Adverse reactions and unintended effects can occasionally occur with toxins for cosmetic use, even although they generally have an outstanding safety profile. As the use of fillers becomes increasingly more common, adverse events can be expected to increase as well. This article discusses complication avoidance, addressing appropriate training and proper injection techniques, along with patient selection and patient considerations. In addition to complications, avoidance or amelioration of common adverse events is discussed.
Key points
- •
Although counterfeit neurotoxin is available through Internet resources, it may not contain the amount of toxin stated on the label; risk of hurting patients makes the use of these products highly unethical.
- •
Adverse events with cosmetic use of neurotoxins and with fillers are commonly associated with intramuscular injections: local pain, infection, inflammation, tenderness, edema, erythema, bleeding, and ecchymosis.
Introduction
Toxins
Background
Forms of botulinum toxin type A (BoNTA) available for cosmetic use include onabotulinumtoxinA (Botox Cosmetic, Allergan, Irvine, CA), abobotulinumtoxinA (Dysport, Medicis Aesthetics, Scottsdale, AZ) and incobotulinumtoxinA (Xeomin, Merz Pharmaceuticals, Greensboro, NC). All of them are produced from fermentation of Clostridium botulinum type A (Hall strain) and may or may not contain accessory or hemagglutinin proteins. Other products marketed for medical (noncosmetic) purposes include onabotulinumtoxinA (Botox) and rimabotulinumtoxinB (Myobloc), which is produced by fermentation of the bacterium Clostridium botulinum type B (Bean strain). Units of potency are specific to each BoNTA product, and the doses or units of biological activity cannot be compared or converted from 1 product to another.
It is important to use only BoNTA products that are approved by the US Food and Drug Administration (FDA). Counterfeit neurotoxin is available through Internet resources and may not contain the amount of toxin stated on the label. Although these products can be bought at a reduced price, the risk of hurting patients makes the use of these products highly unethical.
Pharmacology
All BoNTA molecules consist of 1 heavy chain and 1 light chain polypeptide linked by a disulfide bond. They all share the same 3-step mechanism of action, which inhibits release of acetylcholine from peripheral cholinergic nerve endings at the neuromuscular junction. First, the heavy chain binds to specific surface receptors on nerve endings. The BoNTA molecule then undergoes internalization via receptor-mediated endocytosis and the disulfide bond is cleaved. The light chain undergoes translocation to the cytosol, where it cleaves polypeptides essential for docking, fusion, and release of acetylcholine vesicles through the cell membrane, resulting in chemical denervation of the muscle. Recovery of neurotransmission occurs as the neuromuscular junction recovers from SNAP25 cleavage and as new nerve endings are formed.
Postmarketing safety
Despite the potent systemic toxicity of BoNTA, the intramuscular injection of cosmetic BoNTA has an acceptable safety profile, with one of the largest known therapeutic indices for a prescription drug. It is one of the most studied pharmaceutical agents, with more than 3000 peer-reviewed published articles and more than 30 years of clinical experience. More than 10 million BoNTA treatments were performed worldwide last year. When used according to appropriate standards of care, it is the opinion of this author that BoNTA is one of the safest pharmaceutical agents available. Adverse events related to the cosmetic use of BoNTA reported to the FDA over a 13.5-year period included 36 serious and 995 nonserious adverse events. There were no reported deaths. The nonserious adverse events consisted mainly of lack of effect ( N = 623; 63%) and injection site reaction ( N = 190; 19%).
In contrast, there were 406 reported adverse events related to the use of BoNTA for therapeutic or medical uses, such as cervical dystonia and cerebral palsy. These adverse events included 217 serious adverse events, including 28 deaths caused by respiratory, myocardial infarction, cerebrovascular accident, pulmonary embolism, and pneumonia ; however, many of these patients already had underlying disease, which likely led to or precipitated these serious events. The serious adverse events associated with medical uses of BoNTA are often caused by the use of higher doses used to treat large muscles and diffusion or the spread of the toxin away from the injection site or the result of systemic toxicity. Among patients treated for spasticity with BoNTA, the mean dose of onabotulinumtoxinA was 245 units (standard deviation [SD] 118 units) and abobotulinumtoxinA was 939 units (SD 324 units). In contrast, it is uncommon to use more than 50 units cosmetically.
As a result of these reports, the FDA introduced several revisions to the prescribing information of BoNTA products (including aesthetic products), which includes a Boxed Warning highlighting the possibility of potentially life-threatening effects of distant spread of toxin after local injection, a Risk Evaluation and Mitigation Strategy, which includes a Medication Guide to help patients understand the risks and benefits of botulinum toxin products, and changes to the established drug names to reinforce individual potencies and prevent medication errors.
Indications
With respect to cosmetic use, BoNTA is indicated for the temporary improvement in the appearance of moderate to severe glabellar lines associated with corrugator or procerus muscle activity in adult patients aged 65 years or older ; however, off-label use is widespread and includes the treatment of forehead lines, crow’s feet, bunny lines, upper and lower perioral lip lines, gingival smile, marionette lines, dimpled chin, and vertical neck lines. Its effectiveness and safety in patients older than 65 years has also been shown.
Contraindications
The use of BoNTA is contraindicated in patients with an infection at the proposed site of injection and in individuals with known hypersensitivity to any botulinum toxin preparation or to any of the components in the formulation, including albumin and cow’s milk protein, because serious allergic reactions may occur. Although BoNTA is not recommended for use during pregnancy, there are reports of BoNTA administration during pregnancy for medical conditions without apparent complications. It is unknown whether BoNTA is excreted in breast milk.
Precautions
Because BoNTA diminishes neurotransmitter signaling at the neuromuscular junction, it should be used cautiously in patients with preexisting neuromuscular diseases, such as amyotrophic lateral sclerosis, myasthenia gravis, or Lambert-Eaton myasthenic syndrome. The use of BoNTA has unmasked subclinical Lambert-Eaton myasthenic syndrome. Similarly, patients may experience exaggerated effects in the presence of neuromuscular junction blockers, such as succinylcholine chloride, or other drugs that may interfere with neuromuscular transmission, including aminoglycosides, polymyxin, cyclosporin, chloroquine, quinidine, magnesium sulfate, and acetylcholinesterases. The use of anticholinergic drugs after BoNTA administration may potentiate their systemic anticholinergic effects.
Common adverse events
Common adverse events include local pain, infection, inflammation, tenderness, edema, erythema, bleeding, and ecchymosis, which are commonly associated with intramuscular injections. Other potential concerns include pain and infection. Bruising is of particular concern in the crow’s feet/lateral canthus/orbicularis oculi region. Ecchymosis occurs most frequently in this region, because of the delicate and highly vascular nature of this tissue. During clinical studies, bruising and pain were reported to occur in 6% to 25% of treated patients.
Reports of temporary hypoesthesia at the injection site may be related to local needle trauma and edema but does not seem to be caused by nerve injury. Uncommon psoriasiform eruptions and skin dryness and flakiness in the treated area may be caused by changes in skin secretions caused by BoNTA. A potentially serious adverse event after BoNTA injection is severe headaches. Although not recognized in my own experience, 1 report states that approximately 1% of treated patients developed headaches described as severe and debilitating. They spontaneously resolved after 2 to 4 weeks.
Unintended pharmacologic (toxic) effects
Although cosmetic BoNTA has an exceptional safety profile, misplaced injections may cause unwanted effects in nontarget areas. These effects may be the result of poor injection technique and failure to understand underlying facial anatomy. In addition to improper placement, unintended effects may also result from diffusion or spread of injected BoNTA into nearby functionally important muscle fibers. Injecting larger volumes of BoNTA results in greater diffusion, affecting a larger area. The spread of toxin can also be affected by muscle contraction in the injected area.
Eyelid ptosis is a common complication of BoNTA injections and is most likely to be associated with injections of the orbicularis oculii, corrugator supercilii, and procerus muscles. Eyelid ptosis may also result from BoNTA diffusion to the levator palpebrae superioris and may become apparent within 2 to 10 days of treatment and last approximately 2 to 4 weeks. During 1 clinical trial, blepharoptosis was reported to occur with a frequency of 5.4%. Overaggressive treatment of the frontalis muscles may also result in upper eyelid ptosis as well as brow ptosis. Eyelid ptosis may be mistaken for myasthenia gravis. It has been my impression that most incidences of eyelid ptosis after BoNTA injection are caused by unmasking underlying ptosis, which becomes more apparent after the reduction in muscle tone of the forehead together with the reduce compensatory lifting of the brow and upper eyelid. After the frontalis is weakened, preexisting ptosis becomes more obvious.
The injection of BoNTA in the lower eyelid area may adversely affect the function of the orbicularis oculi muscle. Although uncommon and unrecognized in more than 30,000 injections by me, lagophthalmos after aesthetic procedures is reported to result from paralysis of the orbicularis oculi muscles, which decreases blink strength, and tonic orbicularis contraction, causing dry eyes. The resulting corneal exposure may cause superficial punctate keratitis. Dry eyes in theory can also result from ectropion or abnormal eversion of the lower eyelid so that the inner surface is exposed ; however, a significant amount of neurotoxin would need to be injected periocularly and into the lower lid for this to occur. If it does occur, ectropion requires prompt medical attention to prevent exposure keratitis and corneal injury.
BoNTA injected into the periocular area can also adversely affect tear production, requiring the use of lubricating eye drops. This situation is a result of obstruction of the tear canal caused by paralysis of the orbicularis muscle. The effect of periocular BoNTA injection on tear production was studied in 13 women undergoing bilateral treatment of crow’s feet. The Schirmer test was used to assess tear production. Among the 26 treated eyes, 5 eyes of 3 patients had a significant decrease in Schirmer test results 1 month after injection and 3 eyes of 2 patients had a significant decrease in Schirmer test results at 4 months after injection; however, whether this is clinically significant is debatable, because only 1 patient reported dry-eye symptoms at 4 months.
Injection of BoNTA into the superior rectus muscle for the treatment of strabismus was one of the first therapeutic uses of botulinum toxins. By injecting the toxin into the stronger muscle and weakening it, proper alignment of the eyes can be achieved. Temporary strabismus may also occur after BoNTA injections of the lateral canthal areas as a result of diffusion, causing weakness of the lateral rectus muscles.
Upper lip ptosis has rarely been reported after the injection of BoNTA into the lateral canthal creases and orbicularis oculi muscle and may be related to prior blepharoplasty. In these cases, evidence of lip weakness began after 7 to 10 days and resolved after 5 to 6 weeks. Lip weakness can also occur from perioral injections to soften radial perioral lines and lateral canthal areas. Correction of chin dimpling or peau d’orange skin can be achieved by injecting BoNTA into the medial mentalis muscle; however, lateral injection placement may affect the depressor labii muscles and result in lower lip ptosis.
Large doses of BoNTA injected into the platysma for the treatment of platysmal bands and horizontal neck lines can produce weakness of the neck flexors and dysphagia and hoarseness ; however, routine administration of 30 to 40 units has not resulted in adverse events.
Antibody formation
Formation of antibodies to abobotulinumtoxinA and onabotulinumtoxinA after cosmetic applications has occurred in only a few case reports and seems to be more common with medical applications. Although not a danger to the patient, the development of neutralizing antibodies in theory may limit the therapeutic effectiveness of BoNTA injections. In 1 report, therapeutic failure caused by the development of BoNTA antibodies occurred in 4 patients after 3 to 13 injection sessions over 18 to 65 months, whereas a large meta-analysis reported that 11 of 2240 treated patients (0.49%) became positive for onabotulinumtoxinA antibodies, but only 3 patients became clinically unresponsive to onabotulinumtoxinA.
Ultraviolet light exposure
Using the time course of anhydrosis after intradermal injection of BoNTA, excessive exposure to UV-B radiation reduced the area of sweating by approximately 30%, although the duration of effect remained unchanged. These investigators suggested that exposure to UV-B radiation may result in denaturation or degradation of BoNTA.
Preventing common adverse events
To minimize the risk of bleeding and bruising, medications that may increase bleeding such as aspirin and nonsteroidal antiinflammatory drugs should be stopped at least 10 days before receiving BoNTA injections. If a blood vessel is inadvertently punctured, applying immediate pressure on the injection site may help minimize the development of widespread ecchymosis.
Injection discomfort may be reduced by the use of small-diameter needles, such as 30-gauge to 34-gauge, and changing the needle after 3 or 4 injections. It has been suggested that pinching the skin and the underlying muscle to be injected and slowly injecting the BoNTA solution may help reduce injection pain. Using a similar technique was reported to significantly reduce pain and eliminate unpleasant crunchy sensations.
The application of ice packs on the intended treatment area may also help minimize pain. In 1 study, the application of ice packs 5 minutes before periocular BoNTA injection decreased pain scores by 45%. In another study, the application of ice packs 5 minutes before injecting the lateral canthus with BoNTA significantly diminished pain; however, others have reported that cooling provides only minimal pain relief from periocular BoNTA injections. Topical application of an anesthetic cream has also been suggested to decrease injection pain. The topical application of lidocaine 4% cream significantly reduced the pain associated with the injection of BoNTA in the lateral canthal areas.
It has been suggested that differences in the diluents used to reconstitute BoNTA products may affect injection pain. The results of 2 studies showed that reconstitution of BoNTA using a preservative-containing saline solution significantly decreased the perception of pain perception during multiple BoNTA injections.
Prevention of complications
It has been anecdotally reported that applying digital pressure between the injection site and the eye may reduce the spread of BoNTA. When injecting the glabellar area, the spread of BoNTA may be reduced by squeezing the corrugator muscle between 2 fingers while injecting, although this has not been proved, to my knowledge. Placing corrugator injections 1 cm or more above the supraorbital ridge and using smaller injection volumes also reduces the spread of BoNTA and minimizes the occurrence of eyelid ptosis. The benefit of remaining upright for several hours after treatment remains unclear and likely has no effect on the toxin spread.
Patients with preexisting lower lid laxity such as the elderly may be at greater risk for ectropion. Before periorbital injections are administered, the snap test or lower lid extraction test may help identify patients at risk. Periorbital complications can also be minimized by injecting outside the infraorbital rim or beyond 1 cm lateral to the lateral canthus. Strabismus and diplopia are unlikely during cosmetic injections and are caused by unwanted lateral rectus weakness; these conditions can be guarded against by avoiding injections within the orbital rim and perhaps by applying digital pressure to isolate the drug while injecting. Patients experiencing strabismus or diplopia may require an ophthalmology consultation.
Lip dysfunction may be avoided by minimizing the number of perioral injections and keeping BoNTA injections 5 mm apart. Noticeable brow ptosis can be avoided by conservatively treating the medial forehead and foregoing treatment of the lateral forehead in those with low brows; however, patients with lateral frontalis muscle hyperactivity may develop brow arching, which results in a joker face. These patients may require a dose of 1 to 2 U of onabotulinumtoxinA or 3 to 6 U of abobotulinumtoxinA just over the lateral brow. Close examination before treatment should identify at-risk patients.
Dermal Fillers
Background
The most commonly used dermal fillers are made of hyaluronic acid, a glycosaminoglycan consisting of repeating units of glucuronic acid and N -acetyl-glucosamine. When combined with water, hyaluronic acid forms a viscous gel. Because normal human skin consists to a large extent of hyaluronic acid, it is well suited for use in dermal fillers. Several hyaluronic acid–containing dermal fillers are commercially available (Restylane and Perlane, Medicis Aesthetics, Scottsdale, AZ; Juvéderm Ultra Plus, and Juvéderm Voluma, Allergan, Irvine, CA). These fillers are chemically similar but differ in their physical properties because of different manufacturing processes. Hyaluronic acid dermal fillers may also contain lidocaine (Belotero and Belotero Balance; Merz Aesthetics and Juvéderm Ultra XC; Medicis Aesthetics, Scottsdale, AZ). Hyaluronic acid–containing dermal fillers are not permanent, because they undergo slow degradation by endogenous hyaluronidase.
Calcium hydroxylapatite (Radiesse, Merz Aesthetics) is a suspension of 20-μM to 45-μM particles in a methylcellulose gel. When used for augmentation, it lasts for 10 to 14 months, but its duration has been reported to range significantly longer and shorter than this. Poly- l -lactic acid (Sculptra Aesthetic, Sanofi-Aventis US) is a synthetic peptide polymer with particles sizes of 40 to 60 μM. Both calcium hydroxylapatite and poly- l -lactic acid stimulate collagen neogenesis and are considered semipermanent. One product containing nonresorbable polymethylmethacrylate microspheres suspended in bovine collagen (Artefill, Suneva Medical) is considered a permanent facial filler. It also contains lidocaine.
Contraindications
Contraindications to the use of dermal fillers include hypersensitivity to product components, bleeding disorders, especially with use of cannulas, and a history of anaphylaxis or presence of multiple severe allergies. Because hyaluronic acid–containing products are made by bacterial fermentation, a history of allergy to gram-positive bacterial proteins has been suggested to be a contraindication to their use; however, this seems to be hypothetical. Polymethylmethacrylate is contraindicated for use in lip augmentation and injection into the vermilion or the wet mucosa of the lip. Polymethylmethacrylate and poly- l -lactic acid should not be used in patients with known history of or susceptibility to keloid formation or hypertrophic scarring.
Common adverse events
Similar to treatment with toxins, the introduction of needles into tissue may be associated with local pain, erythema, edema, and ecchymosis. Patients should be cautioned that that these responses may occur but are generally short-lived ; however, these can be reduced or eliminated by a proper injection technique.
I have avoided much of the discomfort associated with dermal filler injection by adopting the use of blunt-tip cannulas (SoftFil, AlphaMedix, Petah Tikva, Israel) for all areas of the face. These cannulas permit deeper placement of fillers, resulting in more immediate results, and virtually eliminate discomfort, bruising, and swelling. The cannula I prefer using is 22-gauge and 70 mm long. As the cannula advances through the facial plane, it gently pushes underlying soft tissue out of the way. By thinning products with 1% lidocaine (0.3 mL), it becomes more malleable and it can be massaged into place after deep injection.
The decision whether to use an anesthetic depends on the patient, the product, and the area to be treated. Some products are manufactured with 0.3% dry preservative-free lidocaine, which significantly reduces injection-related discomfort ; however, in my practice, I premix all filler products with 1% lidocaine. In addition to achieving an anesthetic effect, the addition of lidocaine decreases filler viscosity, further alleviating discomfort. By using blunt-tip cannulas, there is little discomfort when treating cheeks and tear trough, making anesthetics unnecessary.
For very pain-sensitive patients, I use a regional nerve block when treating the lower one-third of the face and especially the lips. I start by applying 2% benzocaine paste (Hurricane, Beutlich Pharmaceuticals, Waukegan, IL) with a cotton swab along the oral buccal mucosal. Five minutes later, I apply 4 intraoral injections of mepivicaine HCl 3% (Isocaine, Henry Schein, Melville, NY) (2 superior and 2 inferior), anesthetizing the labial branches of the inferior orbital nerve and the mental nerves. Mepivicaine is short acting (∼30 minutes) and better tolerated than lidocaine.
A useful technique for avoiding pain is applying a vibrating nerve distracter near and above the injection point during anesthetic injection. Patients are often are unaware that they have been injected. For patients who are resistant to nerve blocks, I apply topical 30% lidocaine cream (SCP, Sherman Oaks, CA) for 20 to 30 minutes. This technique is especially effective for use on the lips, where I place the cream into a special mouth guard, which keeps the product over the labial mucosa.
Complications
Although occurring with an incidence of only 0.001%, focal necrosis has been associated with dermal fillers of all types and represents a serious complication. The vascular nature of the glabella makes it the most common site of necrosis caused by intra-arterial injection of dermal fillers, but intra-arterial injection in the nasolabial fold has also been reported. Inadvertent intra-arterial injection of a polymethylmethacrylate dermal filler in the glabellar area for soft tissue augmentation resulted in blindness and total ophthalmoplegia. These investigators speculated that the injected material entered a peripheral branch of the ophthalmic artery or some anastomosing artery, from where it traveled to the ophthalmic artery and reached the retinal central artery and anterior and long posterior ciliary arteries, where it resulted in blindness, corneal and iris ischemia, and total ophthalmoplegia.
In my experience, the appearance of impending tissue necrosis occurs immediately, within 24 to 48 hours, or is delayed. Occlusion caused by direct intravascular injection results in immediate blanching. Although aspirating before injecting may help prevent intravascular injection, it is difficult to perform with the current filler products. I find it helpful to inject slowly (less than 0.3 mL/min), keep the needle in motion, and avoid injecting near large facial vessels. As mentioned earlier, I premix filler products with 0.3 mL of lidocaine before injection to decrease its viscosity and minimize the risk for occlusion; however, the likely cause of reported necrosis is tissue pressure secondary to local edema or the hydrophilic properties of the product-occluding vessels. For this reason, the onset of symptoms may be delayed 24 to 48 hours.
I have modified a previously published protocol for the recognition and treatment of impending necrosis from soft tissue filler injection with satisfactory results ( Table 1 ). Blanching followed by a dusky or purple discoloration of the injected area indicates vascular compromise. Regardless of filler type, the injection should be immediately stopped and 10 to 30 units of hyaluronidase diluted 1:1 with saline should be injected into each 2-cm × 2-cm area of affected tissue. Because hypersensitivity reactions can occur, skin should be tested before hyaluronidase is injected. Hyaluronidase is also effective for reducing rejection-induced edema. It is likely that an inflammatory response causes an increase in hyaluronan via cytokines and growth factor, which is then mitigated by the hyaluronidase. It is for this reason that I believe it to be effective for the treatment of impending necrosis caused by all filler products, regardless of type.
| Presentation | Immediate or early blanching followed by a dusky or purple discoloration of the area |
| Treatment | Discontinue injection Perform hyaluronidase skin testing Inject 10–30 U of hyaluronidase per 2-cm 2 area Massage 1.25 cm (0.5 inch) of 2% nitroglycerin paste into the area and apply warm compresses Begin aspirin 325 mg and an antacid regimen Apply topical oxygen cosmeceutical therapy twice daily |
| Additional management | Follow patient daily for further signs of occlusion/necrosis Continue hyaluronidase and 2% nitroglycerin paste daily as needed Continue aspirin, antacid, and topical oxygen therapy until wound has healed If edema progresses, place on tapering doses of methylprednisolone Consider hyperbaric oxygen for progressing necrosis resistant to above treatments |
The application of 2% nitroglycerin paste (Nitro-BID, E. Fougera, Melville, NY) to the area of impending necrosis may also be beneficial. The product should be applied daily to the affected area until the capillary refill rate exceeds 2 seconds; however, the use of large amounts of 2% nitroglycerin paste (>1.25 cm [0.5 inch]) may cause systemic effects. Warm compresses may also provide vasodilatation, and daily aspirin may prevent further clot formation. Patients are followed daily for further signs of occlusion or necrosis. If edema is slow to resolve, methylprednisolone is added. Hyperbaric oxygen should be considered if the affected area does not respond to these treatments ( Fig. 1 ).
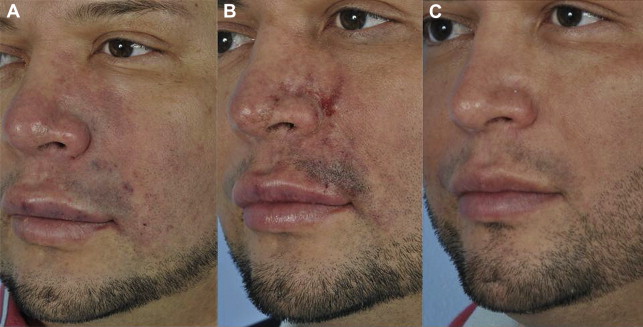
At one time, the reported rate of hypersensitivity reactions to hyaluronic acid was 0.07%, but it has decreased to 0.02%, because more highly purified products contain lower amounts of protein. Immediate hypersensitivity reactions are rarely severe and are more likely to occur with bovine-derived products. Immediate hypersensitivity reactions are also more common with products containing a local anesthetic. Reports of angioedema are rare.
Many reactions that are assumed to be allergic or hypersensitivity responses are most likely caused by bacterial reactions. Local infections are rare but have been associated with the use of unapproved products. Biofilms can present as sterile abscesses or chronic inflammation and infections and may cause a foreign body response, resembling an allergic reaction. Treatment of early infectious complications from biofilm reactions included broad-spectrum antibiotics such as fluoroquinolone or macrolide antibiotics.
In 1 case report, poly- l -lactic acid was used to treat facial lipodystrophy associated with human immunodeficiency virus. Approximately 1 year after the last treatment, the patient complained of hot, hard, painful, and distorting bumps at the injection sites. Examination of the patient revealed warm, erythematous, and fibrotic skin and nodules located on treated areas, which were attributed to a delayed allergic reaction. I believe that hypersensitivity reactions are likely to have an infectious cause and I have developed a protocol for the prevention and treatment of hypersensitivity/infectious reactions ( Table 2 ).
| Before treatment | Cleanse the face thoroughly of all makeup before injection Use benzalkonium chloride wash or povidone-iodine (Betadine) swab to prepare the face just before the treatment Use as few injection sites as possible Avoid bolus injections Avoid injecting into previously placed fillers or through infected tissue Avoid injecting through oral or nasal mucosa Consider prophylactic antibiotics before a dental procedure or if a facial infection occurs within 2 wk of a filler treatment |
| If a red indurated area appears any time after the treatment, regardless of duration | Inject hyaluronidase regardless of filler used 10–30 units mixed 1:1 with saline (Vitrase, ISTA Pharmaceuticals, Irvine, CA) Start antibiotics: ciprofloxacin 500 mg QD (Cipro, Bayer HealthCare Pharmaceuticals, Wayne, NJ) and clarithromycin 500 mg twice daily (Biaxin, Abbott Laboratories, Abbott Park, IL) Avoid all forms of steroids or nonsteroidal antiinflammatory drugs |
| If long-term indurated area or steroids have already been used | Inject 15–40 mg fluorouracil (APP Pharmaceuticals, Schaumburg, IL) Repeat every 4 wk |
| If induration remains persistent despite fluorouracil treatment | Consider laser lysis Consider incision and washing out cavity with antibiotics Surgical excision as last resort |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree




