Fig. 70.1
Blunt amputation after a patient attempted to jump onto a train and was dragged under the wheels. This illustrates why blunt amputations are generally not candidates for replantation
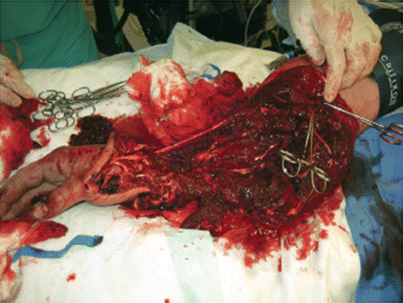
Fig. 70.2
Propellor injury after a patient fell out of a boat
The difficulty with this decision has led to a great deal of research in an attempt to come up with objective scoring systems that will predict those patients who will be best served with early amputation. The good news is that there is no shortage of scoring systems from which to choose: the Mangled Extremity Severity Score, Hanover Fracture Scale, Predictive Salvage Index, Limb Salvage Index, and others. The bad news is that none of them are great in terms of answering your question as they tend to lack sensitivity in addressing which limbs should undergo early amputation. While I think that these scoring systems have limited utility when their quantitative scores are used, they are of some use if you look at them qualitatively as they will tell you the variables that need to be factored into your decision.
Let us start with the easy part. This has nothing to do with the crushed extremity but with the rest of the patient instead. I am referring to the patient’s overall trauma burden. How sick are they? With how many organ systems and body cavities are you contending? Are they in shock and not resuscitating well? It can be unequivocally stated that for the patient in extremis, an expeditious guillotine amputation (i.e., rapidly getting the extremity off, ligating the major vasculature involved, and doing nothing else without any attempt at closure) is the procedure of choice at the first operation. For the patient who is hemodynamically stable but has suffered a severe set of injuries, one should keep in mind the tremendous amount of physiologic reserve it takes to try to heal a mangled extremity. Large amounts of the traumatized muscle and bone exert profound immunomodulatory effects and can also serve as a septic source. Given that, one can see that in the massively injured patient, it is wise to strongly consider early amputation (remember the old principles of priority in trauma training: life, the limb, function, cosmesis). I have had conversations with older, more experienced colleagues who have suggested that an Injury Severity Score greater than 25 is a contraindication to heroic limb salvage. While I think that this number may be a little on the low side, it gives you a frame of reference for the debate. Further, as a patient gets older (and consequently has less in the way of metabolic reserves), one’s threshold for amputation due to overall injury burden should lower.
In considering factors associated with the extremity itself, it is interesting to note that a plurality of trauma orthopods interviewed on decision-making factors for amputation in this scenario felt that the single biggest predictor of the need for early amputation was the loss of neurologic integrity and/or plantar sensation. Clearly, undergoing limb salvage in the hope of ultimately leaving a patient with an insensate, paralyzed extremity is a questionable course to undertake. One should resist the temptation to be dogmatic about this as an absolute indication, however, because stretch injury, compression, and ischemia can all result in transient nerve deficits. In fact, half of patients who present with an insensate foot and undergo limb salvage will regain sensation at 2 years. If the deficit is felt to be permanent, however (i.e., if the posterior tibial nerve is visualized as transected from a crushing injury), it is defensible to say that amputation should be undertaken early rather than late.
Vascular injuries and ischemia time clearly play a large role in limb viability. After 6 h of warm ischemia time, limb salvage is anecdotal. Of note, one rarely commented upon factor is the importance of the length of an interposition graft in a vascular repair. Long grafts can give rise to the phenomenon of “interstate syndrome.” When these high-speed, uninterrupted highway systems were built in the United States, they bypassed many small towns which had previously survived on the business brought to them by visitors passing through on the smaller roads. Just as many of these small towns withered away after potential visitors elected to use the interstates without ever exiting, so to do smaller muscle units wither away as the vascular graft bypasses them without giving off perforators.
The muscular component of the injury is best evaluated by the four “Cs:” color, contracture, consistency, and circulation. As a practical matter, the single best predictor of muscle viability is bleeding during debridement. After removing that muscle which is clearly nonviable, the surgeon needs to ask if the crush injury has left the patient with so little viable muscle that the resulting limb is worth continuing to attempt to salvage. If the answer is in the affirmative, it must be asked anew each time the patient goes back to the operating room for further debridement. These issues are not limited to the extremity, as the earlier scenario involving the city worker at the beginning of this section will attest. This man received a massive blow to the right flank, transecting his liver and macerating part of his lower right abdominal wall. His liver was packed, and he subsequently did well. Over a matter of days, however, he required more and more debridement of the nonviable muscle wall until only the peritoneum remained. He then developed two enterocutaneous fistulae (Fig. 70.3). He underwent a simultaneous fistula resection and abdominal wall reconstruction several months after his initial injury.
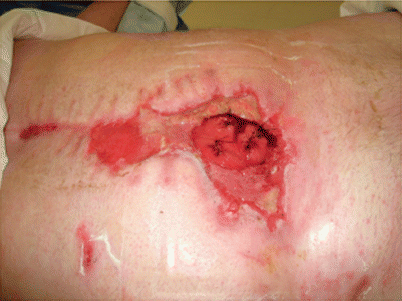

Fig. 70.3
Abdominal wall necrosis with subsequent fistulization after a blow to the right flank by a crossbar on a city garbage truck whose hydraulics broke
Surprisingly, the bony component of the injury is less important than the amount of soft tissue coverage that remains. Simply put, there is no substitute for healthy coverage. This will affect union rates, wound healing, and all of their downstream sequelae.
Finally, we have come to realize that there are social components that go into the likelihood of a successful outcome. Patients with a low frustration threshold, low levels of motivation, and poor social support tend to not do very well with the protracted course entailed with heroic salvage efforts. Before committing the patient to this undertaking, it is wise to have a very frank discussion with them and their family on what can reasonably be expected. They should be made aware of the sobering statistic that at 2- and 7-year follow-up, functional outcomes are equivalent between amputation and reconstruction, and they are not good. By doing so, the surgeon protects the patient from unrealistic expectations and consequently himself or herself.
70.1.2 Compartment Syndrome
It has been said that paranoia is a healthy attitude in a trauma surgeon, and this is especially true in the diagnosis and management of compartment syndromes. They frequently present in a subtle fashion and if missed will render the good technical results of an operation moot, inflict serious morbidity on the patient, and potentially serve as a source of litigation. Given the ease with which they can be overlooked, and the serious consequences when that happens, they are accurately viewed as a bear trap by practicing trauma surgeons.
The first step in managing a possible compartment syndrome is to be aware of the clinical scenarios in which they frequently occur. Two broad categories of insult lead to the increase in intra-compartmental pressures which characterize the condition. The first is relatively straightforward and generally occurs after direct trauma to the injured portion of the extremity. Here, the lacerated muscle and fractured bone bleed into the unyielding fascial compartment and result in the characteristic increased pressure. The crushed and contused muscle will accumulate some degree of hematoma, further exacerbating the condition. The second etiology for compartment syndrome and the most common one seen in my practice is after a limb has undergone a period of ischemia typically after a vascular injury. In this setting, the anaerobic metabolites that build up downstream from a site of vascular occlusion accumulate in proportion to the length of time for which a limb has been ischemic. When the limb is reperfused, these toxic by-products of anaerobic metabolism wash out and cause significant capillary leak and cell swelling. In this setting, the syndrome occurs in the compartments downstream from the site of injury. The occlusion can also arise from extrinsic causes, such as when a cast is applied too tightly. Finally, the most insidious presentation of compartment syndrome can occur with the anasarca seen during and after a massive resuscitation. Here, the muscle compartments swell from an accumulation of third-spaced fluid, frequently aggravated by systemic hypotension and hypoperfusion. A limb which has been ischemic for 2 h is at high risk for compartment syndrome, while 4 h of ischemia time will certainly develop a compartment syndrome after reperfusion. By 6 h of ischemia time, not only is a compartment syndrome a certainty, but the neuromuscular deficits are frequently not reversible even after fasciotomy and limb salvage is anecdotal. It is easy to see how a crush injury can put a patient at risk for any of these causes.
Classically, surgical trainees are taught to watch for the five “Ps” of compartment syndrome: pain, pallor, paralysis, paresthesias, and pulselessness (occasionally, a sixth is included with poikilothermia). A point that needs to be reinforced is that all are not required for the diagnosis. The earliest sign to be seen is that of pain, particularly if it is out of proportion to what would normally be clinically expected. It is especially pronounced if the muscles within the compartment in question are squeezed or stretched. All of the remaining signs indicate that the condition is in a more advanced stage. Finally, it should be noted that a limb will be in the late stages of compartment syndrome before it loses a pulse. The occlusion of capillary perfusion and venous outflow occurs at much lower pressure levels, and the presence of a pulse in the extremity should not put the surgeon’s mind at ease.
Alright, so you have got a patient who has an injury pattern that has you worried and a set of complaints and findings that have got warning bells going off in your head. What do you do now? How do you proceed? Traditional surgical teaching states that compartment pressures should be measured in some way. Some will say that this can be done easily by hooking an IV needle up to a transducer, making sure that it is zeroed, sticking the needle directly into the compartment, and reading the resulting number. This technique is mentioned only to be condemned, however, as the muscle can “cork” the end of the needle and artificially elevate the pressure reading. Another option is to use commercially available specialized needles which have their pressure aperture slightly offset in such a manner that they avoid the problem of muscle plugging. This strategy yields a precise pressure value, albeit at the expense of extra, specialized equipment. If a quantitative strategy is undertaken, an absolute intra-compartmental pressure of 30 mmHg is diagnostic of a compartment syndrome. Additionally, a pressure value which is within 30 mmHg of the patient’s diastolic blood pressure should also be considered to represent a compartment syndrome.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree