This review provides a thorough and clear discussion on the outcomes of stem cells in treating chronic wounds. With recent technological developments that now allow isolation and culture of stem cells, researchers are able to perform vigorous studies on somatic or adult stem cells. Human and animal stem cell studies are discussed with a focus on the basic process of stem cells in wound healing and the authors’ first-hand clinical experience with stem cells used for chronic wound healing.
- •
Stem cell biology and application in wound healing is widely studied for bone marrow-derived stem cells.
- •
Adipose-derived stem cells (ADSCs) are more easily obtained from the donor sites.
- •
In chronic radiation injury, a mixture of ADSC and aspirated adipose tissue may play a pivotal role in wound healing.
Introduction
Wound Healing Process
The wound healing process contains well-organized and integrated patterns of complex biologic, molecular, and gene involvement events of
- •
Cell proliferation
- •
Cell migration
- •
Differentiation capacity of cells
- •
Extracellular matrix (ECM) deposition and degradation.
Within seconds, minutes, and hours after injury, epidermal and dermal cell migration, proliferation, and differentiation initiate re-epithelialization. External injury may trigger tissue hypoxia, which leads to upregulation of local growth factors (cytokines), degradation of ECM, and may ultimately lead to angiogenesis. Newly formed granulation tissue must be sustained to establish the formation of new blood vessels. Many diseases are related to keratinocyte or epidermal cell loss, angiogenesis dysfunction, and incomplete skin regeneration. These disorders including Buerger disease, chronic radiation wounds, and other chronic wound healing problems are under clinical investigation for possible new therapeutic modalities. Autologous adipose-derived stem cells (ADSCs) show great promise as an adjunct in facilitating the healing of these complex wounds.
Pathogenesis of Intractable or Nonhealing Wounds
Wound healing in skin comprises 4 major distinctive, but overlapping phases:
- 1.
Hemostasis
- 2.
Inflammatory
- 3.
Proliferation
- 4.
Remodeling.
In a chronic wound, remarkable changes related to senescence, ischemia, matrix biology, and bacterial colonization transform the normal progression of wound healing into a self-continuing cycle of inflammation and injury. Injured tissue induces tissue hypoxia, which leads to upregulation of growth factors; degradation of ECM activates local angiogenesis. Stem cells have shown to promote angiogenic processes. Stem cells also may be differentiated into various cells including fibroblasts, which are major component of dermis.
Chronic wounds include venous ulcers, pressure ulcers, and diabetic ulcers, which may account for 90% of all nonhealing wounds.
Venous ulcers are highly prevalent in woman and are related to circulatory insufficiency and venous hypertension located in and around the malleolus.
Pressure sores commonly occur in patients with impaired mobility and under prolonged medical and surgical care, such as extensive surgery, time spent in an intensive care unit, and poor systemic nutrition status. Pressure ulcers arise from external force, friction, and shear forces that affect segmental and perforator arteries leading to tissue necrosis particularly over bony prominences.
Diabetic ulcers result in long-term complications in diabetic patients. Most ulcers in patients with diabetes are located in pressure points of the foot and can arise from peripheral neuropathy, foot deformity, and local minor trauma. In a diabetic wound, ischemia can result from a macrovascular, microvascular, and neuropathic disease, or all the aforementioned.
Chronic wounds are complicated and etiologically multifactorial. Ischemia-reperfusion injury may play a major and important role in etiology. Ischemic stresses are altered in elderly people, partly because of the combination of ischemia and oxidant stresses in the ischemia-reperfusion injury. Negative pressure wound therapy may prevent systemic damage caused by ischemic reperfusion, as demonstrated in a pig model.
Wound healing contains multifactorial local and systemic factors. Wound bed preparation involves management of the following parameters :
- •
Tissue viability
- •
Involvement of infection
- •
Moisture balance
- •
Wound edges.
In addition, local alteration of metalloproteinases, integrins, chemokines, and growth factors on the wound surface must be controlled.
Treatment of Chronic Wounds
Treatment of chronic wounds is affected by optimal environmental conditions, the healing process, and subsequent scarring. Surgical or nonsurgical debridement, reduction of edema, diminishing bacterial burdens, maintaining the favorable moisture balance, and removal of undermining tissues are included in standard wound care.
Wound characteristics can be modified with a variety of recently developed topical dressings such as impregnated gauze, film, hydrogel, hydrocolloid, and alginate. Advanced therapies include mechanical devices providing topical negative pressure and bioengineered tissues of various types. Even though vigorous development of such materials, devices, and products are underway, surgical debridement is still required to facilitate wound healing. Debridement can reduce the bioburden of a wound selectively and effectively. Necrotic tissue, which must be eliminated because it behaves as a substrate for proliferating bacteria that strives for the same nutrients and oxygen molecules essential for wound healing, is best removed with debridement.
Stem Cells in Wound Healing
Stem cells are able to
- •
Undergo self-renewal
- •
Be cell proliferative
- •
Differentiate into multiple lineages of cell and tissue phenotypes.
Recent technological developments now allow isolation and culture of stem cells, which has enabled researchers to perform vigorous studies on somatic or adult stem cells.
Adult stem cells can differentiate into adipogenic, osteogenic, and neurogenic cell types. The autologous adult stem cells are readily available from the patient, thus not affected by ethical and immunologic barriers. Mesenchymal stem cells are first isolated from bone marrow and extend to almost all organs and tissues. Bone marrow-derived stem cells (BMMSCs) from a single donor can differentiate into liver epithelium, lung, gastrointestinal tract, and skin. BMMSCs are relatively easily obtained from bone-marrow aspirates of donor patients. BMMSCs are isolated and expanded in vitro by subculturing. Human BMMSCs (hBMMSCs) are resistant to 20 Gy radiation in vitro; immediately after exposure to 20 Gy radiations and reconstruction with hBMMSCs and angiogenic growth factor (basic fibroblast growth factor, [bFGF]), artificial dermis demonstrated improved wound healing in 10 days.
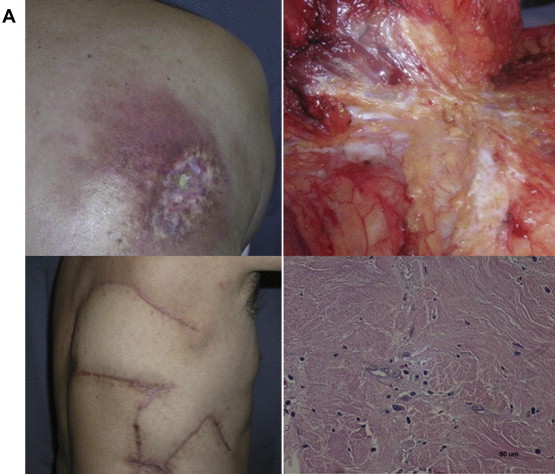
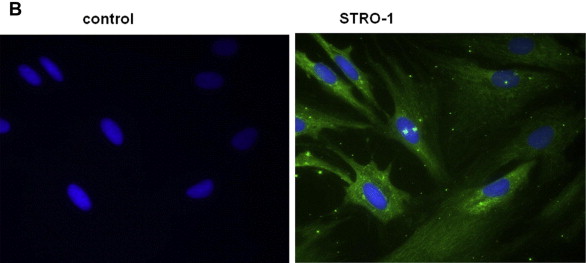
A 58-year-old male patient had received 45 Gy of radiation and subsequently developed chest pain. He then underwent percutaneous transluminal coronary angioplasty under fluoroscopy, which was unexpectedly prolonged as a result of intraoperative complications. Immediately after the procedure, the patient noticed strong, deep, penetrating pain in his back. He subsequently developed a localized wound deep into the costal cartilage and a surrounding erythematic lesion, which was apparent at first follow-up surgical visit. The wound was widely excised including the necrotic cartilage and was reconstructed with a bilobed latissimus dorsi musculocutaneous flap. The scar tissue resulting from exposure to original radiation was further analyzed via histologic analysis. Small spindle-shaped cells were concentrated within the fibrotic tissue and Stro-1, which is one of the immunoreactive markers of mesenchymal stem cells. This finding suggested that stem cells may mediate a radiation injury ( Fig. 1 ). FLOAT NOT FOUND
BMMSCs can synthesize higher amount of collagen, fibroblast growth factor (FGF), and vascular endothelial growth factor (VEGF), suggesting a potential benefit in accelerating wound healing. hBMMSCs are able to accelerate wound healing with a collagen sponge with bFGF. Over a 1-year observation of autologous bone marrow cell treatment for 3 nonhealing wounds, which previously failed to heal with the use of bioengineered skin or skin grafting, complete closure and dermal rebuilding in all cases was demonstrated with autologous bone marrow cell. Reduced scarring and engraftment of grafted cells was observed. Fibrin polymer spray, in which fibrinogen contains cultured autologous BMMSCs, can adhere to the wound bed and retain the cell viability and migration. Cultured BMMSCs are used for severe radiation-associated wounds with numerical dosimetry-guided surgery followed up to 11 months. Also, cultured BMMSCs are effective for intractable wounds when soaked in an artificial dermis. In addition, direct injection of cultured BMMSCs into the wound area in normal diabetic mice promoted wound healing through the release of proangiogenic factors on differentiation ; In streptozocin (STX)-induced diabetic rats, BMMSC double-system injections through the tail vein and local injection at the wound site augmented growth factors such as EGF, VEGF, platelet-derived growth factor-BB markedly and transforming growth factor-β and keratinocyte growth factor (KGF) moderately. BMMSCs induce potent differentiation and proliferation under low oxygen tension with cell morphologic and cell cycle changes and differentiation capacity toward bone and fat.
Furthermore, hBMMSCs transplantation to an adult rabbit incisional wound demonstrated tensile strength 80 days after surgery. Systemic administration of rat BMMSCs for 4 days promotes wound healing, increases production of collagen, rapid maturation of wound, and increases the tensile strength.
Stem cells are found in very low amounts among the nucleated cells from the bone marrow and even lesser amounts in the peripheral blood. Fibrocytes originate in bone marrow and are found in peripheral blood, wounds, and tissue remodeling. A recent study of peripheral blood demonstrated that fibrocytes are able to proliferate cells, and induce re-epithelialization and angiogenesis in a diabetic mouse.
ADSCs
Fat tissues contain multipotent cells, with similar wound healing effects of BMMSCs, stimulating human dermal fibroblasts. They have the capacity to differentiate into adipogenic, chondrogenic, myogenic, and osteogenic cell lineage when cultured in vitro with similar cell surface cluster of differentiation antigens. ADSCs are obtained from either liposuction procedure or solid fat tissue. ADSCs are called adipose-derived regenerative cells because they contain heterogeneous inducible regenerative cells. ADSCs are more easily obtained from the donor sites than BMMC and are extensively proliferative ex vivo in vitro. Fat tissue plays an important role in regulating energy balance and substance metabolism, and its biologic function in terms of metabolism, hormone, and signaling are varying. In vitro, assay depending on the anatomic location of the donor sites, age, and gender may differ in yielding and differentiation capacity. ADSCs are approximately 5000 CFU-F per gram of adipose tissue whereas the estimated CFU-F per milliliter of bone marrow is 100 to 1000. Comparing lipoaspirate and excised adipose tissues, aspirated adipose tissue yields more preadipocytes within 60 minutes after extraction and at 24 hours storage at 4° than extracted tissue. Although distinction of preadipocytes from ADSCs via lipoaspiration is still unknown, it is acknowledged that preadipocytes can be used for fat-based regeneration. There are several reasons that ADSCs are most favorable cell sources for regeneration:
- •
The lipoaspiration procedure is relatively common for surgeons.
- •
ADSCs contain superior potentials to induce both angiogenesis and vasculogenesis.
- •
In vivo studies prove the efficacy and effectiveness using ADSCs.
- •
ADSCs are easily cultured and have high-affinity with three-dimensional scaffolds and other cells.
ADSCs in Wound Healing
ADSCs are available for promoting angiogenesis, secreting growth factors and cytokines, and differentiating into multiple cell types on stimuli. ADSCs can promote human dermal fibroblast proliferation by directly contacting cells and paracrine activation in re-epithelialization phase of wound healing. Two-dimensional gel electrophoretic gel proteomic analysis of the intracellular protein of BMMSCs and ADSCs revealed that the proteins were similar, which suggests that ADSCs can replace BMMSCs in cell therapy. In mice, wounds healed faster when treated with ADSCs than with normal process. In nonirradiated and locally irradiated wounds in mouse, ADSCs can be differentiated to keratinocytes and produce KGF as well as vascular endothelial growth factor (VEGF). ADSCs can release angiogenic factors in ischemic injury. ADSCs in acellular dermal matrix provide a framework for support of the regenerative capacity in wound healing. With appropriate scaffolds, ADSCs can cause regeneration and induce wound healing. ADSCs with skin substitute containing human ECM can produce subcutaneous, dermal, and epidermal regenerated tissues. Using clinically available human acellular dermal matrix of cadaveric donors, ADSCs demonstrated accelerated wound healing by 7 days postoperatively and microvascular endothelial phenotype for 2 weeks, indicating the direct vascular networking in tissue regeneration with no systemic distribution other than surgical engrafted sites.
In a diabetic mouse model, noncultured excised ADSCs with commercially available bilayer artificial dermis can secrete several growth factors and cytokines for 4 days in cell culture supernatants, and histologic examination demonstrated advanced granulation tissue formation, capillary formation, and epithelialization for 2 weeks postoperatively.
In a pig multiple full-thickness wound model, cultured ADSCs with platelet-rich plasma can enhance the wound healing process in terms of VEGF concentration in the fluid and cosmetic appearance. ADSCs are able to demonstrate antioxidant effect via dermal fibroblasts and keratinocytes in a paracrine manner, and hypoxia increases VEGF and bFGF levels as well as induces cell proliferation.
Adipose-derived regenerative cell therapy for chronic radiation injury
Background of Adipose-Derived Regenerative Cell Therapy
Although literature emphasizes the merit and advantage of the use of ADSCs or adipose-derived regenerative cells (ADRCs) in wound healing, at present, there are few clinical applications. Autologous cultured BMMSCs are now being used to treat severe radiation injury. The purified autologous lipoaspirates are injected to improve radiation ulcers.
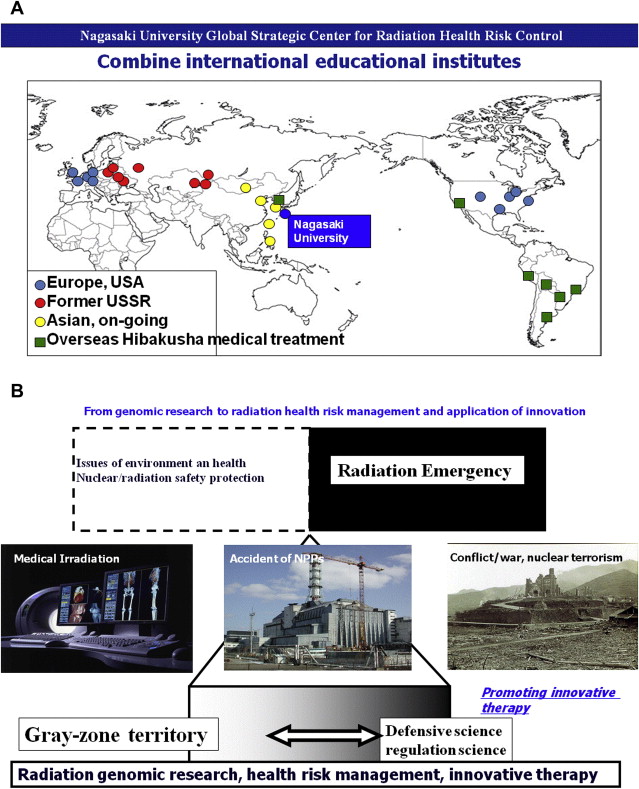
Nagasaki University Global Strategic Center for Radiation Health Risk Control
This clinical study project is designed under Nagasaki University Global Strategic Center for Radiation Health Risk Control, which is one of the global center-of-excellence programs funded by the Ministry of Education, Culture, Sports and Technology from 2007 to 2011. The aim of this program is to focus on global assessment of radiation and effect to health risk, to overcome the legacy of atomic bombing, to establish the scientific basis of human safety to radiation, and to promote international collaboration that will nurture the experts in this field. To accomplish these aims 3 research projects are set:
- 1.
Radiation and nuclear basic life science research
- 2.
Atomic bomb disease medical research
- 3.
International radiation health science research.
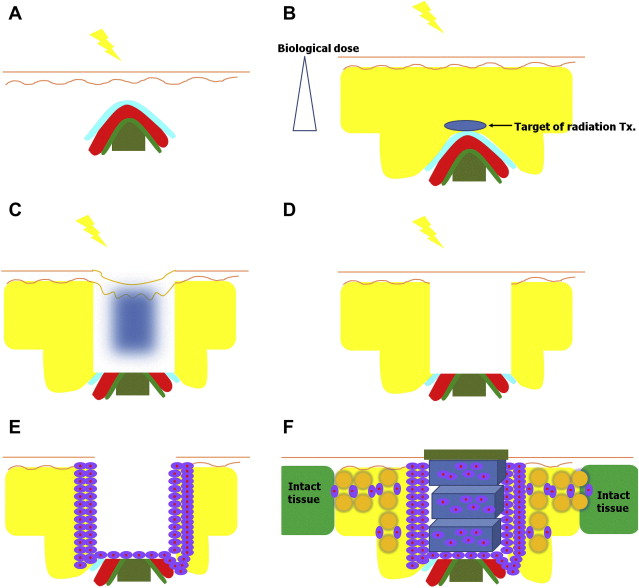
The goal of this project was to establish the scientific basis of radiation health risk control to contribute to society through Hibaku/Hibakusha (radiation-exposed) medicine. Radiation based life science research investigates external and internal irradiation, mechanisms of chronic low-dose radiation, risk assessment and management, susceptibility, and racial difference in individual risk assessment. Atomic bomb disease medical research deals with aging of the atomic bomb survivors who have progressive cancer, multiple cancer, and psychosomatic effects and also treating foreign Hibakusha atomic bomb survivors. International radiation health science research handles broad aspects of threat of nuclear events, global nuclear power plant (NPP) accidents and disasters, and world health organization-radiation emergency medical assistance and networking (WHO-REMPAN) collaboration center activity. Through WHO-REMPAN collaboration center, development and clinical application of regenerative medicine is further promoted ( Fig. 2 ). Because acute systemic radiation injuries are rare and unpredictable, the possibility of applying patients’ own fat tissue–derived stem cell therapy of their subcutaneous tissues not exposed to radiation for chronic local radiation injury is attempted.
Patient Enrollment
Five clinical treatment experiences with autologous noncultured ADSCs for more than 18 months, which is considered the final phase of wound healing remodeling process, mean patients’ follow-up period was 2 years ± 6 months (2 years 10 months to 1 year 9 months) postoperatively. All 5 patients, who were women, healed uneventfully, average healing 8 ± 2.2 weeks, among which 1 patient was treated for her consistent pain in her left toes as a result of thromboangiitis due to Buerger disease. Mean age of the patients were 64.4 ± 22.0. There is no recurrence or abnormal wound healing during follow-up periods in any of the patients.
Surgical Procedure, Scaffold (Artificial Dermis), and Growth Factor
In adipose-derived stem cell transplantation and postoperative management, an artificial dermis (Terudermis, Olympus-Terumo Biomaterials Corp, Ltd, Japan) was used as a scaffold. Terudermis comprises 2 layers: a lower layer of bovine atelocollagen and an upper layer comprising a silicone sheet, which protects against infection and dryness from the outside.
- •
After minimum debridement, Terudermis was multilayered and stacked over freshly debrided wounds.
- •
The silicone sheets were removed except top Terudermis.
- •
Two-thirds of isolated ADSCs were injected around the debrided wound at the base of the wounds, and into Terudermis.
- •
Another one-third of ADSCs was mixed with the autologous adipose tissue, which was rinsed with a lactated Ringer solution.
- •
In the Celution system, after isolating ADSCs, the disposable cell collection plastic case was again used to mix the suctioned fat, which is rinsed separately in the 50-cc syringe and repeated until the oil droplets are removed.
- •
After being mixed, it was injected into a zone of hard fibrotic tissue around the debrided wounds in 2-cm width in all directions.
Detailed surgical procedure is described in the literature. Briefly, surgical debridement is limited to the most severely affected area in depth and in width. The cells are injected at the edges and the base, evenly washed fat tissue mixed with cells are injected to bridge between the wound and intact tissue ( Fig. 3 ).