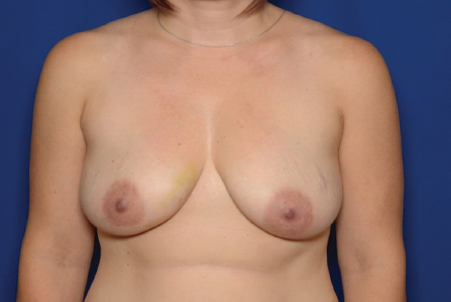
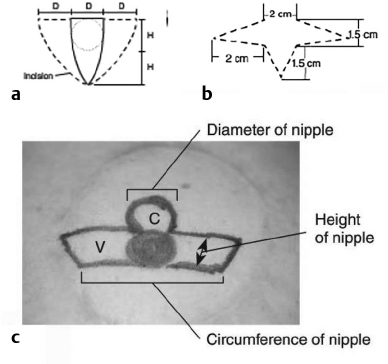
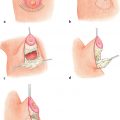
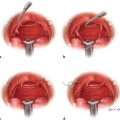
12 Breast Reconstruction Abstract This chapter will review surgical options for breast reconstruction including expander-based and autologous reconstructions, and the indications and limitations of each, as well as the role of radiation. The reader will be able to manage the mastectomy site and the irradiated bed for purposes of reconstruction, and will be able to create stepwise surgical reconstructive plans and manage complications. Keywords: breast reconstruction, tissue expansion, nipple reconstruction Six Key Points • Reconstruction can be either implant based or autologous. • Implant reconstruction in irradiated beds is associated with higher complication rates. • Cancer can be upstaged on final pathology, which must be considered when performing an immediate reconstruction. • Tissue expanders can be overfilled. • Food and Drug Administration (FDA) recommends MRI screening for silent ruptures 3 years after placement of silicone implants, and every 2 years after that. • Latissimus flaps can be more reliable in patients with a BMI > 35. A 45-year-old female presents for unilateral breast reconstruction, without radiation (Fig. 12.1). 1. The patient presents prior to mastectomy to discuss reconstructive options. What do you tell her? First, confirm the oncologic plan and whether postoperative radiation is needed. A targeted history should be performed looking specifically for intrinsic (such as bleeding or thrombotic disorders) and extrinsic factors (such as smoking) as they may affect the reconstructive options. Her goals for reconstruction should be identified, including whether she would prefer implant-based reconstruction or autologous reconstruction. 2. Would you offer her immediate reconstruction? “Immediate” reconstruction can refer to either a reconstructive procedure at the time of mastectomy or a definitive reconstruction at the time of mastectomy. There are two reasons to stage reconstruction with a tissue expander first, then definitive reconstruction. This first is to confirm the oncologic status. The second reason is to confirm that the mastectomy flaps are viable, and to allow for debridement of the mastectomy flaps if they are not. RATIONALE: Oncologic status must be confirmed, because the diagnosis can change on final pathology. Core biopsy can be inaccurate—approximately 18% of specimens with a core needle biopsy diagnosis of atypical ductal hyperplasia will be upstaged to either ductal carcinoma in situ (DCIS) or invasive cancer, and approximately 20% of DCIS specimens will be upstaged to invasive cancer, depending on associated clinical and histological characteristics.1–5 A change in final pathology may result in the need for further surgical intervention. 3. How do you determine what expander to use? The expander size is determined by the base width. This can be determined by the width of the breast, but with a large breast, the width can extend laterally on the chest wall and the expander itself can have the lateral aspect extending to the axilla. Once a subpectoral pocket is created, the width of the pocket can be measured with a ruler, and the implant can be sized. 4. How do you cover the implant? The implant can be either entirely submuscular or partially submuscular. In an entirely submuscular implant, the pectoralis muscle fibers remain attached to the rib caudally, and serratus muscle and fascia is elevated laterally and sewn to the lateral edge of pectoralis major muscle. In a partially submuscular implant, the pectoralis muscle fibers are detached from their rib attachments, and an acellular dermal matrix sling is placed. 5. How do you decide whether to use a complete submuscular placement or a subpectoral pocket? A complete submuscular placement provides two distinct advantages: it reduces the risk of infection and the risk of lateral displacement of the implant. The limitations of the approach are that the expander may be positioned more cephalad than with a subpectoral pocket. A subpectoral pocket is created when the caudal attachments of the pectoralis major muscle to the ribs are released. The expander has muscular coverage superiorly and medially. Use of a “sling,” or suturing acellular dermal matrix to the edge of pectoralis muscle, allows some coverage of the implant inferiorly and prevents caudal displacement of the implant. The advantage of this approach is that the implant can be positioned caudally near the intermaxillary fixation (IMF) and has less of a tendency for cephalad displacement, and allows more significant lower pole expansion, but there is a higher risk of expander exposure and seroma. 6. What are the risks of decellularized dermal matrix? There is a higher risk of seroma with AllodermTM (Life-Cell, Branchburg, NJ)6 and there is the risk of “red breast syndrome”.7 Red breast syndrome is a nonpainful erythema of the skin overlying acellular dermal matrix, not associated with warmth of the skin. Because it is acellular, there is minimal risk of rejection, and it allows from vascular ingrowth.8 7. Do you overfill the expander? Expanders are typically overfilled by approximately 20%. After the overexpansion, the pocket is allowed to mature for at least 1 month, although some wait longer. 8. She is expanded and you place an implant. How do you decide whether to use a saline or silicone implant? The risks and benefits of saline versus silicone implants are reviewed with the patient. Silicone implants have “silent ruptures” in which the shell ruptures are undetected on physical examination, and unnoticed by the patient, as opposed to saline, which gets resorbed by the body. Food and Drug Administration (FDA) recommendations are for silicone implants to be monitored by MRI for silent rupture 3 years after placement, and every 2 years after that. RATIONALE: In the overwhelming majority of cases, the decision to use a silicone or a saline implant is made by the patient, and the plastic surgeon’s role is to provide information about the risks and benefits of saline or silicone implants. It is important to note for young patients with breast cancer that while silicone implants are FDA approved for reconstructive purposes in any age, they are approved for women age 22 years and older for cosmetic purposes. 9. After reconstruction, she desires nipple areolar complex reconstruction. What are the options? There are over 15 described nipple reconstruction methods. Of these, the most popular are the skate flaps, the star flap, and the C-V flap. 10. Draw it. The drawings can be seen in Fig. 12.2.
Questions
Case 1
Plastic Surgery Key
Fastest Plastic Surgery & Dermatology Insight Engine