Congenital melanocytic nevi are benign neoplasms, which present early in life at various anatomic locations with a variety of clinical presentations. When evaluating large or giant nevi, it’s important to use a multidisciplinary team and rule out comorbid conditions, such as neurocutaneous melanosis. Surgical interventions include direct excision, serial excision, and tissue expansion. For more complex lesions, tissue expansion is a versatile and effective option for many areas of the body. Most large or giant lesions require serial excision and long-term follow-up.
Key points
- •
Congenital melanocytic nevi are classified primarily by size and number of satellite lesions, which divides them into small, medium, large, or giant nevi.
- •
The goals of treatment of congenital nevi, especially in the pediatric population, are to minimize the risk of malignant transformation, preserve function, and to optimize aesthetic outcomes.
- •
Minimally invasive treatments, such as dermabrasion, chemical peels, and laser ablation will not decrease the risk of malignant transformation after treatment.
- •
Optimal surgical treatment varies greatly with anatomic location, and therefore, the treatment algorithm should account for the size and anatomic location of the lesion.
Introduction
Congenital melanocytic nevi (CMN) are classically defined as benign neoplasms and more specifically, clusters of nevus cells present at birth, or within the first year of life. , These lesions range significantly in location, size, associated systemic disease, and psychological burden. Throughout the past decades, there has been immense progress in the surgical treatment of these lesions, especially following the advent of tissue expansion and the use of local tissue rearrangement. The fundamental concepts of plastic and reconstructive surgery stand true when preparing for surgical intervention. The goal of this article is to provide a comprehensive understanding of the pathogenesis, presentation, and various surgical options for the management of CMN, with a focus on large and giant variants.
Epidemiology
Although most cases of congenital nevi are sporadic, there are occasionally familial associations. Approximately 1% of the general population has small CMN lesions. Large CMN, which are greater than 10 cm in diameter, occur in approximately 1 in 20,000 live births. Giant CMN, or greater than 40 cm in diameter, are even less common and are found in 1 in 500,000 live births. ,
Embryology, Histology, and Genetics
Embryologically, CMN develop during 5 to 24 weeks of gestation. The melanocytes begin in the neural crest as melanoblasts and migrate to various sites of the body such as the skin, mucous membranes, pia and arachnoid mater. Once at their final location, melanoblasts then differentiate to become dendritic melanocytes. When these cells fail to grow, migrate, and develop normally, a congenital nevus results. Nevus cells can be separately classified from developmentally normal melanocytes because of a lack of dendrites and an overall rounder shape. , Both large and small nevi most commonly consist of nests of nevomelanocytes seen at the epidermal–dermal junction or within the epidermis; however, nests can also be seen in the reticular dermis, which is more commonly associated with congenital nevi rather than acquired nevi. Additionally, CMN cells can be seen spanning collagen bundles of reticular dermis and they have the ability to infiltrate various glands, hair follicles, vessels, and nerves. In general, large CMNs have variable macroscopic morphology and are more complex histologically than the small CMNs.
Genetically, previous literature has widely reported the presence of N-Ras proto-oncogene (NRAS) and B-Raf proto-oncogene (BRAF) gene mutations in patients with congenital nevi. Furthermore, CMNs have been shown to evolve from somatic mutations in the NRAS or BRAF genes. Specifically in large and giant nevi, NRAS mutations were demonstrated in 94.7% of cases. Given the known association with these genes, recent investigation has attempted to target NRAS for melanoma gene therapy in patients diagnosed with neurocutaneous melanosis.
Clinical characteristics
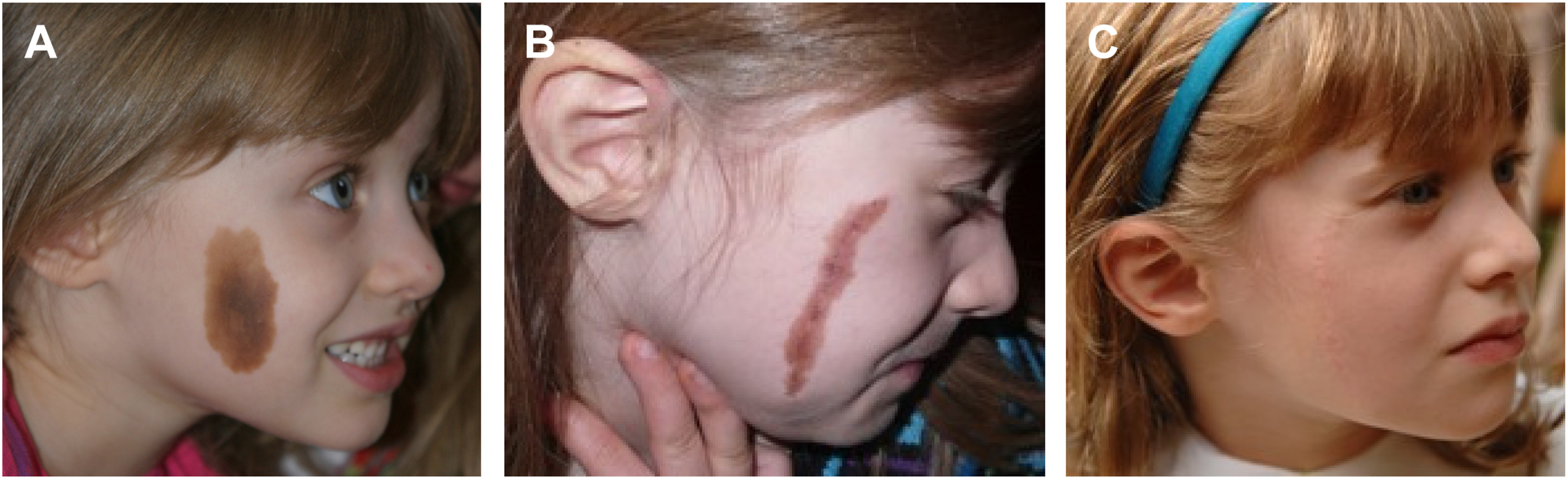
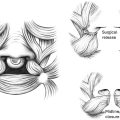
The clinical presentation of CMN can vary significantly not only in size and location but also color ( Fig. 1 ). The lesions typically appear light to dark brown in color and even have a bluish hue in darker skinned individuals. Some lesions change color over time and can appear lighter in color at birth and tend to darken over the first few months of life. These lesions are typically round or oval with well-defined borders. The classification of nevi has varied throughout the literature, but most recently Krengel and colleagues considers size, location, number of satellite nevi, and additional morphologic characteristics ( Fig. 2 ). , Using this classification for adults, small nevi are defined as less than 1.5 cm in diameter, medium nevi between 1.5 to 20 cm, and large nevi measure from 20 to 40 cm. Giant CMN are defined as either greater than 40 cm in diameter in adulthood or greater than 2% of the total body surface area in children , , , , . Where small and medium nevi have clear, demarcated borders and appear more homogenous, giant CMN have more irregular borders, nodular surfaces, and asymmetry in contrast.
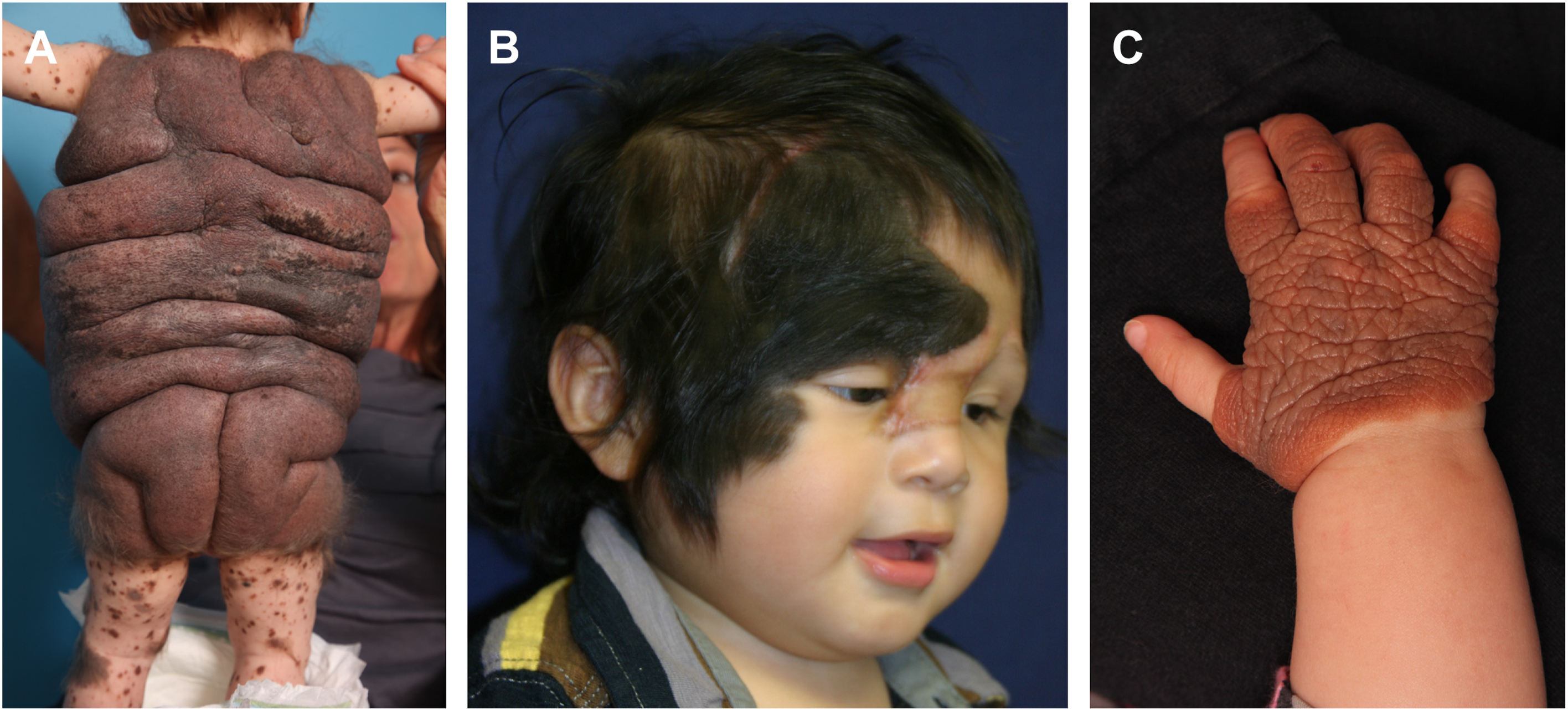

Regarding the anatomic location, the most common location is the trunk for large and giant nevi. This is followed by the extremities and then the head and neck region. It is common for the giant CMN to cross anatomical zones (ie, nevi may involve both the trunk and extremities), for which these patterns have been classically named. For example, the “bathing trunk”, “vest”, and “glove-stocking” distributions are common. Approximately 80% of patients with giant CMN will have associated satellite lesions. ,
Workup
When evaluating a patient with a CMN, there are multiple considerations that will determine the diagnostic workup and final treatment plan for each patient. For those with small or medium sized nevi, it is reasonable to assess for comorbidities and determine benefits of surgical excision versus conservative management, especially in the pediatric populations. Circumstances that would warrant surgical excision of small or medium nevi, include concern for malignancy, peer bullying or patient’s desires for excision (if of appropriate age for assent). When evaluating large or giant nevi, it is important to employ a multidisciplinary approach, which may include a dermatologist, plastic surgeon, radiologist, or neurologist when indicated. When evaluating a large or giant CMN, an MRI should be strongly considered to rule out neurocutaneous melanosis, which will be discussed below. Long-term follow- up is important for monitoring the signs of malignant transformation.
Features that should prompt biopsy and possibly early excision are known dysplasia, concern for melanoma, or concerning clinical features, such as ulceration, varied pigmentation within a singular lesion, evolution of shape or size over time, and abnormal nodularity on the face. While biopsy should be considered in the face of rapidly enlarging or changing nodules, proliferative nodules are commonly seen amidst large and giant nevi.
Malignant Transformation
The discussion of malignant transformation to melanoma or sarcoma has been controversial when determining whether to surgically intervene with CMN lesions. Less than 0.5% of melanomas arise in children but 33% of those that do, are thought to arise from CMN. The presence of melanoma is always reported as arising from the main lesion and rarely from the satellite lesions. Malignancy in CMN patients is most often seen in the trunk or head and neck regions. The risk of malignant transformation of small nevi is variable and ranges from 0% to 5% and increases to 4% to 6% for large nevi. The largest risk, however, is in patients with comorbid neurocutaneous melanosis (see below). Large melanomas occur deep in the dermal-epidermal junction or else are in an extra-cutaneous location (ie, CNS retroperitoneum) and will typically occur in the first decade of life. Clinical presentation of malignant transformation may not differ to an extreme from the original lesion, but it is important to recognize any significant changes over time, including change in pigmentation, change in the rate of growth, change in shape, change in surface texture, or nodularity, or change in subjective findings (ie, pain, pruritus, or bleeding). No studies have definitively shown that the excision of large CMN reduces the rate of conversion; however, this is difficult to prove because the excised lesion may have transformed later. For patients who do not undergo complete excision of these lesions, long-term follow-up is critical.
In the early 2000s there were numerous articles investigating the incidence of melanoma in patients with large congenital nevi. A systematic review of 14 studies with a total of 6571 patients reported that 0.7% of the patients developed melanoma. This translated into a 465-fold increased relative risk for patients with large congenital nevi during childhood and adolescence (median age of 7 years) developing these malignancies. In this study, 67% of the melanoma lesions arose from the primary CMN. A second systematic review (8 studies with 432 patients), which investigated malignant transformation in large congenital nevi, reported a rate of 2.8% of patients ultimately developing melanomas. Eighty-three percent of these malignancies originated in the primary nevus. The authors used a standardized morbidity ratio to conclude that the incidence of melanoma in these patients was “2599 times higher than would be expected in 120 individuals in the general population”. Two additional studies in the same period reported retrospective and prospective cohort outcomes with rates of malignant transformation at 2.9% (20) and 2.3% respectively , with a morbidity ratio of 324, , . The majority of melanomas occurred in the primary nevus and the risk of malignancy was greater with larger numbers of satellite lesions. Although there were varying outcomes amongst the articles, they highlight the possibility of melanoma development and emphasize the importance of patient counseling when deciding whether or not to prophylactically excise a congenital nevus.
In recent years, the literature echoed these findings of risk of melanoma development. One single center retrospective review reported the rate of malignant transformation of giant congenital nevi (including melanoma, rhabdomyosarcomas, and peripheral nerve malignancies) occurring in 4.6% of the patients over a 20 year span. Again, median age was in childhood at 3.17 year old and the majority of the lesions arose from the primary CMN. In a larger meta-analysis, the rate of malignancies was found to be fewer at 1.15% amongst 7915 patients with CMN. Additionally, the risk ratio in large or giant nevi was 21.9 compared with small and medium nevi, and the age of diagnosis was 9.5 years. For those patients diagnosed with melanoma, the mortality rate was found to be a startling 33%. There have been a handful of case reports also, identifying children with transformation of a congenital nevus to melanoma.
It can be expected that individual studies will result in varying rates of malignant transformation. However, throughout the years the literature continues to demonstrate an ever-present risk of children or adolescents developing a malignant lesion from a preexisting congenital nevus, even if the risk is small. This evidence accentuates the importance of close monitoring, long-term follow-up, and a high index of suspicion from providers for potential melanoma or rhabdomyosarcoma in patients with congenital nevi.
Neurocutaneous Melanosis
Neurocutaneous melanosis (NCM) is a condition where there is abnormal deposition of melanocytes into the leptomeninges (pia and arachnoid) and brain parenchyma. Approximately 15% of patients with large or giant CMN are found to have NCM. It is most common in the temporal lobe when it is isolated. The majority of these patients are asymptomatic; however, the greatest potential risk of NCM is malignant transformation, leading to a central nervous system malignancy. Additional morbidities that can occur include seizures, hydrocephalus, central nervous system (CNS) disturbances, and learning or developmental delays. As mentioned previously, the current recommendation for MRI screening is for patients with large nevi, especially in the posterior midline of the trunk or those with greater than 10 satellite nevi.
Management
The ultimate goals of treatment of congenital nevi, especially in the pediatric population, are to minimize the risk of malignant transformation, preserve function, and to optimize aesthetic outcomes. It is important to consider these factors when deciding whether to intervene while simultaneously weighing the morbidity of multiple procedures. Studies have demonstrated that those with untreated large and giant lesions suffered from bullying, higher rates of depression, and lower self-esteem. Additionally, patient-reported outcome-studies showed less psychological impact of the resulting scar following excision compared to the original lesion. A recent study in Spain also investigated the psychosocial impact of large congenital nevi. Based on patient reported outcomes, the authors concluded that although it took an average of 5.5 operations for complete excision, 70% of the patients and caregivers felt that the surgeries did not negatively impact their quality of life and that surgical intervention should begin as young as possible. They also noted that the younger the patient’s age at the time of the index surgery, the higher the surgeon’s satisfaction with the outcome.
The toll of psychological impact, however, needs to be balanced with the patient’s candidacy for operative intervention in addition to creating the foundation for a long-term surgeon–patient relationship.
Surgical Timing
Surgical timing is another topic of heavy discussion amongst surgeons who manage CMN. Some argue to wait until the child can personally advocate for excision while others strongly urge complete excision before school age. It is most common, however, to begin reconstruction early in life. When determining whether a patient is an appropriate surgical candidate, it is important to not only consider clinical indications and medical comorbidities but also social history and the presence of family support.
Minimally Invasive Techniques
Treatment of CMN includes surgical and nonsurgical interventions. Of the minimally invasive treatments, dermabrasion, chemical peels, and laser ablation are the most commonly discussed. These techniques are more often employed by dermatologists and less so by plastic surgeons. Although the physical appearance of the nevi may show a slight improvement, none of these methods will result in complete resolution of the lesion. Furthermore, there have even been in vitro studies demonstrating the worsening of malignant potential via laser therapy due to exposure to thermal energy. Another potential risk of these techniques is making sequential monitoring more difficult by altering the appearance of the native lesion. These methods will not decrease the risk of malignant transformation after treatment.
Excisional Techniques
Only direct excision has the potential to alter the rate of malignant transformation, although no study to date has demonstrated a lower risk of melanoma. Surgical excision is size-dependent and is the most common management of most nevi, especially when discussing large and giant CMN lesions. Amongst the surgical options, the most commonly described are single-stage excision, serial excision with or without tissue expansion, tissue expansion with local tissue rearrangement, or excision with skin grafting. Classically, if the lesion is believed to be able to be excised in 2 to 3 procedures, serial excision is recommended ( Fig. 3 ). At least 6 months between procedures is recommended.