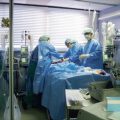
Fig. 44.1
Intestines are covered with a sterile plastic bag
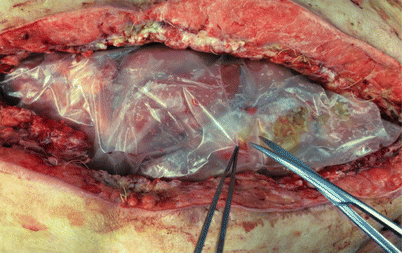
Fig. 44.2
Cuts are made on the plastic bag to allow better drainage of fluid
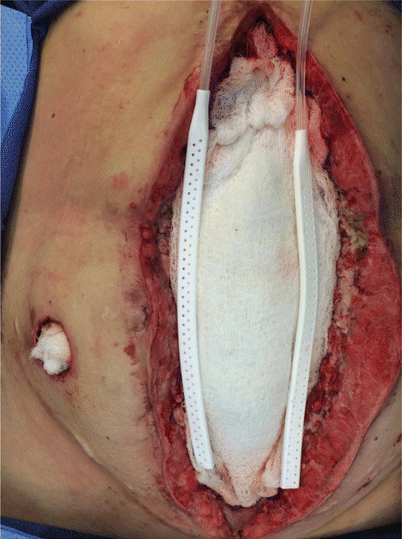
Fig. 44.3
A moist Kerlix gauze is placed over the plastic bag, and two drains are placed between gauzes
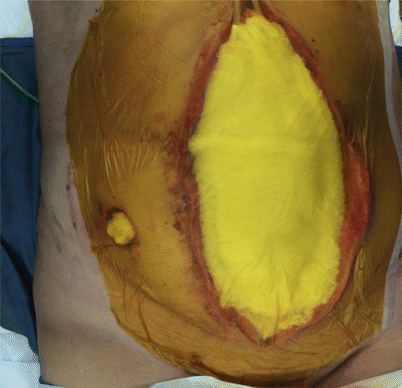
Fig. 44.4
Finally the gauze and the drains are covered with sticky plastic
Once the patient is resuscitated, he or she should be taken back to the operating room for definitive treatment and closure. Continue to attempt to perform a definitive closure at the first take back, the second take back, or even on the third or fourth take back. Sequential closure of the fascia should be attempted as well when unable to close at once. Starting at the most inferior and superior portion of the midline will make the closing process less difficult. If nothing else, you will reduce the defect and make it easier to eventually close it completely. On occasion, the intestines are so swollen, or there is continuation of intra-abdominal pathology that you are unable to close the fascia at all. In such cases you can use a temporary Vicryl mesh over the omentum or often directly over the intestines. If you have an ability to close the skin and subcutaneous tissue over the fascial defect, without major tension, this will be the preferred method, knowing that there will be a major hernia that you will deal with at a later time. Once you have committed to open abdomen management, it is very reasonable to use the wound VAC. Depending on the infectious status of the wound, the VAC can be changed every 2–3 days. In these situations, I prefer to use an irrigation. In many patients you may need to eventually cover the defect with skin graft.
On occasions you may be able to close fascia primarily by performing adjunct procedures such as lateral compartment release. This is a potentially risky procedure at this stage, as it may be complicated with skin and subcutaneous necrosis and you “burn the bridge” for future reconstructions. For this reason, I rarely perform lateral compartment release in the early stages of the management of the open abdomen. While there is always an option of using biologic mesh at this stage as a bridge, this should be your last resort of action.
44.2 General Principles of Management of Post-DCS Consequences
Post-DCS consequences can be challenging but should be understood by each surgeon who embarks on it. As described in the previous section, the process of closing the abdomen should start when the DCS is performed. There are a number of questions that need to be answered in this process, but the main ones are as follows: how to redefine the anatomy and new “physiology,” when should we perform the definitive surgery, how to plan and execute the operation and the intraoperative decisions, how to predict/prevent and how to deal with postoperative complications, how to ensure full recovery of the patient to normal functional status, and finally how long we need to follow up the patients?
While it is clear that there are many ways to manage these patients, the basic principles are the same, but the specific approaches to these complex patients depend on the concurrent presence of enterocutaneous fistulas (ECF) and/or enteroatmospheric fistulas (EAF), obesity, stomas, malnutrition, infection, and sepsis, as well as the overall physiology of the patient.
All these factors will dictate your surgical approach. However, whatever surgical approach you use and whenever you attempt to close the abdomen, the management of these patients should be done in a stepwise fashion. Each phase should be well planned and understood by the surgeon and surgical team, including nursing and anesthesia team. Oftentimes these patients are operated on in the same hospital, so nursing and other services know these patients well; thus it is important to keep them in the loop.
While there is always room for flexibility on surgical approach for individual surgeon, disciplined protocols and a well-planned surgical strategy, particularly in patients with large abdominal wall defects complicated by fistulas or stomas, make the operative management process easier and may improve postoperative outcomes. Such a strategy has been described in a six-step strategy for management of enterocutaneous fistulas, known as “SOWATS” (S = sepsis control, O = nutrition optimization, W = wound care, T = timing, A = anatomy, and S = surgery) 2,3. We have expanded this approach to a nine-step strategy and call it “ISOWATS PL” where I = identification and diagnosis of the postoperative fistula, S = sepsis and source control, O = optimization of nutrition, W = providing and ensuring wound care, A = redefining the anatomy and understanding the pathology at hand, T = timing of definitive surgery and/or takedown of fistulas, S = definitive surgery and surgical creativity, P = postoperative care, and L = long-term follow-up. Adhering to all nine steps of the “ISOWATS PL” maybe difficult at times as certain patients often require emergency surgery and you do not have the luxury to plan the entire process, but all attempts should be made as the process of re-operations is planned, structured, and executed carefully.
44.3 Redefining the Anatomy and New Physiology
Most patients undergo some form of radiologic study, CT scan being most predominant. In our recent study of 176 patients, the most common preoperative investigation performed was a computed tomography (CT) scan, followed by an MRI (Unpublished study performed at the university of Arizona by the author). Although nearly 15 % of patient population in our above mentioned study, presented with coexisting fistula, fistulogram was rarely performed. Barium studies and/or upper GI with small bowel follow-through has been mostly substituted with CT scan, although barium study has its relevance particularly in colonic fistulas, stomas, or when there is suspicion for other pathology of the colon.
44.4 Timing to Definitive Repair
We have previously described that the decision if and when to re-operate on patients with complex abdominal wall defects should be individualized and represents one of the most important steps in the surgical management of these patients. We base this decision on many factors but particularly on the comorbid diseases and on the anatomy of the surgical problem. In addition to considering the clinical status and physiology of the patient, one has to remember that these large defects can be functionally devastating and lead to further weight gain and more problems and potentially may lead to major morbidity. Patients with serious comorbid diseases such as extreme obesity, severe heart disease, high-grade liver cirrhosis, or lung disease (dependent upon oxygen therapy at home), unless they have symptoms of gastrointestinal obstructions, should be carefully evaluated before the decision of whether to operate is made.
I believe that at times, the strategy for these patients should be “more is better,” and often the definitive surgery is the only choice in the management. The definitive surgery should be performed earlier rather than later, assuming that the patient is not prohibitively at high risk for major complications.
While timing when to repair large abdominal wall hernias is less debatable, timing of taking down fistulas is more contentious. Delaying surgery anywhere from 12 to 36 months to improve the outcomes in patients with ECF has been suggested, although prolonging surgery for longer than 1 year following ECF diagnosis doubles the risk of postoperative refistulization. Waiting longer than 36 weeks increases the reported risk for fistula recurrence to 36 %, compared to 12 % if the operation is performed prior to 36 weeks. There are data or clinical predictive models to guide such decision, and thus the individual patient’s condition is the main factor that should be used as a guide.
In our experience, the optimal time for abdominal wall reconstruction is 6–12 months after the first procedure (when adhesions are less prominent), but this is at best an estimation. The presence of the so-called pinch sign (i.e., easy retraction of the skin or skin graft over the defect) is a good indicator that the adhesions are subsiding and that it is appropriate to schedule the abdominal reconstruction.
44.5 Operative Approach
Decision to operate is made jointly by the patient and the surgeon; definitive reconstruction technique is the next challenge to be faced. Most patients who have previously undergone large abdominal surgeries have a midline abdominal incision, so their lateral abdominal wall is usually free of scars and defects, thereby providing a well-vascularized soft tissue donor site, unless the patient has had lateral incision(s) or stoma(s). Unless the patient has a giant hernia with loss of abdominal domain, the abdominal wall can be anatomically restored with minimal tension and without compromising the integrity of the abdominal muscles, vessels, and nerves. The goals of the operation are to establish gastrointestinal (GI) tract continuity; obtain full closure of the abdominal wall; avoid the postoperative abdominal compartment syndrome; minimize recurrence of fistulas, hernias, and wound infections; and strive to restore the patient’s functionality. In patients with frozen abdomen or when a split-thickness skin graft (STSG) exists, dealing with adhesions, resecting fistulas, and performing the anastomosis require experience, and even entering the abdomen may prove challenging.
44.6 Definitive Abdominal Wall Reconstruction
Creating a new abdominal wall may represent a serious surgical challenge, and both the surgeon and the patient should be prepared for a lengthy procedure (i.e., entering the abdomen, taking down the adhesions, resecting the fistulas, and performing the anastomosis). Some authors have suggested that reconstruction should be performed by another team, such as plastic surgeons. On occasion, I have used the principle of “damage control on demand” by abbreviating the operation and returning the next day or so to completely inspect the previous work such as anastomosis again and ensuring that there are no missed enterotomies before performing the final closure.
44.6.1 Use of Native Tissue
You should strive to use native tissue to repair major defects if this does not create undue tension. If that is not possible, you should use a synthetic or biologic prosthesis. In most patients, some sort of combination of reconstruction techniques will be needed, that is, reducing the defect by transposing native tissue toward midline and the reinforcing with prosthesis. If the midline tissue cannot be easily approximated or if mesh reinforcement is needed (as it is in almost all abdominal wall defects larger than 6 cm), then other techniques must be considered. For example, if midline tissue cannot be easily approximated, then bilateral lateral component release should be done and a neo-abdominal wall reestablished. Tissue transposition of myocutaneous flaps through lateral component separation is the procedure of choice in my practice.
44.6.2 Other Adjunct Procedures
Other methods can be used to reconstruct the complex abdominal wall defects such as local advancement or regional flaps, distant flaps, or combined flap and mesh; however, which technique is used will depend on the pathology at hand and your expertise. In Type I defects with stable skin coverage, bridging the fascial gap with prosthetic material or autologous tissue is sufficient, whereas in Type II defects with absent or unstable skin coverage, fascial repair alone is inadequate, and the repair must be done with tissue utilizing more complex reconstruction techniques (e.g., regional or distant flaps, either alone or in combination with mesh). Vascularized flaps provide healthy autologous tissue coverage and usually do not require any implantation of foreign material at the closure site. Small and midsize defects can be repaired with pedicle flaps within the arch of the rotation of the flap. In extensive upper midline abdominal wall and thoracoabdominal defects, a free flap that offers a completely autologous, single-stage reconstructive solution is the best option available.
44.6.3 The Component Separation Technique
Component separation results in medial advancement of intact rectus myofascial units bilaterally, enabling the closure of defects of up to 10 cm in the upper abdomen, 20 cm in the mid-abdomen, and 6–8 cm in the lower abdomen. The component separation technique is based on an enlargement of the abdominal wall surface by separating and advancing the muscular layers. Some form of component separation, alone or in combination with other adjunct procedures, has become common practice. During the component separation technique (CST) for abdominal wall reconstruction, you should dissect out and develop anterior abdominal skin flaps laterally from the chest wall to the anterior superior spine. After that you need to divide the aponeurosis of the external oblique muscle longitudinally 2 cm laterally to the lateral edge of the rectus sheath, which will allow the mobilized rectus myofascial component to be mobilized medially and facilitate the approximation of the midline with sutures (Figs. 44.5, 44.6, 44.7, and 44.8). Every effort should be made to preserve the skin perforators as you dissect and create the mucocutaneous flaps. This will greatly reduce skin and subcutaneous necrosis.
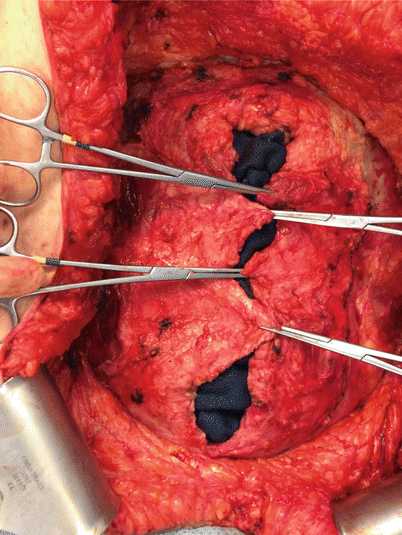
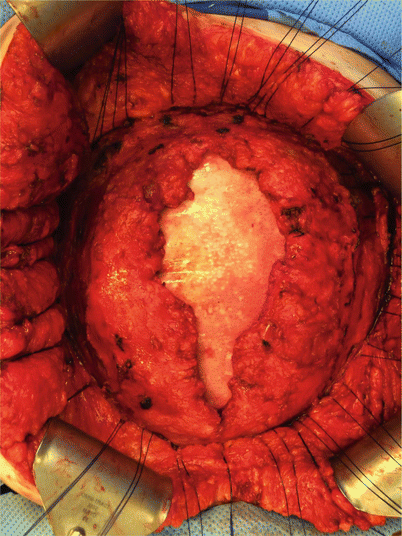
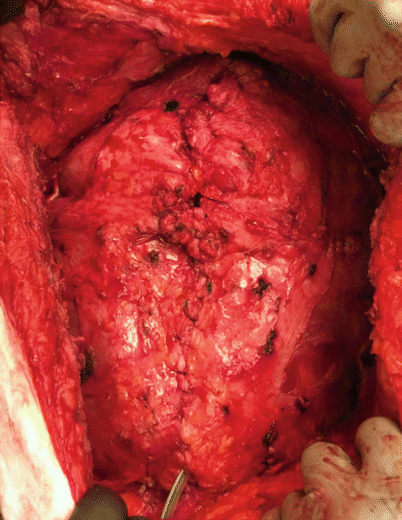
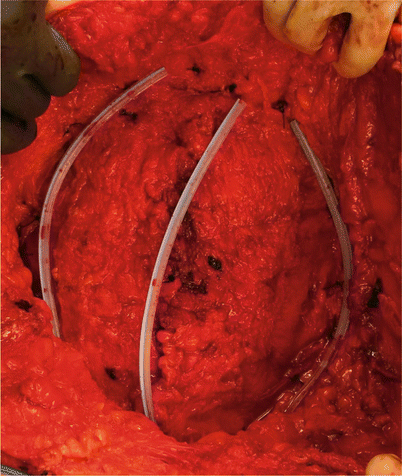

Fig. 44.5
Following anterior component separation, try to approximate the edges of the fascia

Fig. 44.6
Secured underlay mesh needs to be tight when all sutures are placed under some tension and pulled laterally

Fig. 44.7
Completed closure of the abdominal defect after underlay mesh placement and bilateral component release

Fig. 44.8




Drains are placed under the skin and subcutaneous tissue to reduce seromas
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree