Charles procedure | 27 | NR | 8.5 kg | None |
| Karri et al. (2011) | Retrospective | 27 | Lower extremity | Charles procedure | 48 | NR | NR | None |
| Author (Year) | Study Design | Number of Patients | Lymphedema Site | Specific Surgical Procedure | Follow-Up Time, Months | Measurement Technique | Volume Reduction, % |
|---|---|---|---|---|---|---|---|
| Liu et al. (2005) | Prospective | 11 | Upper extremity | Liposuction | ∗ | Circumference | ∗ |
| Brorson et al. (2006) | Prospective | 35 | Upper extremity | Liposuction | 12 | Water displacement | 103 |
| Qi et al. (2009) | Prospective | 11 | Upper extremity | Liposuction, myocutaneous flap transfer | 26 | Circumference | 18 |
| Damstra et al. (2009) | Prospective | 37 | Upper extremity | Suction-assisted lipectomy | 12 | Water displacement | 118 |
| Schaverien et al. (2012) | Prospective | 12 | Upper extremity | Liposuction | 36 | Water displacement | 123 |
| Granzow ∗∗ (2014) | Retrospective | 10 | Upper extremity (n=6), lower extremity (n=4) | Suction-assisted lipectomy | 32 | Circumference | 111% (UE), 87% (LE) |
∗∗ Study included a discussion of patients who had tissue transfer and lymphatic venous anastomosis.
| Author (Year) | Study Design | Number of Patients | Lymphedema Site | Specific Surgical Procedure | Follow-Up Time, Months | Measurement Technique | Volume Reduction, % |
|---|---|---|---|---|---|---|---|
| Weiss et al. (2002) | Prospective | 12 | Upper extremity | Autologous lymphatic tissue transplantation | 96 | Circumference | 22 to 31 |
| Wongtrungkapun (2004) | Prospective | 10 | Lower extremity | Lymphonodovenous implantation | 4.5 | Circumference | 3.5 cm at knee, 7.37 cm at 16 cm below knee, 2.75 at metatarsal level |
| Becker et al. (2006) | Retrospective | 24 | Upper extremity | Lymph node transplantation | 96 | Circumference | Reduction to normal (n=10), some reduction (n=10), no change (n=2) |
| Belcaro et al. (2008) | Retrospective case-control | 9 | Lower extremity | Autologous lymphatic tissue transplantation (n=9) vs. control (n=8) | 120 | Water displacement | Increase of 13 |
| Hou et al. (2008) | Randomized controlled trial | 15 | Upper extremity | Autologous bone marrow stromal cell transplantation (n=15) vs. CDT (n=35) | 12 | Circumference | 81 |
| Lin et al. (2009) | Retrospective | 13 | Upper extremity | Vascularized lymph node transfer | 56 | Circumference | 51 |
| Gharb et al. (2011) | Prospective | 21 | Upper extremity | Vascularized lymph node transfer | 40 | Circumference | NR |
| Cheng et al. (2012) | Prospective | 6 | Lower extremity | Vascularized lymph node transfer | 8.7 | Circumference | 64% above knee, 64% below knee, 67% above ankle |
| Cheng et al. (2013) | Prospective | 10 | Upper extremity | Vascularized lymph node transfer | 39 | Circumference | 40 |
| Author (Year) | Study Design | Number of Patients | Lymphedema Site | Follow-Up Time, Months | Measurement Technique | Volume Reduction, % |
|---|---|---|---|---|---|---|
| Koshima et al. (2004) | Retrospective | 52 | Lower extremity | 15 | Water displacement | 42 |
| Matsubara et al. (2006) | Retrospective | 9 | Lower extremity | 21 to 87 | Circumference | >5 cm (n=6), 2 cm (n=2), no effect (n=3) |
| Damstra et al. (2009) | Prospective | 10 | Upper extremity | 12 | Water displacement | 2 |
| Demirtas et al. (2009) | Retrospective | 42 | Lower extremity | 12 | Circumference | 59 |
| Campisi et al. (2010) ∗ | Retrospective | 1800 | Upper and lower extremities | 120 | Water displacement | 56 (83% with 67% reduction) |
| Chang (2010) | Prospective | 20 | Upper extremity | 18 | Perometer | 35 |
| Maegawa et al. (2010) | Retrospective | 111 | Lower extremity | NR | Circumference | Percentage not reported average 872 ml reduction |
| Mihara et al. (2010) | Retrospective | 11 | Lower extremity | 23.6 | Circumference | 92 |
| Narushima et al. (2010) | Prospective | 14 | Upper extremity (n=2), lower extremity (n=12) | 809 | Circumference | 11 |
| Furukawa et al. (2011) | Prospective | 9 | Upper extremity | 17 | Circumference | >50 in 77.8% of patients |
| Yamamoto et al. (2011) | Retrospective | 20 | Lower extremity | 8.9 | Circumference | 11 |
| Auba et al. (2012) | Prospective | 12 | Upper extremity (n=7), lower extremity (n=5) | 24 | Circumference | 1.18 cm circumference |
| Mihara et al. (2012) | Prospective | 6 | Lower extremity | 10 | Circumference | NR |
| Ayestaray et al. (2013) | Prospective | 4 | Head and neck | 12 | Circumference | 3.7 |
| Boccardo et al. (2013) | Retrospective | 23 ∗∗ | Lower extremity | 42 | Circumference | 80 |
| Chang et al. (2013) | Prospective | 100 | Upper extremity (n=89), lower extremity (n=11) | 12 to 36 | Perometer | 42 (upper extremity) 7 to 42 (lower extremity) |
∗ Selected among duplicate studies with overlapping patients.
∗∗ Study included patients treated in a preventative manner, lymphedema patients included.
Surgical intervention for lymphedema has been widely reported in the literature, but only two such studies were found to use any patient-reported QOL metrics. Among reports of excisional surgical procedures to treat lymphedema, only one study reported any assessment of QOL; however, this study did not report what instrument was used. In a study of 35 patients who received liposuction for the treatment of chronic lymphedema, QOL was evaluated using the Psychological General Well-Being Index (22 items) and the Hospital Anxiety and Depression Scale (14 items). Six months after liposuction, the patients’ total scores and general health scores in the Psychological Well-Being Index had improved since baseline measurements, but this change was not statistically significant. Scores on the Hospital Anxiety and Depression Scale decreased, although the change was not statistically significant, at six months after the surgery but returned to their baseline levels at 12 months after the surgery.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree