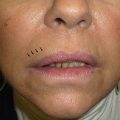
*Of note, the mineralocorticoid axis (renin-angiotensin-aldosterone) is almost NEVER suppressed in “exogenous adrenal insufficiency” → almost never get true adrenal (Addisonian) crisis with hypotension, coma.
• Basics: hypothalamus releases corticotropin releasing factor (CRH) → anterior pituitary releases ACTH → adrenal glands release cortisol
• HPA axis (CRH → ACTH → cortisol) is suppressed by use of exogenous CS
■ Hypothalamus: first to be suppressed, but quickest to recover
■ Adrenals: last to be suppressed, but slowest to recover
• Mineralocorticoid axis (renin-angiotensin-aldosterone) is NOT suppressed by exogenous CS used in dermatology → true adrenal (Addisonian) crisis w/ severe hypotension and coma is extremely uncommon in secondary exogenous adrenal insufficiency, because of the preserved MC axis function
• Exogenous adrenal insufficiency (HPA axis suppression) typically seen in patients taking pharmacologic CS doses for ≥3 to 4 weeks
■ Abrupt cessation of CS (always taper if CS course is >4 weeks)
■ Major stressor (surgery, trauma, or illness)
■ Daily dose given at any time other than the morning
• QOD (alternate day) dosing → ↓risk of nearly all major complications
■ ↓risk of: HPA axis suppression, growth suppression, HTN, opportunistic infections, and electrolyte disturbances
■ Does not lower risk of: cataracts or osteoporosis
• Two clinical presentations of exogenous adrenal insufficiency:
■ Steroid withdrawal syndrome (SWS): most common presentation; presents with (p/w) arthralgias, myalgias, mood changes, headache, fatigue, and anorexia/nausea/vomiting; no change in serum cortisol level, but rather ↓available intracellular CS
■ Adrenal (Addisonian) crisis: extremely uncommon; life-threatening; p/w symptoms of SWS + hypotension, ↓↓↓cortisol levels
Bone effects
• Osteoporosis: QOD dosing does NOT ↓risk; consider calcium + vitamin D and/or bisphosphonates, teriparatide, nasal calcitonin; greatest reduction in bone mass occurs in first 6 months; ↑fracture risk in postmenopausal women; greatest absolute loss of bone mass occurs in young men (they have highest baseline bone mass)
• Osteonecrosis: usually at least 2 to 3 month courses; proximal femur most common
Contraindications
Systemic fungal infections, herpes simplex keratitis, and hypersensitivity reactions
Pregnancy
Category C, but likely safe for short courses if needed (severe PUPPP or gestational pemphigoid, for example)
Clinical use
Systemic steroids are used in autoimmune bullous dermatoses, connective tissue disorders (treatment of choice in dermatomyositis), vasculitides, neutrophilic dermatoses, allergic contact dermatitis, papulosquamous dermatoses, and various other dermatoses
• Pemphigus: start at 1 mg/kg daily in divided doses and increase up to 2 mg/kg daily (if needed) for 4 to 6 weeks, consolidate dose to once a day and taper quickly to 40 mg daily, and slowly thereafter; a steroid-sparing agent should either be started at the get-go or before tapering
• Toxicodendron dermatitis: be careful not to stop oral CS too early because of the ↑likelihood of flare; best option is a 3 week tapering course starting at about 1 mg/kg daily
• Note that oral CS ↓acute pain in herpes zoster, but likely do not prevent postherpetic neuralgia
• Longer duration of treatment = ↑SE risk
• Divided dose regimens are more effective, but have a higher risk of SEs than single dose regimens (best taken in AM to simulate body’s diurnal variation of cortisol production)
• Alternate day (QOD) dosing: the antiinflammatory effects of CS last longer than the HPA axis suppressive effects → QOD dosing helps maintain control of disease activity after course with daily CS
Intramuscular CS
• Unique adverse effects: cold abscesses, subcutaneous fat atrophy, crystal deposition, menstrual irregularities, and purpura
• Main advantages (vs oral CS): compliance, can be given in setting of nausea/vomiting
• Main disadvantages (vs oral CS): ↑HPA axis suppression because levels are constant throughout the day (↑frequency of IM injections → ↑risk of HPA axis suppression), and less ability to precisely taper
■ Per Wolverton, do not use long-acting IM CS (such as Kenalog) more than 3 to 4×/year
Topical CS
• Most commonly used in dermatology for various conditions including dermatitis and psoriasis
• Of note, more potent topical steroids (e.g., clobetasol) and those in more highly absorbed bases (e.g., gels and ointments) are more likely to → adverse cutaneous effects
■ Remember that topical steroids do not usually cause systemic symptoms!
Monitoring
Consider monitoring:
• Fasting glucose levels, blood pressure (mild ↑ is ok), triglycerides, weight, height/weight for children, DEXA scans (T score < −2.5 = osteoporosis), MRI if pain in hip/shoulder/knee (osteonecrosis), and slit-lamp examination q6–12 months
• TB screening and chest X-ray
• Tests to evaluate adrenal insufficiency
■ AM cortisol: primary screening tool, >10 mcg/dL = good basal adrenal function
■ 24 hour urine free cortisol: more accurate test for basal adrenal function (advantage); main disadvantage is patient compliance with 24 hour urine collection
■ ACTH stimulation: most commonly used provocative test for adrenal function; check basal cortisol level → then inject ACTH → check cortisol levels at 30 and 60 minutes
■ Others: insulin hypoglycemia, metyrapone, and corticotropin-releasing factor
2.4 Immunomodulatory agents
Janus kinase inhibitors
Tofacitinib
• FDA approved for moderate to severe rheumatoid arthritis (RA) patients who have failed MTX
• Topical and oral have been tested in psoriasis; reports of oral used in alopecia areata
• Most common SEs: URI, mild headaches, and nausea
• May have ↓hemoglobin and mean neutrophil count, but usually normalize on treatment
• May have ↑LDL, HDL, CK, TGs, and LFTs
Azathioprine
Mechanism of action
• Azathioprine’s active metabolite, 6-TG (thioguanine), is produced by the hypoxanthine guanine phosphoribosyltransferase (HGPRT) pathway and shares similarities with endogenous purines → therefore, it gets incorporated into DNA and RNA → inhibits purine metabolism and cell division (particularly in fast-growing cells that do not have a salvage pathway, like lymphocytes)
• Xanthine oxidase and thiopurine methyltransferase (TPMT) convert azathioprine into inactive metabolites
• Diminishes T-cell function and antibody production by B-cells
Important pharmacology points
• TPMT activity is reduced in certain populations, and functional enzyme allele sequencing is available
■ ↓activity of TPMT (measured by allele activity) or ↓xanthine oxidase (as a result of allopurinol or febuxostat) → ↑azathioprine levels → ↑risk of life-threatening myelosuppression
■ ACE inhibitors, sulfasalazine, and concomitant use of folate antagonists also increases risk of myelosuppression
• Azathioprine may decrease anticoagulant effects of warfarin and reverse neuromuscular blockade
Side effects
• Leukopenia, thrombocytopenia, and immunosuppression (correlates with low TPMT activity)
• Squamous cell carcinoma (SCC) and lymphoma (particularly non-Hodgkin’s B-cell lymphoma)
■ No clear evidence of increased risk for dermatologically dosed indications
• Infection (particularly human papilloma virus, herpes simplex, and scabies)
• Hypersensitivity syndrome (usually between first and fourth week of therapy and more common in patients who are receiving concomitant cyclosporine or MTX)
• Gastrointestinal SEs – most common adverse effect of azathioprine – include nausea, vomiting, and diarrhea (often present between first and tenth day of therapy); also gastritis and pancreatitis
• Transaminase elevation and severe hepatocellular toxicity are rare
Important monitoring points
• Baseline pregnancy test (for women of childbearing potential; pregnancy category D) and tuberculin skin test (strongly consider performing depending on clinical situation)
• Annual complete physical examination with particular attention to possible lymphoma and squamous cell carcinoma
• CBC with differential and liver function tests every 2 weeks for the first 2 months and every 2 to 3 months thereafter
Cyclosporine
Mechanism of action
• Forms a complex with cyclophilin, which inhibits calcineurin – an intracellular enzyme – which in turn reduces the activity of NFAT-1 (transcribes various cytokines, such as IL-2)
• ↓IL-2 production leads to decreased numbers of CD4 and CD8 cells.
Important pharmacology points
• Cyclosporine should ideally be gradually tapered while an alternative therapy is instituted to prevent flaring
• Maximum dermatologic dose = 5 mg/kg daily and can be used continuously for up to 1 year according to the FDA (2 years for worldwide consensus data)
■ Cyclosporine lipid nanoparticles formulation maximum dermatolyte dose = 4 mg/kg
• For obese patients, ideal body weight should be used to calculate starting dose
Indications
• Off-label uses include atopic dermatitis, chronic idiopathic urticaria, pyoderma gangrenosum, lichen planus, bullous dermatoses, autoimmune connective tissue diseases, neutrophilic dermatoses, and pityriasis rubra pilaris among others
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree