The primary function of the palate is to separate the nasal and oral cavities and facilitate the production of normal speech. The velar levator veli palatini (LVP) muscular sling is the palate speech motor. In the cleft palate, the LVP muscles are aberrantly sagittally oriented, which prevents their normal cranial, posterior, and lateral pull on the soft palate, resulting in velopharyngeal incompetence. Palatoplasty techniques enlist the same 3 principles: closure of the nasal mucosa, reorientation and repair of the LVP muscle sling, and closure of the oral mucosa. The primary outcome measures for palatoplasty are speech quality and palatal integrity.
Key points
- •
The palate is divided functionally into the hard palate and soft palate. The hard palate serves as the main structural separation of the oral and nasal cavities and is a growth center for the maxilla. The soft palate is the speech motor and main contributor to velopharyngeal competence.
- •
The levator veli palatini (LVP) muscles, which form a transverse muscular sling in the middle of the velum, are the principal motors of the velar component of velopharyngeal closure. In the cleft palate, the LVP muscles are sagittally oriented, running posterior to anterior, and insert onto the posterior edge of the hard palate. This aberrant orientation of the LVP muscles prevents their normal cranial, posterior, and lateral pull on the soft palate, and velopharyngeal competence is compromised.
- •
Cleft palate repair must include complete release of LVP muscles from their abnormal attachments to the (1) posterior edge of the hard palate anteriorly, (2) tensor veli palatini aponeurosis anteriorly, and (3) superior pharyngeal constrictor muscles laterally.
- •
Cleft palate is both a malformation and deformation defect with a spectrum of tissue deficiency. Tissue augmenting palatoplasty techniques, such as buccal myomucosal flaps, can serve as supplemental tissue to facilitate closure of the nasal and/or oral mucosa of the hard and/or soft palate without tension, the need for relaxing incisions, or hard palate dissection.
- •
Primary outcome measures for palatoplasty include speech quality and palatal integrity.
Introduction
The primary function of the palate is to serve as a physical separation of the nasal and oral cavities and to facilitate the production of normal speech. An overt cleft palate is both a physical defect of varying degree and improper orientation of the submucosal muscles of the velum. The incidence of cleft palate, taking into account racial and gender differences in predominance, ranges from 1:500 to 1:2000. While cleft palate etiology is multifactorial, including the complex interaction of genetic aberrations and environmental factors, there is but one universal surgical goal of palatoplasty, to restore the form and function of the palate. This entails reconstruction of the normal anatomic relationship of the palatal tissues and muscles within the velum, which in turn permits the dynamic function of the velopharyngeal valve and the production of speech. In order to adequately understand the various palatoplasty surgical techniques, it is essential to first understand the normal anatomy and dynamic function of the structures that comprise the palate. Herein, the authors will begin with a discussion of the anatomy relevant to palatoplasty, cleft palate classification, and timing of cleft palate repair. The range of surgical palatoplasty techniques is broad and has evolved over time. These techniques will be discussed in their historical context followed by a more detailed description of the current preferred surgical techniques of cleft palate repair.
Relevant anatomy
The palate, based on the nuances of palatal embryogenesis, is divided into a primary palate and secondary palate. The primary palate is the premaxilla, which includes the alveolus and bony palate anterior to the incisive foramen. The secondary palate is posterior to the primary palate and consists of the bony palate posterior to the incisive foramen and the soft palate/velum. The main blood supply to the hard and soft palate is the greater palatine artery and ascending palatine artery, respectively. The greater palatine artery enters the hard palate via a foramen at the lateral posterior edge of the hard palate. The ascending palatine artery passes around the superior pharyngeal constrictor muscle and below the levator veli palatini (LVP) muscle near the skull base to supply the soft palate.
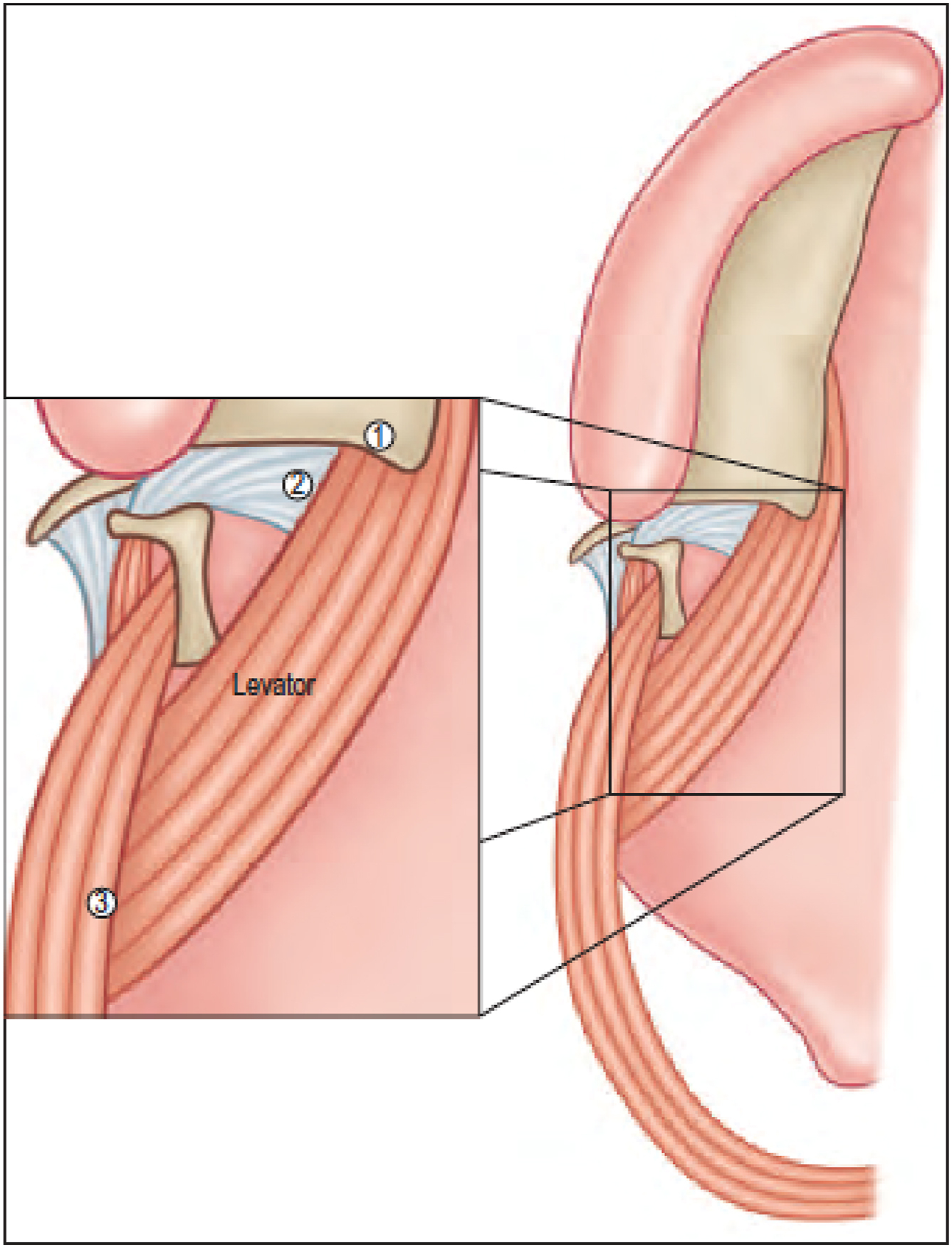
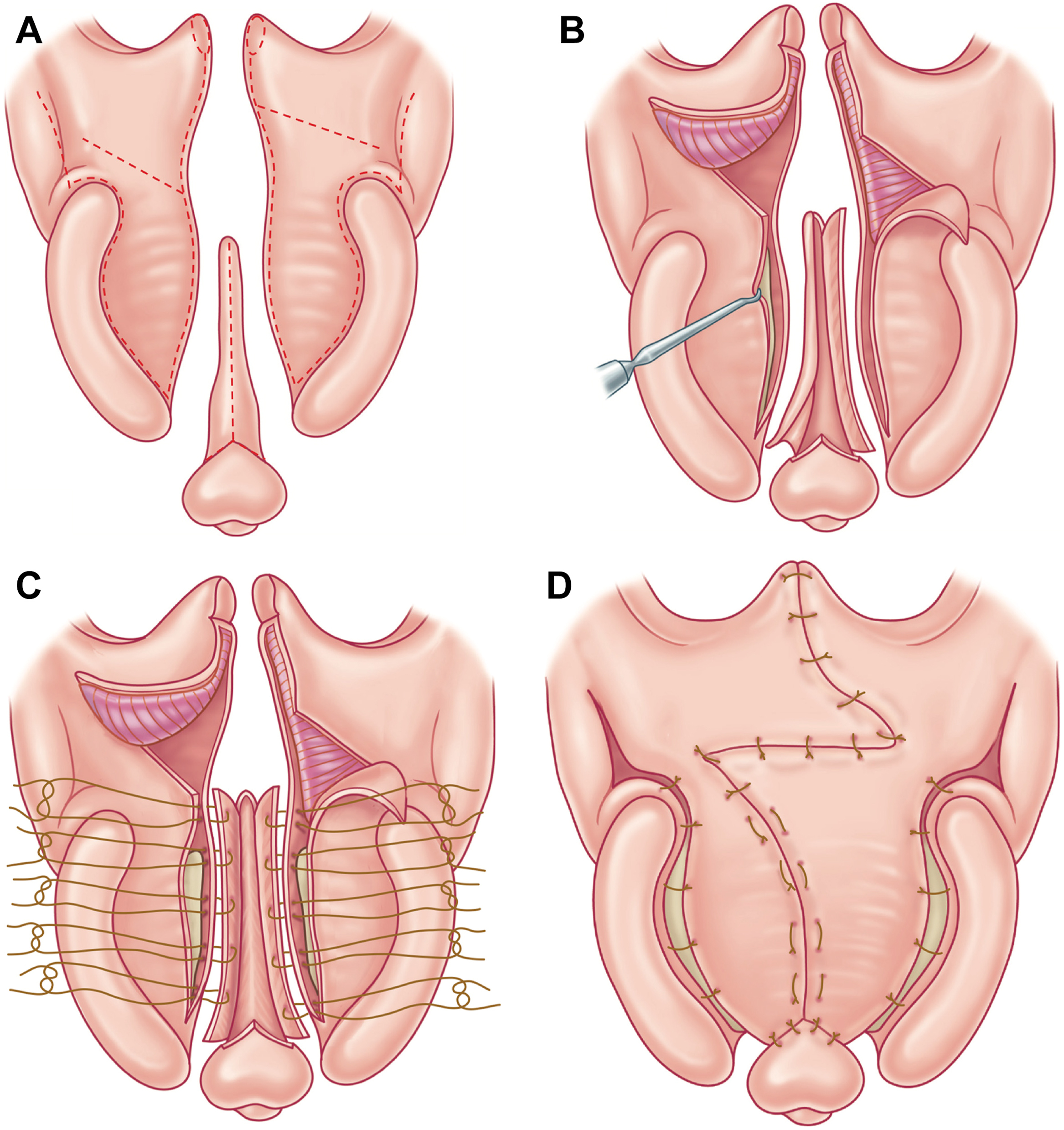
The soft palate is located posterior to the hard palate and consists of 5 paired muscles (LVP, tensor veli palatini, palatoglossus, palatopharyngeus, and muscularis uvulae) sandwiched between nasal mucosa cephalad and oral mucosa caudally ( Fig. 1 A ). From a functional perspective, the soft palate can be divided into anterior (25%), middle (50%), and posterior (25%) sections. The anterior 25% of the soft palate is relatively static and contains the tensor veli palatini (TVP) muscular aponeurosis. This TVP originates from the greater wing of the sphenoid bone and superolateral aspect of the eustachian tube, descends in a vertical trajectory to hook around the hamulus, and ultimately transforms into a fibrous aponeurosis that joins its contralateral counterpart in the anterior 25% of the soft palate (see Fig. 1 A; Fig. 2 ). Some opine that normal anatomic continuity and orientation of the TVP muscles is critical for the optimization of eustachian tube function. , The middle 50% of the soft palate houses the LVP muscles. These muscles originate from the cranial base, more specifically the posteromedial aspect of the eustachian tube, and travel in a caudal, anterior, and medial direction to insert into their contralateral partner muscle in the midline of the soft palate (see Figs. 1 A and 2 ). This creates a transverse/horizontal muscular sling that not only supports the soft palate at rest, suspending it from the cranial base, but also functions as the velar motor, powering palatal elevation during speech by exerting force on the soft palate in a cranial, posterior, and lateral direction. Continuity and normal transverse orientation of the LVP muscular sling is necessary for velopharyngeal competence, which is the soft palate’s ability to make contact with the posterior pharyngeal wall, preventing air escape through the nose during enunciation of specific phonemes. Others opine that proper anatomic continuity and orientation of the LVP muscles is also critical for the optimization of eustachian tube function. The palatopharyngeus and palatoglossus muscles are also found in the middle 50% of the soft palate. The palatopharyngeus muscle boasts an inferior head that takes origin from the oral surface of the LVP muscle and a superior head that takes origin from the nasal surface of the LVP and courses posteriorly and inferiorly as the posterior tonsillar pillars. , The palatoglossus muscles originate from the dorsolateral transverse fibers of the tongue and travel cephalad as the anterior tonsillar pillars before fanning out and inserting in the middle 50% of the soft palate oral to the LVP muscles as the most superficial muscle of the velum. , The posterior 25% of the soft palate contains the muscularis uvulae muscles. The muscularis uvulae muscle takes origin from the anterior aponeurosis of the TVP muscles and courses posteriorly within the palate as a paired midline structure, inserting at the base of the uvula (see Fig. 1 A).
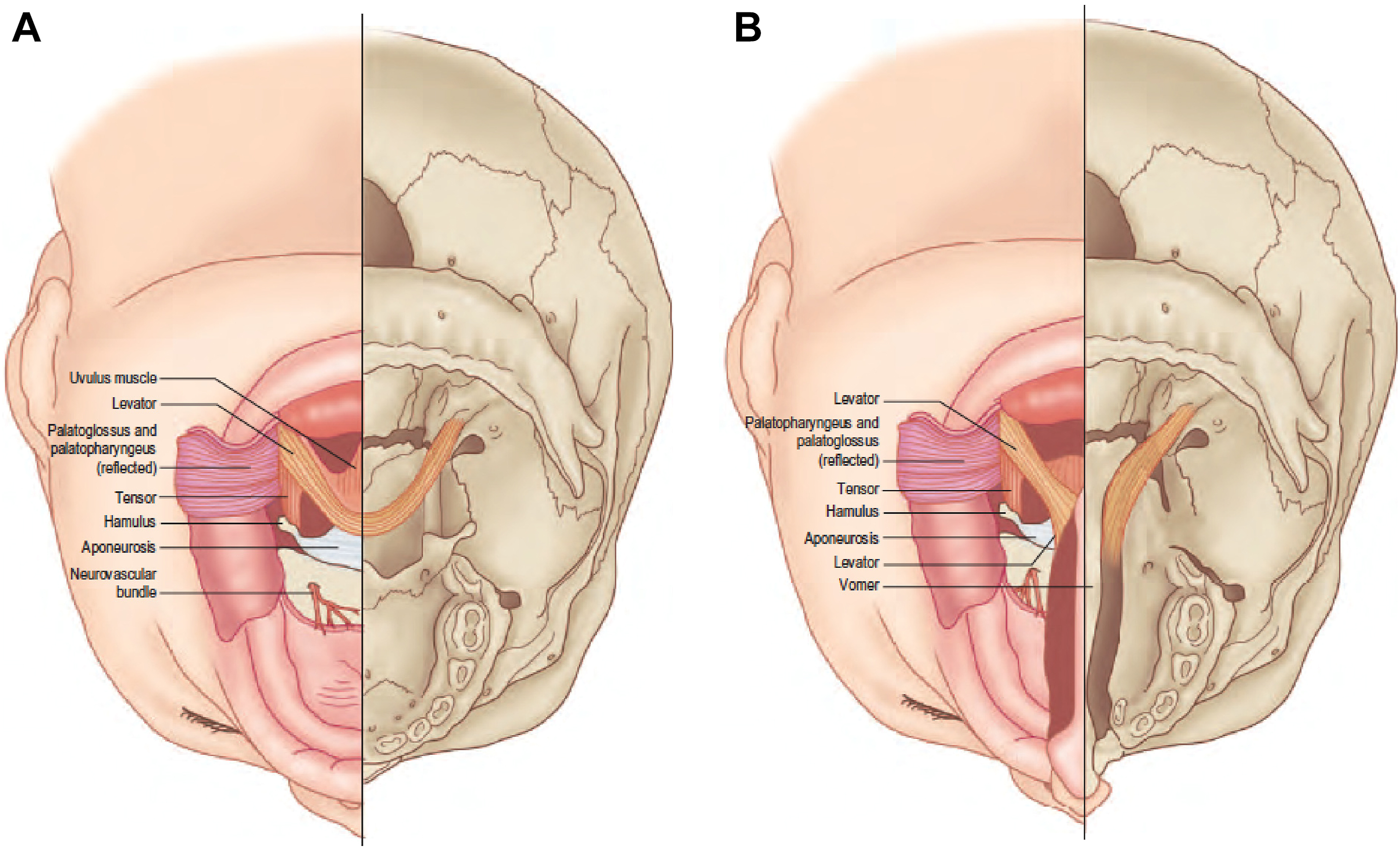
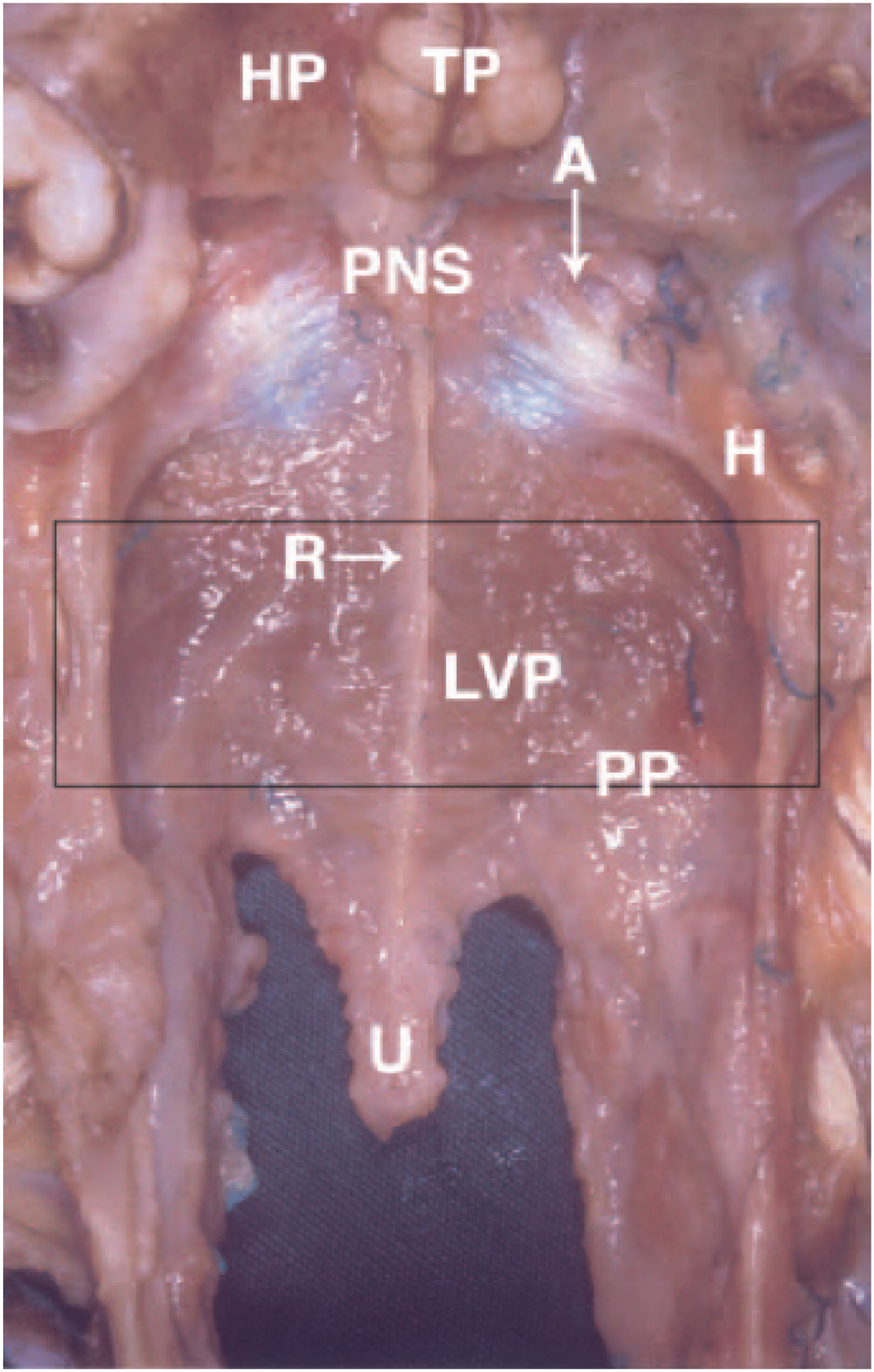
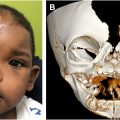
In the cleft palate, the normal anatomic paths of the muscles housed within the velum are disrupted. In particular, the LVP muscles, which are arguably the most important soft palate muscles for velopharyngeal competence, are no longer joined with one another transversely but are instead positioned sagittally from posterior to anterior boasting 3 abnormal attachments: (1) posterior edge of the hard palate anteriorly, (2) TVP aponeurosis anteriorly, and (3) superior pharyngeal constrictor muscles laterally ( Figs. 1 B and 3 ). When oriented in this clefted way, the LVP muscles are unable to fully exert their normal cranial, posterior, and lateral pull on the soft palate and velopharyngeal competence is compromised.
Cleft palate classification
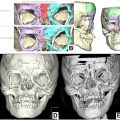
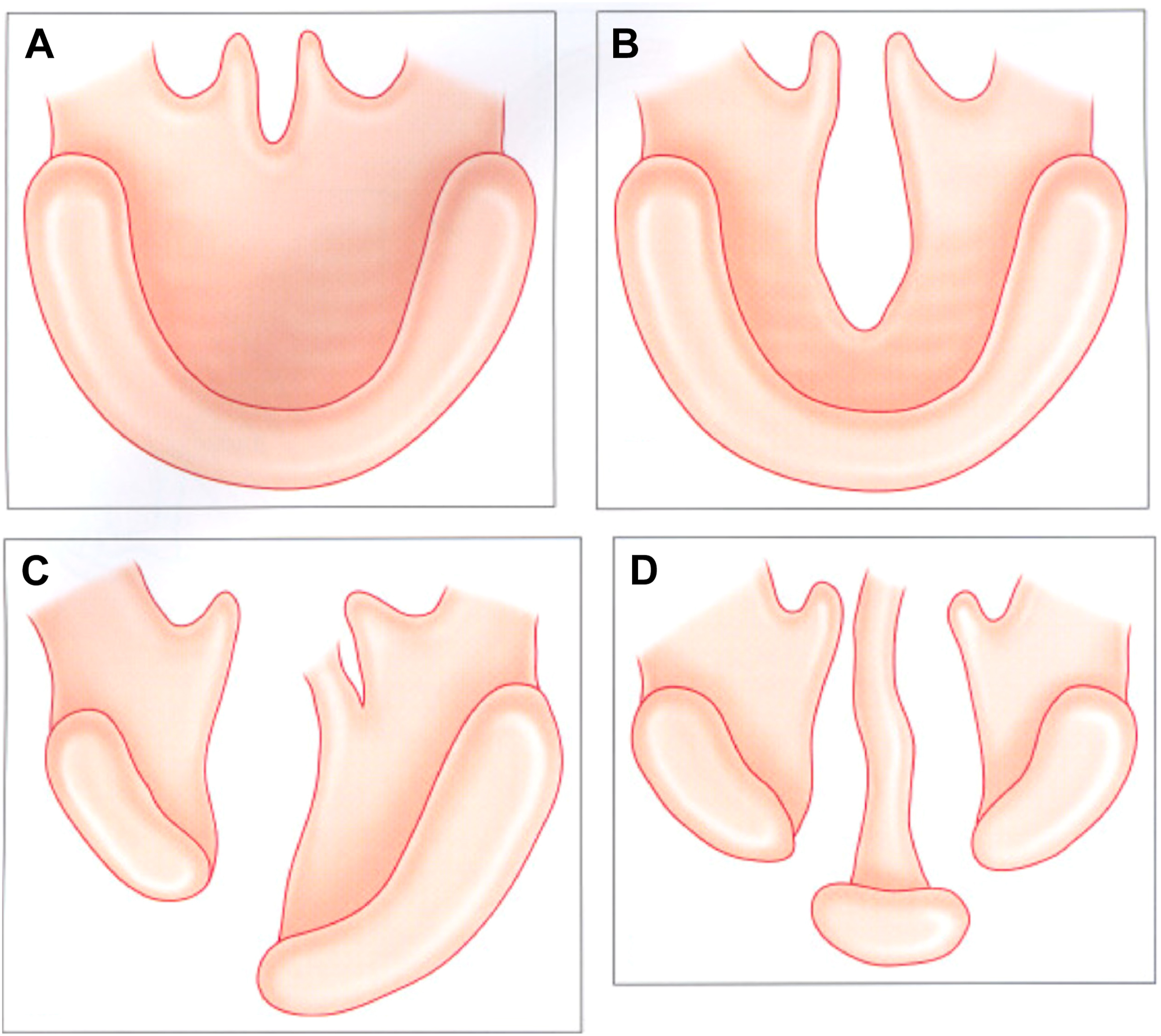
Multiple classification systems exist for describing overt clefts of the palate. The Veau classification is both simple and intuitive and is the author’s preferred classification system based on these attributes and the fact that it is derived from the embryologic origins of cleft palate. The Veau classification system denotes 4 distinct types of overt cleft palate ( Fig. 4 ). Veau I cleft is an isolated cleft of the soft palate. Veau II cleft is a cleft of the soft palate and hard palate (to some variable degree). Veau III cleft is a complete unilateral cleft of the alveolus, hard, and soft palate. Veau IV cleft is a complete bilateral cleft of the alveolus, hard, and soft palate. One type of cleft palate that is not addressed by the Veau classification system is submucous cleft palate (SMCP). SMCP occurs when the muscles of the soft palate are clefted and improperly oriented (to some variable degree), but the oral and nasal mucosas between which they are located are intact. This is classically characterized by the presence of a bifid uvula, a zona pellucida within the soft palate, and a notch at the posterior edge of the hard palate. These 3 static/nondynamic clinical signs are typically necessary to diagnose an overt SMCP. SMCP, however, can also be detected by the presence of a dynamic vaulted/V-shaped appearance of the soft palate with elevation, as with phonation or with gag. The V-shaped appearance of the velum is a visual indication of the aberrant orientation and attachments of the underlying LVP muscles, which are abnormally sagittally oriented from the skull base toward the posterior edge of the hard palate. An occult SMCP is an SMCP that lacks the typical static clinical signs (bifid uvula and zona pellucida) and is only revealed with the dynamic vaulted/V-shaped pattern of palatal elevation on velar activation.
Timing of cleft palate repair
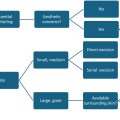
Functionally, the palate is divided into the hard palate, which serves as the main structural separation of the oral and nasal cavities (and a growth center for the maxilla) and the soft palate, which is the speech motor and main contributor to velopharyngeal competence. Similarly, considerations regarding cleft palate repair are often divided into repair of hard palate and repair of the soft palate. Because maxillary growth and speech development do not occur in unison, there is significant debate as to the ideal timing of cleft palate repair. Some argue that by 1 year of age, an unrepaired soft palate cleft will compromise speech development and, thus, favor early palatoplasty (repair by 9–12 months of age). Others argue that early palatoplasty, given its surgical manipulation of the hard palate, can disrupt midface growth and, thus, favor late palatoplasty (repair after 18 months of age). Others attempt to uncouple the possible detrimental effects of late soft palate repair (compromised speech development) from those of early hard palate repair (maxillary growth restriction) and favor various methods of 2 stage cleft palate repair. The authors hold that while the optimization of speech development and minimization of maxillary growth restriction must both be taken into account when approaching cleft palate repair, optimization of normal speech development must take priority as the decreased neural plasticity associated with increased age makes addressing disorders of speech significantly more challenging than addressing possible maxillary growth restriction, which can be corrected with orthognathic surgery at the time of skeletal maturity. It is the authors’ standard practice to repair Veau III and IV palatal clefts in a 2 staged manner: (1) repair of the hard palate via a single-layer vomer flap closure at the time of cleft lip repair, and (2) soft palate repair as a second-staged operation at approximately 1 year of age. This technique, which will be detailed further, attempts to minimize the risk of maxillary growth restriction, by negating the elevation of hard palate mucosal flaps with lateral relaxing incisions, while still ensuring soft palate repair by the time of normal speech and language acquisition.
Evolution of palatoplasty
In order to advance the field of palatoplasty, the surgical history must first be understood. It is the senior author’s opinion that the development of palatoplasty can be divided into 5 evolutionary phases, beginning with attempts to simply occlude the palatal defect with the use of an obturator, to our current evolutionary phase of tissue augmentation palatoplasty ( Box 1 ). The first published report of cleft palate repair was by Le Monnier in 1764, in which a raw edge was made at the medial cleft margins and the edges were sutured together. The first reported hard palate repair was by Johann Friedrich Dieffenbach in 1828. Based on these and other reports of early attempts at cleft palate repair, it became readily apparent that functional outcomes were poor when palatoplasty was limited to simply pulling marginal cleft tissue together under tension. The first modern report of cleft palate repair was by von Langenbeck in 1859, in which repair was achieved by the creation and medialization of bipedicled mucoperiosteal flaps. The von Langenbeck technique was later modified by Veau, Thomas Kilner, and William Wardill by instituting an anterior V-Y closure of the hard palate mucoperiosteal flaps, leaving a large anterior raw surface to heal by secondary intention, in an attempt to increase palatal length (Veau-Wardill-Kilner VY pushback). While this modification led to a slight decrease in rates of velopharyngeal insufficiency (VPI), speech outcomes remained suboptimal. This was likely due to both inadequate length of the velum (VY pushback was ultimately limited by immobility of the palatal neurovascular pedicles), and poor velar function because of not reconstructing the LVP muscular sling and reorienting the LVP muscles to their normal anatomic position.
- 1.
FORM : Occlude the defect—“mind the gap”
- 2.
STATIC FUNCTION : Midline closure and attempting to lengthen the palate
- 3.
DYNAMIC FUNCTION : The Levator Veli Palatini (LVP) and intravelar veloplasty
- 4.
FORM/FUNCTION : Furlow double opposing Z-plasty
- 5.
TISSUE AUGMENTATION/REPLACEMENT
Awareness of the anatomy, importance, and impact of normal orientation of the LVP muscles on postpalatoplasty speech outcomes came into focus starting in the 1960s. , The concept of an intravelar veloplasty (IVVP), correcting the abnormal sagittal orientation of the LVP muscles and reconstructing the LVP sling by repositioning them transversely in the middle 50% of the velum, was reported and popularized. In 1994, Cutting and Somerland described the importance of what they termed a radical IVVP. John Grant later published data showing improved speech outcomes and a significant reduction in secondary speech surgery, with radical versus conventional IVVP. He also showed improvement in speech with secondary revision palatoplasty with a radical IVVP for patients with unsatisfactory speech outcomes following primary palatoplasty.
The next largest advancement in the evolution of palatoplasty came with the development of the double opposing Z-plasty repair of the soft palate by Leonard Furlow in 1976. The beauty of this operation is its ability to address both the form and function of the palate with simultaneous lengthening of the soft palate and reorientation of the LVP muscles to their correct anatomic position with less dissection of the LVP muscles from the mucosa compared to traditional forms of IVVP. Furthermore, by avoiding the traditional straight-line incision and closure of the soft palate, this technique results in less scar contracture and postoperative shortening of the palate over the course of the healing process. Given the intrinsic nature of a z-plasty, which increases the length by recruiting from the width, the double opposing Z-plasty provides a secondary pharyngoplasty effect with velopharyngeal port constriction, which aids in achieving velopharyngeal competence. One caveat to this is that, when applied to wide clefts, the Furlow palatoplasty technique may require lateral relaxing incisions to achieve midline closure, although this has not been shown to compromise postoperative outcomes.
The final and current evolutionary phase of palatoplasty is that of tissue augmentation. Historical doctrine has been that a cleft palate harbors normal tissue quantity (normal formation) but abnormal tissue configuration (failure of fusion). Many cleft surgeons today, however, readily appreciate a spectrum of tissue deficiency in cases of cleft palate, suggesting that cleft palate is actually both a malformation and deformation defect. Previous palatoplasty techniques have not addressed the paucity of palatal tissue and have instead relied on relaxing incisions to facilitate repairs under tension. These techniques, used to approximate available palatal tissue, leave raw areas left to scar, and in the case of the hard palate, can result in maxillary growth restriction. In addition to the paucity of native palate tissue, the repositioning of the LVP muscles results in significant “dead-space” between the nasal and oral mucosal layers at the hard–soft palate junction. The volume of this “dead-space” has been estimated to be approximately 12 cm 3 . , If left unaddressed, this “dead-space” scars in and contracts, which can result in shortening of the palate, impaired palate mobility, and relapse migration of the LVP muscles anteriorly back to a sagittal orientation, all of which could compromise long-term velopharyngeal competence and impede the primary goal of cleft palate repair. , , Historically, the senior author addressed this paucity of native palatal tissue with the use of acellular dermal matrixes (ADMs). , While the use of ADMs can effectively augment and reinforce a tenuous mucosal lining repair, it is nonvascular and has the potential to induce additional scarring within the velum. Due to this potential risk, the senior author now solely uses ADMs for the prevention or repair of hard palate fistulae.
The most recent revolutionary advances in palatoplasty address the aforementioned paucity of native palatal tissue, tenuous palate repairs under tension, and “dead-space,” with tissue augmentation. This includes using the vascularized buccal fat pad flap (BFPF) and the buccal myomucosal flap (BMMF). Popularized by Levi and colleagues, the vascularized BFPF can be harvested and transferred to augment palate repair and obliterate dead space. The BFPF serves as a vascularized “dead-space” filler while also providing an additional barrier to fistula formation. The BMMF, while first described by Mukherji in 1969, and later by Kaplan, Maeda, and Jackson, has been traditionally used as an adjunct, along with pedicled facial artery musculomucosal flap (FAMM) flaps, for fistula repair. More recently, the benefit of utilizing BMMFs at the time of primary palatoplasty, to augment the palatal repair thus alleviating the need for relaxing incisions and providing tension-free repairs, has been realized. The BMMF can be designed and rotated in a variety of different configurations to serve as additional useable nasal and oral mucosa during cleft palate repair. Mann , demonstrated that the use of BMMFs in primary repair of the hard and soft palates can avoid the need for relaxing incisions, and the negative effects associated with them, even when repairing wider clefts with the Furlow technique. An additional benefit of utilizing the BMMF at the time of primary palatoplasty is the fact that the BMMF stretches as the patient grows. It is postulated that this built-in “tissue expander” can help prevent the later development of VPI that is sometimes seen in patients with initially successful palate reconstructions as they grow. In addition to their use at the time of primary palatoplasty and during fistula repair, BMMFs have also proven extremely useful in the treatment of VPI. In patients with a functional LVP muscle sling, secondary palatal lengthening can be achieved via a transverses full-thickness disjunction of the soft palate from the hard palate, and subsequent reconstruction of the resultant nasal and oral mucosal defects with bilateral BMMFs. Use of BMMFs in this manner results in functional velar lengthening and posteriorization of the LVP sling, and has the potential to nearly eliminate the need for pharyngoplasty (sphincter pharyngoplasty and pharyngeal flaps), removing the life-long risks of pharyngoplasty in the VPI population. ,
Operative techniques
All palatoplasty techniques enlist the same 3 principles: closure of the nasal mucosa, reorientation and repair of the LVP muscle sling, and closure of the oral mucosa. The specific palatoplasty technique employed to achieve these goals is determined by both the cleft anatomy and surgeon preference. Herein, we will describe in detail the most popular/classic hard and soft palate repair techniques, the authors preferred technique of staged palatoplasty, and details of tissue augmenting palatoplasty techniques.
Classic Palatoplasty
Hard palate flap design and markings
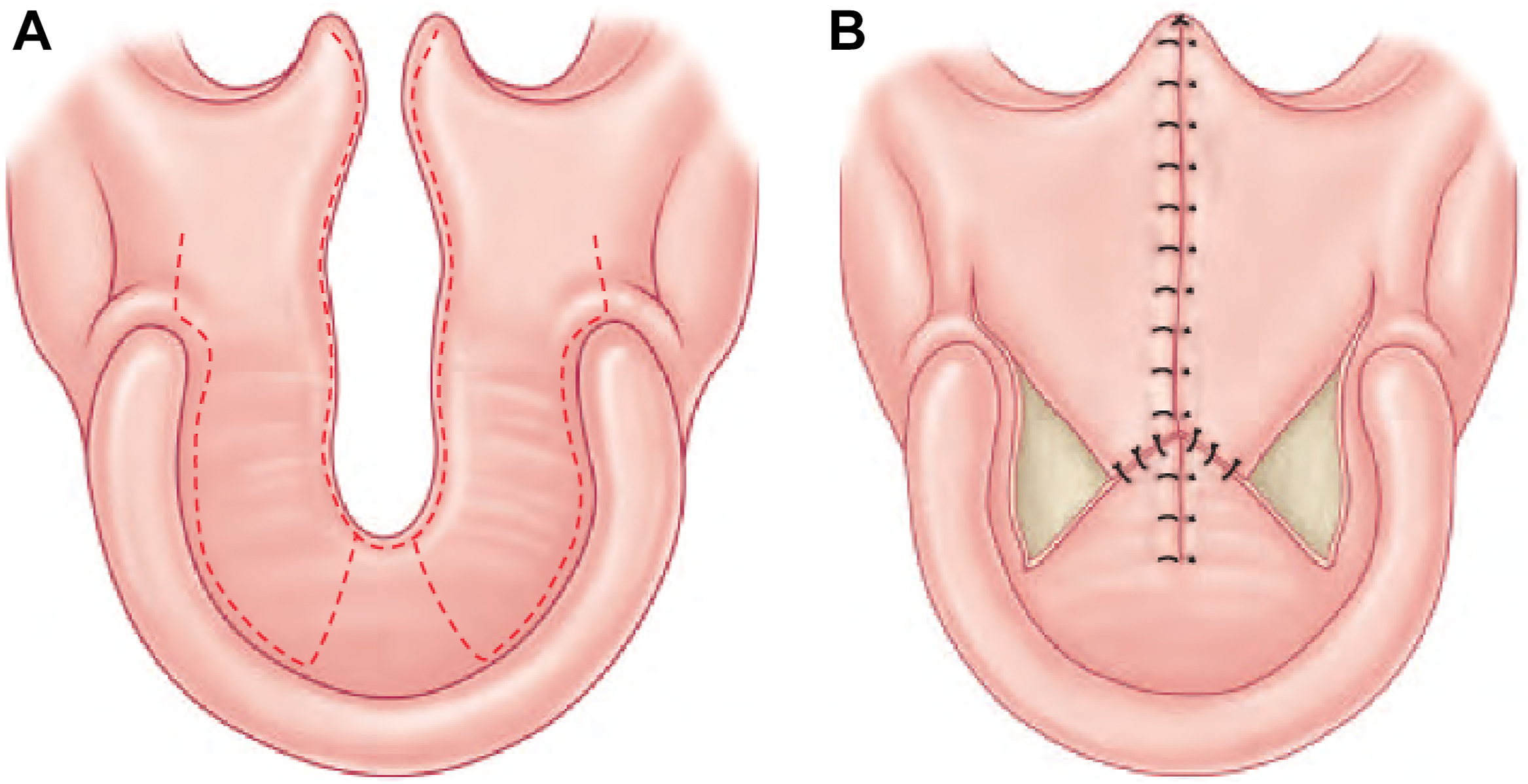
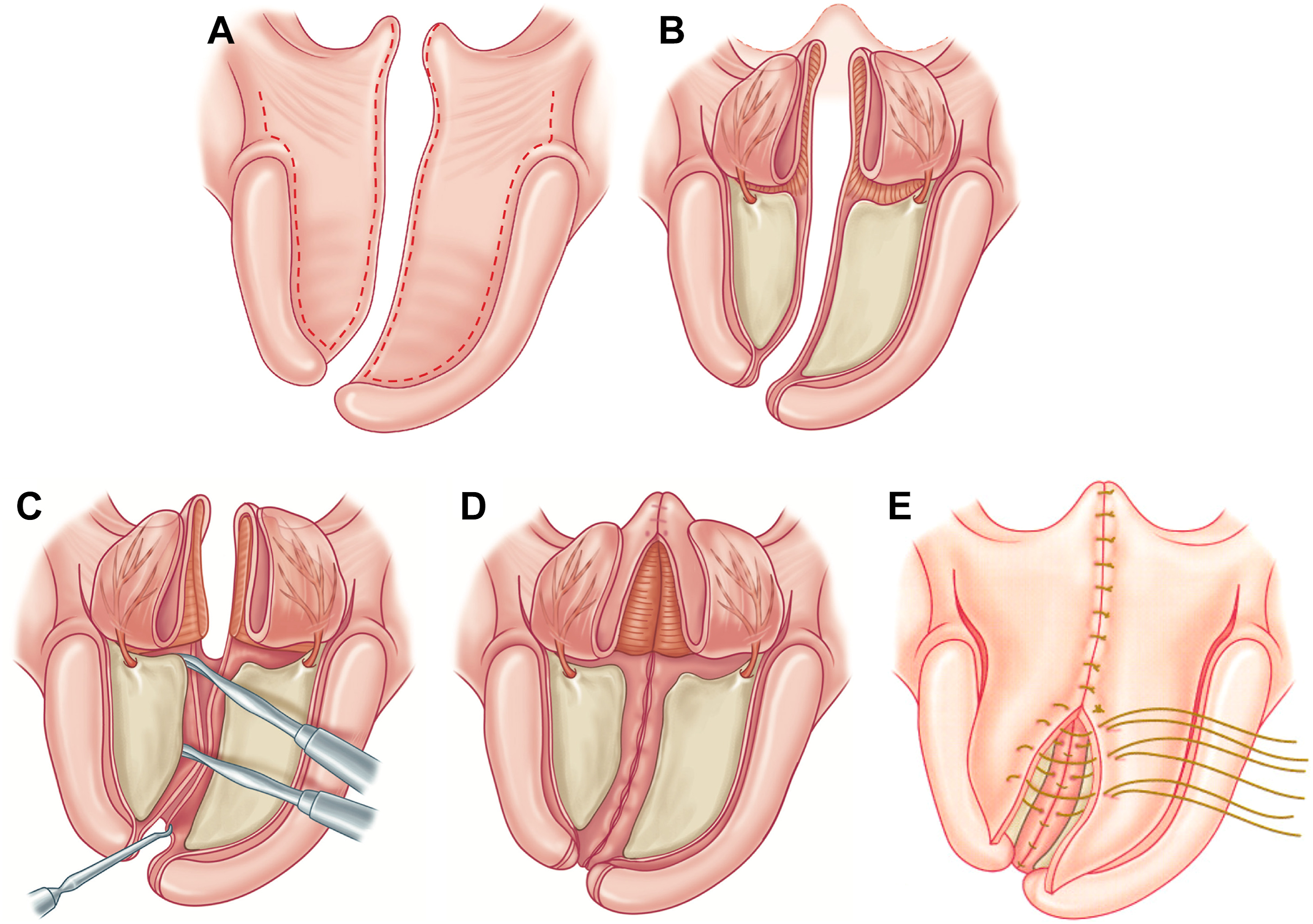
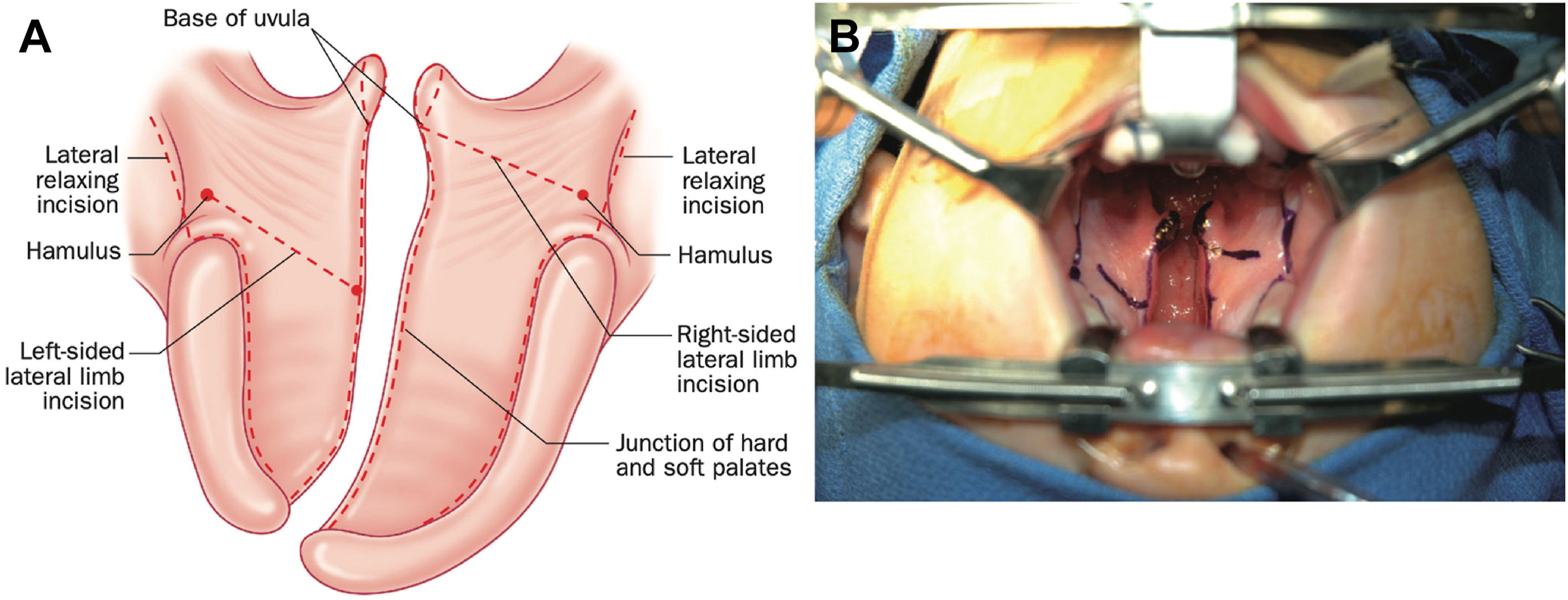
Repair of the hard palate is classically performed with elevation and medialization of mucoperiosteal flaps via bilateral lateral relaxing incisions along the junction of the hard palate and attached gingiva of the alveolus. These flaps can be designed as either unipedicled or bipedicled and are used in combination with vomer flaps for closure of the nasal mucosa. Occasionally for narrow or highly vaulted clefts, the hard palate flaps can be elevated from the medial margin and closed without the need for lateral alveolar relaxing incisions. Unipedicled flaps allow for increased flap mobility to repair wide clefts but require division of the anterior pedicle. Unipedicled flaps are employed in the Veau-Wardill-Kilner VY pushback repair ( Fig. 5 ) and Bardach’s 2 flap palate repair ( Fig. 6 ). , Bipedicled flaps require less dissection and result in a smaller final area of exposed hard palate, but afford less flap mobility. Two bipedicled flaps are typically used for repair of Veau II clefts. For Veau III clefts, a combination of either unipedicled and/or bipedicled flaps can be utilized. Commonly, a bipedicled flap is used on the major segment, and a unipedicled flap is used on the minor segment and rotated to span the cleft and offset the oral and nasal mucosa lines of closure ( Fig. 7 ). Two unipedicled flaps are commonly used for repair of Veau IV clefts, with each flap being inset into the premaxillary segment in order to minimize closure tension at the incisive foramen ( Fig. 8 ). Of note, if the premaxilla is positioned too anteriorly to permit complete oral and nasal lining closure at the anterior hard palate, premaxillary setback can be considered.

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree