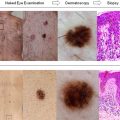
Lack of information and misinformation abounds regarding the potential risks of malignancy, management approaches, benefits of surgical intervention, follow-up strategies, and overall prognosis for individuals with congenital melanocytic nevi (CMN). This review is intended to provide answers to questions that frequently arise shortly after the birth of individuals with CMN, especially of larger types.
- •
Congenital melanocytic nevi (CMN) are classified by their projected adult diameter. They display a spectrum of clinical presentations, including nodular variants, multiple medium CMN and nevus spilus-like CMN.
- •
Leptomeningeal melanocytosis affects 5–15% of patients with CMN >20 cm. Of these, two thirds develop mild to severe neurological symptoms within five years.
- •
The lifetime risk of melanoma (cutaneous and extracutaneous) in patients with CMN >20 cm is probably less than 5%. Melanomas developing within CMN >40 cm usually arise deep to the dermo-epidermal junction and tend to develop in childhood.
- •
Staged excision after previous tissue expansion is a reasonable therapeutic option for large CMN of the trunk, scalp, and forehead.
- •
CMN support groups have been founded in several countries, and the parents of a newborn child with a CMN should be encouraged to join these groups.
Introduction
This article is intended to provide a practice-oriented update on the management of congenital melanocytic nevi (CMN). It is clear that the complexity of the management issues increases with larger CMN, and it is for this reason that this article focuses on these larger CMN. However, many of the management principles outlined in this article may also be relevant for smaller CMN. The scaffold for the article is based on 5 key questions, which are raised in almost every case. The answer to these questions permits the clinician to create a structured management approach for any newborn child with a CMN.
Question 1: what type of CMN is present and what is its morphology?
The diagnosis of a CMN is usually straightforward. The assessment of the nevus should take into account both its primary and secondary features. The primary features include the location and largest diameter of the nevus. In addition, the presence and number of accompanying smaller (satellite) nevi should be recorded. Traditionally, CMN larger than 20 cm projected adult size (PAS) are designated as large CMN (LCMN) (all metrical data in this article refer to the largest diameter the CMN is expected to attain in adulthood). From birth to adulthood, the area of the head enlarges by a factor of 2.8, trunk and arms by a factor of 8, and legs by a factor of 12. Consequently, a CMN in a newborn can be expected to reach 20 cm PAS if its largest diameter is around 6 cm on the legs, 7 cm on the trunk and arms, or 12 cm on the face and scalp. The secondary features include the morphologic spectrum of CMN, which can be quite diverse, and comprise variations in color, hairiness, surface topography (ie, rugosity, including mammillated and verrucous changes), and nodularity (ie, palpable dermal or subcutaneous nodules). It is becoming increasingly apparent that these secondary features affect management and thus should be recorded and also included in epidemiologic studies so as to determine any association with complications such as malignancy. In fact, a revised CMN classification system is currently under development, which takes both primary and secondary features into account and classifies nevi on the basis of size and morphology.
On rare occasions, newborns may exhibit unusual CMN variants. The following CMN features may raise difficulties for the clinician:
- 1.
CMN with extensive dermal and/or subcutaneous nodules: Characteristic examples with this morphology are depicted in Fig. 1 . They mostly affect CMN larger than 40 cm PAS and are often situated on the back, buttocks, genitalia, or, more rarely, the head/face. Histologically, these tumors consist of monomorphous, often epithelioid nevomelanocytes of variable density, which, at least in older lesions, are embedded in a myxoid stroma. The cells may show variable degrees of peripheral nerve sheath differentiation, neurotization, and mesenchymal or lipomatous changes. Larger lesions are sometimes confused for plexiform neurofibroma. A characteristic variant in the genital area has been described as bulky naevocytoma of the perineum. Although most of the dermal or subcutaneous nodules in CMN are present at birth and remain relatively stable, others may develop during infancy or early childhood and enlarge rapidly or even ulcerate. It is quite probable that the former (stable) and the latter (proliferative) nodules represent a congenital and a tardive variant of the same histogenetic process. In enlarging nodules, excision is often necessary to exclude the possible diagnosis of melanoma. The histologic spectrum of proliferative nodules in CMN ranges from benign to atypical variants, the latter exhibiting a certain degree of atypicality of the dermal melanocytes, a blurred demarcation of the underlying nevus, and infrequent mitoses. In cases of doubt, molecular differentiation from melanoma may be supported by comparative genomic hybridization from paraffin-embedded tissue.
Fig. 1
CMN with extensive dermal and subcutaneous nodules: ( A ) face, ( B ) upper back, and ( C ) genitogluteal area.
- 2.
Multiple medium CMN ( Fig. 2 ): This variant usually looks much less outstanding than a giant CMN but often carries with it a worse prognosis. These patients manifest only a few (between 3 and 10) medium-sized CMN on the skin (1.5–20 cm PAS). However, multiple foci of abnormal/excessive deposits of melanocytes are often present on the leptomeninges of these patients. These central nervous system (CNS) melanocytic deposits can lead to symptomatic neurologic involvement. The presence of CMN in the skin and CNS is known as neurocutaneous melanocytosis (NCM). The literature review by Kadonaga and Frieden reports that approximately two-thirds of patients with NCM had a giant CMN on the trunk with multiple satellites, and one-third had the variant manifesting multiple medium CMN.
Fig. 2
Multiple medium CMN.
- 3.
Nevus spilus–like variants: Rarely, large or giant CMN may show features reminiscent of speckled lentiginous nevi (SLN, also known as nevus spilus) ( Fig. 3 ). Some investigators regard nevus spilus–like variants of CMN as true (large) SLN. However, even if congenital, typical SLN represents a characteristic entity that can in most cases be easily separated from conventional CMN. SLN are defined by the presence of grouped macular or papular melanocytic nevi on a light brown (café au lait macule like) background. The morphologic spectrum of SLN does not include hypertrichosis. In larger SLN, the background café au lait–like lesion sometimes exhibits a sharp midline demarcation and a “checkerboard pattern” distribution. Twin spotting has been discussed as a reason for hybrid forms of large SLN and conventional CMN with hypertrichotic plaques. As yet, there are no indications that nevus spilus–like variants of CMN differ from other forms of CMN in terms of risk of malignancy ; death from melanoma has been reported. To the authors’ knowledge, no cases of leptomeningeal melanocytosis have been observed in patients with nevus spilus–like variants of CMN.
Fig. 3
Nevus spilus–like CMN.
- 4.
Congenital blue nevi: Congenital dermal melanocytoses, including blue nevi of the epithelioid and spindle cell type, nevus of Ota, and Mongolian spot, represent a distinct group of melanocytic neoplasms ( Fig. 4 ). In almost all instances, a clear-cut distinction from a conventional CMN is possible. On the level of pathogenesis, blue nevi (as well as uveal melanoma) frequently exhibit GNAQ mutations, in contrast to NRAS mutations, which is the prevailing finding in CMN.
Fig. 4
Large congenital blue nevus of the spindle cell type. The patient underwent operation at age 40 years to alleviate symptoms arising from leptomeningeal involvement. However, he succumbed to CNS melanoma shortly thereafter.
- 5.
Regressive CMN variants is a preliminary term to describe CMN with spontaneous regression. The regression is more often partial than complete. Observations of regression may be made in CMN of all types and sizes, and complete regression is most often seen in scalp nevi. Regression may present as gradual fading of the nevus color or with a halo phenomenon ( Fig. 5 A). In some instances, the reaction involves inflammation and fibrosis. Subtle regressive changes may be indistinguishable from the frequent finding of lightening of the nevus color that commonly occurs during childhood and adolescence (see Fig. 5 B, C). CMN in fair-skinned children, especially with the Celtic complexion (red hair, freckles), almost always lighten (Krengel, unpublished results, 2012).
Fig. 5
Regressive CMN. ( A ) Spontaneous partial regression of a CMN, similar to the halo phenomenon occurring in acquired melanocytic nevi. ( B , C ) Significant spontaneous lightening of a CMN in the course of 4 years.
Question 1: what type of CMN is present and what is its morphology?
The diagnosis of a CMN is usually straightforward. The assessment of the nevus should take into account both its primary and secondary features. The primary features include the location and largest diameter of the nevus. In addition, the presence and number of accompanying smaller (satellite) nevi should be recorded. Traditionally, CMN larger than 20 cm projected adult size (PAS) are designated as large CMN (LCMN) (all metrical data in this article refer to the largest diameter the CMN is expected to attain in adulthood). From birth to adulthood, the area of the head enlarges by a factor of 2.8, trunk and arms by a factor of 8, and legs by a factor of 12. Consequently, a CMN in a newborn can be expected to reach 20 cm PAS if its largest diameter is around 6 cm on the legs, 7 cm on the trunk and arms, or 12 cm on the face and scalp. The secondary features include the morphologic spectrum of CMN, which can be quite diverse, and comprise variations in color, hairiness, surface topography (ie, rugosity, including mammillated and verrucous changes), and nodularity (ie, palpable dermal or subcutaneous nodules). It is becoming increasingly apparent that these secondary features affect management and thus should be recorded and also included in epidemiologic studies so as to determine any association with complications such as malignancy. In fact, a revised CMN classification system is currently under development, which takes both primary and secondary features into account and classifies nevi on the basis of size and morphology.
On rare occasions, newborns may exhibit unusual CMN variants. The following CMN features may raise difficulties for the clinician:
- 1.
CMN with extensive dermal and/or subcutaneous nodules: Characteristic examples with this morphology are depicted in Fig. 1 . They mostly affect CMN larger than 40 cm PAS and are often situated on the back, buttocks, genitalia, or, more rarely, the head/face. Histologically, these tumors consist of monomorphous, often epithelioid nevomelanocytes of variable density, which, at least in older lesions, are embedded in a myxoid stroma. The cells may show variable degrees of peripheral nerve sheath differentiation, neurotization, and mesenchymal or lipomatous changes. Larger lesions are sometimes confused for plexiform neurofibroma. A characteristic variant in the genital area has been described as bulky naevocytoma of the perineum. Although most of the dermal or subcutaneous nodules in CMN are present at birth and remain relatively stable, others may develop during infancy or early childhood and enlarge rapidly or even ulcerate. It is quite probable that the former (stable) and the latter (proliferative) nodules represent a congenital and a tardive variant of the same histogenetic process. In enlarging nodules, excision is often necessary to exclude the possible diagnosis of melanoma. The histologic spectrum of proliferative nodules in CMN ranges from benign to atypical variants, the latter exhibiting a certain degree of atypicality of the dermal melanocytes, a blurred demarcation of the underlying nevus, and infrequent mitoses. In cases of doubt, molecular differentiation from melanoma may be supported by comparative genomic hybridization from paraffin-embedded tissue.
Fig. 1
CMN with extensive dermal and subcutaneous nodules: ( A ) face, ( B ) upper back, and ( C ) genitogluteal area.
- 2.
Multiple medium CMN ( Fig. 2 ): This variant usually looks much less outstanding than a giant CMN but often carries with it a worse prognosis. These patients manifest only a few (between 3 and 10) medium-sized CMN on the skin (1.5–20 cm PAS). However, multiple foci of abnormal/excessive deposits of melanocytes are often present on the leptomeninges of these patients. These central nervous system (CNS) melanocytic deposits can lead to symptomatic neurologic involvement. The presence of CMN in the skin and CNS is known as neurocutaneous melanocytosis (NCM). The literature review by Kadonaga and Frieden reports that approximately two-thirds of patients with NCM had a giant CMN on the trunk with multiple satellites, and one-third had the variant manifesting multiple medium CMN.
Fig. 2
Multiple medium CMN.
- 3.
Nevus spilus–like variants: Rarely, large or giant CMN may show features reminiscent of speckled lentiginous nevi (SLN, also known as nevus spilus) ( Fig. 3 ). Some investigators regard nevus spilus–like variants of CMN as true (large) SLN. However, even if congenital, typical SLN represents a characteristic entity that can in most cases be easily separated from conventional CMN. SLN are defined by the presence of grouped macular or papular melanocytic nevi on a light brown (café au lait macule like) background. The morphologic spectrum of SLN does not include hypertrichosis. In larger SLN, the background café au lait–like lesion sometimes exhibits a sharp midline demarcation and a “checkerboard pattern” distribution. Twin spotting has been discussed as a reason for hybrid forms of large SLN and conventional CMN with hypertrichotic plaques. As yet, there are no indications that nevus spilus–like variants of CMN differ from other forms of CMN in terms of risk of malignancy ; death from melanoma has been reported. To the authors’ knowledge, no cases of leptomeningeal melanocytosis have been observed in patients with nevus spilus–like variants of CMN.
Fig. 3
Nevus spilus–like CMN.
- 4.
Congenital blue nevi: Congenital dermal melanocytoses, including blue nevi of the epithelioid and spindle cell type, nevus of Ota, and Mongolian spot, represent a distinct group of melanocytic neoplasms ( Fig. 4 ). In almost all instances, a clear-cut distinction from a conventional CMN is possible. On the level of pathogenesis, blue nevi (as well as uveal melanoma) frequently exhibit GNAQ mutations, in contrast to NRAS mutations, which is the prevailing finding in CMN.
Fig. 4
Large congenital blue nevus of the spindle cell type. The patient underwent operation at age 40 years to alleviate symptoms arising from leptomeningeal involvement. However, he succumbed to CNS melanoma shortly thereafter.
- 5.
Regressive CMN variants is a preliminary term to describe CMN with spontaneous regression. The regression is more often partial than complete. Observations of regression may be made in CMN of all types and sizes, and complete regression is most often seen in scalp nevi. Regression may present as gradual fading of the nevus color or with a halo phenomenon ( Fig. 5 A). In some instances, the reaction involves inflammation and fibrosis. Subtle regressive changes may be indistinguishable from the frequent finding of lightening of the nevus color that commonly occurs during childhood and adolescence (see Fig. 5 B, C). CMN in fair-skinned children, especially with the Celtic complexion (red hair, freckles), almost always lighten (Krengel, unpublished results, 2012).
Fig. 5
Regressive CMN. ( A ) Spontaneous partial regression of a CMN, similar to the halo phenomenon occurring in acquired melanocytic nevi. ( B , C ) Significant spontaneous lightening of a CMN in the course of 4 years.
Question 2: what is the risk of CNS involvement?
The hallmark of NCM is the presence of an abnormal deposition of nevomelanocytes along the leptomeninges. This is almost always associated with large (or multiple smaller) CMN on the skin. By logistic regression analysis, the number of satellite nevi has been determined as the predominant significant risk factor for NCM. The statistical risk of a given newborn with a CMN larger than 20 cm PAS was 5 times higher for children with more than 20 satellites than that in children with less than 20 satellites (12.5% vs 2.5%). The presence of melanin deposits in the pia mater of the brain or spine is demonstrable by magnetic resonance imaging (MRI) in approximately 5% to 15% of children with CMN larger than 20 cm PAS ([26/379], [49/1008], [18/120] ). The presence of melanocytes in the CNS may not result in any neurologic squeal; however, it is not uncommon to develop neurologic symptoms ranging from mild to severe. The numerical ratio between asymptomatic and symptomatic NCM is in the order of 1/3 (asymptomatic) to 2/3 (symptomatic NCM). In one study, the rate of progressing from asymptomatic NCM to symptomatic NCM was 7% during a mean follow-up of 5 years. The reported ratios between asymptomatic and symptomatic NCMs are influenced by the clinical threshold criteria used to delineate symptomatic disease and the meticulousness of the neuropsychological examinations.
Symptoms of NCM are usually induced by CNS melanocyte hyperplasia or due to the development of melanoma within the CNS. They include seizures as well as symptoms arising from raised intracranial pressure. Symptoms manifested themselves in 72% within the first 2 years of life (including mild developmental delay) and in 89% within 5 years of life. The long-term prognosis of NCM remains to be determined. Although many symptoms can currently be pharmacologically or surgically controlled, death resulting from progressive neurologic complications or due to CNS melanoma is quite common. Kinsler and colleagues reported that 39% of MRI-positive patients required neurosurgical interventions in the course of their disease and that approximately 8% of symptomatic patients with NCM died of neurologic complications during a mean follow-up of 8.35 years. In another study by Bett, 20.7% of symptomatic patients with NCM died of neurologic complications and 13.8% died of CNS melanoma during a mean follow-up of 5.6 years.
An MRI scan should be obtained for any patient manifesting concerning neurologic symptoms. In addition, an MRI should be considered in asymptomatic patients with CMN larger than 40 cm PAS, especially if they also have more than 20 satellites and in patients with multiple medium CMN or in patients with multiple satellites only. Because myelinization of the brain may obscure the ability to detect subtle NCM deposits, it may be ideal to obtain screening MRI scans between the fourth and eighth month of life. With that said, in most cases, MRI scans appear to be equally sensitive at a later age. MRI scan for NCM-positive patients is valuable in that it can help guide management, including deciding on the feasibility of neurosurgical interventions aimed at alleviating symptoms. In asymptomatic patients at increased risk for NCM, a negative MRI result can help reassure both parents and physicians. On the other hand, if the screening MRI scan in an asymptomatic patient proves positive, it may result in heightened anxiety and may appropriately delay elective excision of cutaneous CMN until after it is confirmed that the NCM is stable and nonprogressive.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree