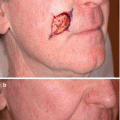
Fig. 16.1
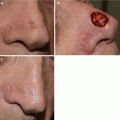
Biopsy proven lentigo maligna melanoma with ill-defined clinical borders
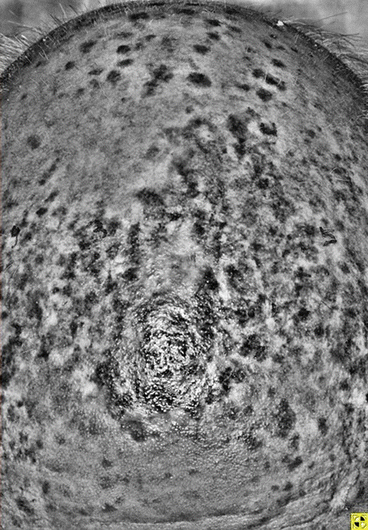
Fig. 16.2
Wood’s light examination of pigmented lesion
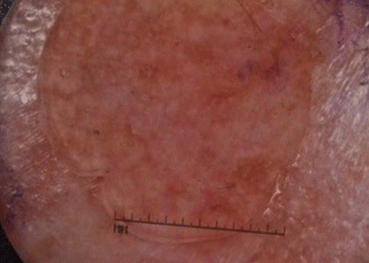
Fig. 16.3
Dermoscopic examination of pigmented lesion
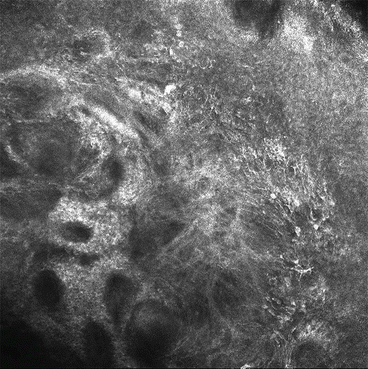
Fig. 16.4
Reflectance confocal imaging of pigmented lesion
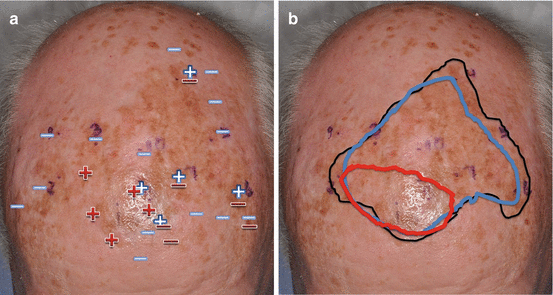
Fig. 16.5
(a) Scouting biopsies (Red:  negative for melanoma
negative for melanoma  positive for melanoma) guided by confocal microscopy (Blue:
positive for melanoma) guided by confocal microscopy (Blue:  negative for melanoma
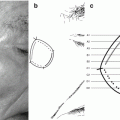
negative for melanoma  positive for melanoma). (b) Presurgical mapping of melanoma borders using Wood’s light examination (black line), dermoscopy and confocal microscopy (blue line), and scouting biopsies (red line)
positive for melanoma). (b) Presurgical mapping of melanoma borders using Wood’s light examination (black line), dermoscopy and confocal microscopy (blue line), and scouting biopsies (red line)
 negative for melanoma
negative for melanoma  positive for melanoma) guided by confocal microscopy (Blue:
positive for melanoma) guided by confocal microscopy (Blue:  negative for melanoma
negative for melanoma  positive for melanoma). (b) Presurgical mapping of melanoma borders using Wood’s light examination (black line), dermoscopy and confocal microscopy (blue line), and scouting biopsies (red line)
positive for melanoma). (b) Presurgical mapping of melanoma borders using Wood’s light examination (black line), dermoscopy and confocal microscopy (blue line), and scouting biopsies (red line)A staged excision was then performed. In the initial stage, the entire lesion plus a narrow margin was excised down to the deep dermis and superficial subcutaneous plane (Fig. 16.6) with orientation preserved with suture markings. The specimen was sent rush for paraffin-embedded pathology step sections from tip-to-tip in a breadloaf fashion. Because only a small portion of this large lesion was sampled, this sectioning approach allowed for comprehensive pathologic assessment, including additional information on tumor thickness and initial margin assessment (i.e. the distance of excision margin to the tumor, or its trailing edge). Pathology showed residual lentigo maligna with isolated atypical intraepidermal melanocytes focally within 1 mm of side margins. Based on these findings, a second and final staged excision was performed and sent for rush permanent sections to achieve sufficiently therapeutic lateral and deep margins (Fig. 16.7). Final pathology showed all margins were cleared with no melanoma seen.
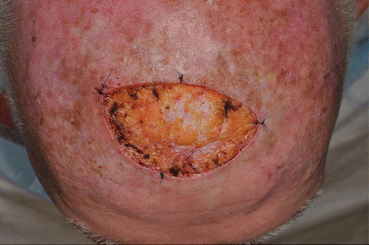
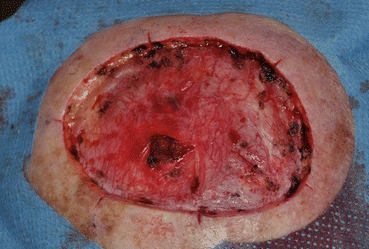

Fig. 16.6
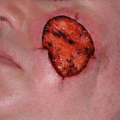
Surgical defect after initial staged excision

Fig. 16.7
Final excision with clear histologic margins
Once the final excision is completed and the surgical margins are histopathologically clear, wound reconstruction can be performed. In this case, the surgical defect measured 7.2 × 5 cm. After discussion of reconstruction options, the patient opted to allow the defect to heal by second intention after wound reduction with a purse-string suturing technique (Fig. 16.8).
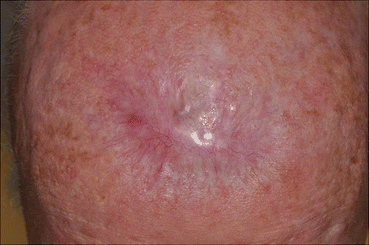

Fig. 16.8
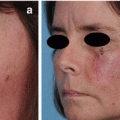
The final defect was reduced with purse string suturing technique and allowed to heal by second intention
Discussion
For LM/LMM, surgical excision is the treatment of choice and is associated with the highest cure rates, primarily because it allows for complete histological evaluation of margins and removal of periadnexal melanocytes and invasive components of lesions [1–3]. While the proper surgical margin can vary widely, excision with adequate margins results in good cure rates [4–7]. Multiple factors make determining the true margin of these lesions for pre-surgical planning difficult. LM/LMM (especially on head and neck) often occur in the background of photodamaged skin, as demonstrated in this case, with solar lentigines, seborrheic keratoses, actinic keratoses, and other chronic-UV light changes obscuring true LM/LMM margins [8]. They often have unpredictable subclinical extension of atypical junctional melanocytic hyperplasia beyond the visible pigmented margins. Since these lesions most often occur on the head and neck, maximal preservation of cosmesis and functionality are additional priorities for the surgeon during pre-operative planning.
As visual assessment of tumor borders is often inaccurate, pre-operative assessment to estimate true margins can be aided by the following techniques: Wood’s lamp examination, dermoscopy, confocal microscopy, and scouting biopsies [9]. Wood’s lamp light (320–400 nm) takes advantage of melanin’s absorption of radiation in the UV range. Lesions that possess increased melanin will appear brighter in contrast to normal skin when exposed to Wood’s lamp light [10]. As a result, the instrument provides increased contrast between normal and pigmented skin, allowing for improved delineation of LM/LMM clinical borders.
Dermoscopy is a non-invasive technique to examine pigmented anatomic structures of the epidermis, dermoepidermal junction, and superficial papillary dermis not visible to the naked eye [11]. For experienced users, dermoscopy has been shown to be more accurate than clinical examination for diagnosis of melanoma [12]. Specific features in helping to identify LM/LMM on dermoscopy include asymmetric pigmented follicular openings, pigmented lines forming rhomboidal structures, slate-gray globules, circles within circles, atypical blood vessels, and slate-gray dots [13]. In a study comparing LM/LMM clinical borders identified by routine clinical examination, Wood’s lamp, and dermoscopy prior to Mohs surgery excision, the borders determined by both dermoscopy and Wood’s lamp were larger than those by routine examination [14]. The borders determined by dermoscopy were also larger than those of Wood’s lamp. Still, most lesions had to undergo an additional 5 mm excision beyond the clinical border even defined by dermoscopy. These findings are further evidence of both the improved accuracy of Wood’s lamp and dermoscopy, as well as of the common subclinical extension of LM/LMM.
Reflectance confocal microscopy (RCM) allows for non-invasive, video-rate imaging of thin sections of human skin in vivo [15]. With a penetration depth of approximately 250 μm (superficial reticular dermis), an axial resolution of 3–5 μm, and a lateral resolution of approximately 1 μm, RCM images are comparable in resolution to routine histology sections [16]. Natural cutaneous chromophores and their varying refractive indices provide the contrast of RCM imaging; melanin, in particular, serves as a strong natural contrast agent [17]. Morphologic features on RCM positively predictive of malignant melanoma include disarray of epidermal honeycomb pattern, pagetoid cells in epidermis, non-edged dermal papillae, cellular atypia at the dermal-epidermal junction, atypical melanocytic nests, and bright nucleated cells in upper dermis [18, 19] In a study involving 51 patients with dysplastic nevus syndrome, RCM served as a helpful adjunct to serial imaging and was shown to have a sensitivity and specificity for the diagnosis of malignant melanoma of 100 % and 69 %, respectively [19]. Such diagnostic accuracy, in addition to its non-invasive real time imaging, allows RCM to play an important adjunct role in helping to define LM/LMM borders.
Histopathology examination with or without immunostaining remains the gold standard for defining benign versus malignant melanocytic lesions. In background skin with severe sun-damage, the specificity in diagnosing melanoma may be reduced when using dermoscopy and confocal microscopy. In such circumstances, scouting punch biopsies are helpful in identifying subclinical extension of LM/LMM lesions. In patients that were found to have melanoma in situ at margins after standard excision, Dengel et al presented the technique of placing 2-mm punch biopsy sites (which heal rapidly and scar minimally) in a ring 1 cm beyond the scar and/or residual melanoma lesion [20]. The use of ultraviolet light (Wood’s lamp) to guide scout punch biopsy sites has also been reported. UV light-assisted scout biopsy margin mapping is shown to avoid repeat surgery and may reduce recurrence [21].
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree