○ Cells originate in the cuboidal basal layer and flatten out as they ascend to the surface – four to five layers/strata (deep to superficial): stratum basale, s. spinosum, s. granulosum, s. lucidum (only on palmoplantar surfaces), and s. corneum
○ Stratum basale: mitotically active cuboidal cells from which the upper layers of the epidermis are derived
♦ Attached to dermis by hemidesmosomes
♦ Keratins 5 and 14 produced here
♦ Cellular proliferation stimulated by various factors, including trauma and UV (↑ornithine decarboxylase expression is associated with (a/w) proliferative states)
➔ Ornithine decarboxylase is inhibited by corticosteroids, retinoids, and vitamin D3
♦ 10% of cells in the basal layer are stem cells, which give rise to other stem cells and transient amplifying cells that can still replicate, but only for a few cycles, until they reach a terminal differentiation phase, where they move upwards and eventually desquamate
♦ Transit time from basal layer to stratum corneum = 14 days; transit through the stratum corneum/desquamation = 14 days (total = 28 days from basal layer to desquamation)
○ Stratum spinosum: named for the “spiny” appearance of intercellular desmosomal connections on microscopy
♦ Contain multiple types of intercellular junctions
♦ Terminal keratinocyte differentiation 2° to ↑intracellular calcium in suprabasal epidermis
♦ Odland bodies (lamellar granules) are produced by Golgi bodies in spinous layer
➔ Primarily contain ceramide (most important lipid involved in epidermal barrier function; the most prevalent/important lipid in the stratum spinosum), along with glycoproteins, glycolipids, and phospholipids
➔ Are specialized lysosomes that exert most of their action in the stratum corneum, by discharging ceramides and other lipids to the extracellular space of the junction between the stratum granulosum and stratum corneum ➔ ceramides help form the cornified cell envelope (see below), and eventually replace the cell membrane
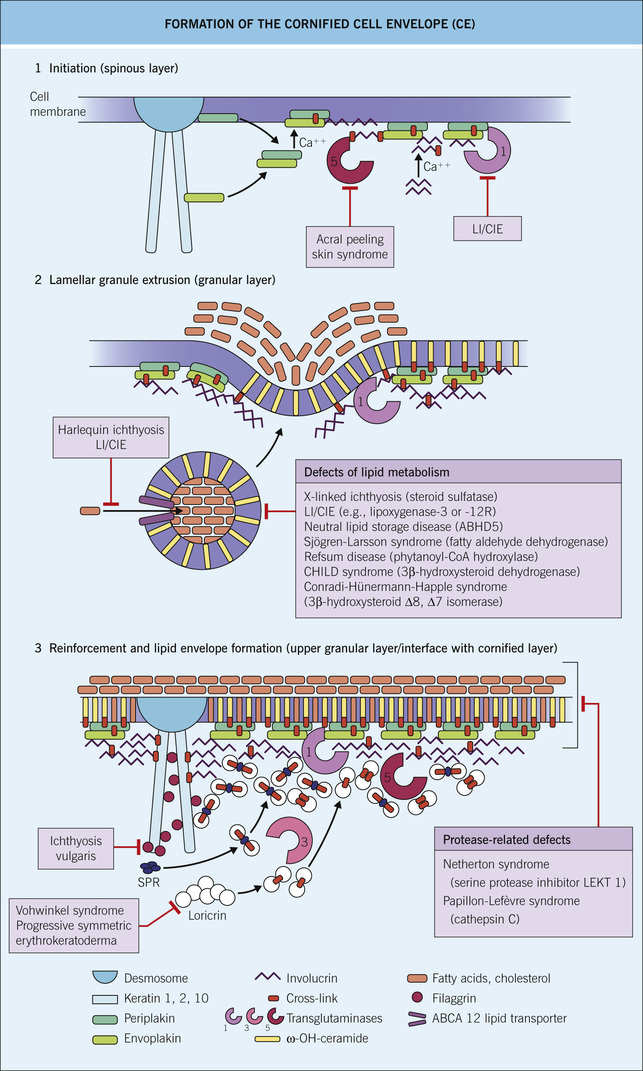
➔ Flegel’s disease and Harlequin ichthyosis are 2° to ↓lamellar granules
➔ X-linked ichthyosis occurs due to absent steroid sulfatase in lamellar granules
○ Stratum granulosum: flattened cells with prominent basophilic keratohyaline granules, which contain profilaggrin (converted to filaggrin at junction of stratum granulosum and stratum corneum), loricrin, keratin intermediate filaments, and involucrin
♦ Cells begin to lose nuclei, but keep overall structure
♦ Cornified cell envelope production primarily takes place in the granular layer (Fig. 1-2)
➔ Cross-linked protein and lipid structure encased in extracellular lipids forming a strong polymer that eventually replaces the plasma membrane
⟐ Process starts with envoplakin, periplakin, and involucrin scaffolding along the inner cell membrane (which is eventually replaced by ceramides from lamellar granules)
⟐ Further reinforcement by cross-linking loricrin (#1 component of cornified envelope, first appears in granular layer; mutated in Vohwinkel syndrome variant lacking deafness), small proline-rich proteins, keratin, and filaggrin
⟐ Cross-linking occurs via transglutaminase I ➔ γ-glutamyl lysine isopeptide bonds (Boards factoids: TG-1 is mutated in lamellar ichthyosis; TG-3 is antigenic target in dermatitis herpetiformis)
⟐ Other components include envoplakin (helps connect desmosomes to cornified envelope), periplakin, elafin, and others
⟐ Outer surface of the cornified envelope is ultimately surrounded by lipids (primarily ceramide) = cornified lipid envelope
⟐ Ultimately provides strong water-impermeable outer barrier
○ Stratum corneum: outermost layer, which serves as a mechanical barrier between the epidermis and the environment
♦ Composed primarily of protein-rich corneocytes (“bricks”; contain NO nuclei; keratin filaments attached to cornified envelope) embedded in a lipid matrix (“mortar,” cornified lipid envelope)
♦ Serves as a barrier to water loss (conditions that perturb the skin ➔ ↑transepidermal water loss) and toxins/infectious agents
○ Epidermal cells of importance
♦ Keratinocytes are the primary cells of the epidermis and produce proteins (e.g., keratin filaments) and lipids important for barrier function
➔ Keratins: intermediate filaments that comprise the primary cytoskeleton of the epidermis (see Table 1-3)
⟐ Type I keratins: low-MW; acidic; K9-28, K31-40 (hair keratins); chromosome 17
⟐ Type II keratins: high-MW; basic; K1-8, K81-86 (hair keratins); chromosome 12
⟐ Basic structure is an α-helical rod domain (consisting of heptad amino acid repeats) divided into four segments (1A, 1B, 2A, and 2B) that are interrupted by three nonhelical segments (“linkers”)
⟐ Functional unit consists of heterodimers of type I and type II filaments that form tetramers and ultimately filaments
⟐ Anchored to plasma membrane by desmosomes
➔ Keratinocytes produce IL-1, IL-6, IL-8, IL-10, IL-12, and TNF-α, among others
➔ Keratinocytes respond to IL-2, IL-4, IL-13, IL-22, and TNF-α, among others
➔ Neural crest-derived melanin-producing dendritic cells found in the stratum basale (≈1 : 10 ratio with keratinocytes, when viewed in 2-dimensional plane)
⟐ c-kit activation is needed for melanocyte development/migration; piebaldism occurs as a result of c-kit loss ➔ impaired melanocyte migration and proliferation; c-kit mutations are a/w mucosal and acral melanoma
➔ Each melanocyte interfaces with 36 keratinocytes when analyzed three-dimensionally (epidermal melanin unit)
➔ Melanin is produced in melanosomes (lysosome-type organelles) from its precursor, tyrosine, through a multistep enzymatic process involving tyrosinase (copper-dependent enzyme)
⟐ Tyrosine ➔ (tyrosinase-dependent step) DOPA ➔ (tyrosinase-dependent step) DOPAquinone ➔ pheomelanin (yellow/red; made by round melanosomes) or eumelanin (black/brown; made by elliptical melanosomes)
⟐ Melanosomes are transported along dendritic processes and transferred to keratinocytes through phagocytosis of dendrite tips
⟐ Racial variation in pigmentation: identical melanocyte density in dark and light skinned individuals; melanosomes in darker skinned individuals are larger, darker (↑melanin), more stable, and are transferred individually (vs smaller, lighter, less stable, and clustered melanosomes in lighter skin)
⟐ Melanin production is stimulated by melanocyte-stimulating hormone (MSH) and ACTH activity on MC1-R on melanocytes; also stimulated through various pathways induced by UV radiation
⟐ MC1-R loss of function mutations ➔ ↑pheomelanin:eumelanin ratio (phenotype = red hair/fair skin, ↑risk of melanoma)
⟐ Melanin absorbs UV ➔ protects against UV-induced mutations
⟐ UV exposure ➔ immediate tanning (from oxidation of existing melanin) and delayed tanning (requires new melanin synthesis)
➔ Other high-yield examination facts:
⟐ Defects in enzymes required to convert tyrosine to melanin ➔ oculocutaneous albinism; OCA1 (Tyrosinase), OCA2 (P gene), OCA3 (TRP-1)
⟐ Defects in packaging of melanosome-specific proteins ➔ Hermansky-Pudlak syndrome (HPS1 > HPS3 > other gene mutations)
⟐ Defects in lysosome and melanosome trafficking to dendrites ➔ Griscelli (MYO5A, RAB27A, and MLPH mutations) and Chédiak-Higashi syndrome (LYST mutations)
♦ Langerhans cells (LCs): major antigen presenting cells (APC) of the skin
➔ Dendritic histiocytes characterized by reniform (kidney shaped) nuclei, and tennis racket-shaped Birbeck granules seen on electron microscopy
➔ Interact with keratinocytes via E-cadherin
➔ Positive immunostains: CD207 (langerin; most sensitive IHC stain; specific for Birbeck granules), CD1a, S100, CD34, vimentin, and actin
➔ Originate from CD34+ progenitor cells in bone marrow like other monocytes/macrophages
➔ Found mainly in stratum spinosum, where it first encounters and processes antigens, and subsequently migrates to the lymph nodes to activate T-cells
➔ Downregulated in skin after UV exposure ➔ ↓immune surveillance
➔ See p. 24 for further discussion of function
♦ Merkel cells: slow-adapting mechanoreceptors found in fingertips, lips, oral cavity, and hair follicle ORS
➔ Found in stratum basale; communicate with neurons
➔ CK20+ in perinuclear dot pattern sensitive/specific for Merkel cells; also (+) for neurofilaments, S100, synaptophysin, chromogranin A, vasoactive intestinal peptide, neuron-specific enolase, and calcitonin gene-related peptide
■ Basement membrane zone (BMZ) (Fig. 1-3 and see Table 1-2)
○ Semipermeable barrier between epidermis and dermis that also serves to adhere basal keratinocytes to the underlying dermis
○ Key steps within each location:
♦ Basal keratinocyte/hemidesmosome: intracellular keratin filaments (K5 and K14) attach to electron-dense hemidesmosomal plaques (plectin and BPAG1 [BP230]) on the basal plasma membrane ➔ hemidesmosomal plaque proteins bind to intracellular portions of the anchoring filaments (BPAG2 and α6β4 integrin)
♦ Lamina lucida: extracellular portion of anchoring filaments (BPAG2, α6β4 integrin, and laminin 332) extend from the hemidesmosome down to the lamina densa; the thin filaments result in an electron-lucent region; is the weakest portion of BMZ ➔ is zone of separation in salt-split skin and also in suction blisters
♦ Lamina densa: anchoring filaments attach to type IV collagen (#1 component) and other proteins (laminin 332, laminin 331, and nidogen) in the lamina densa ➔ results in attachment between basal keratinocyte and lamina densa
♦ Sublamina densa: loops of type VII collagen (anchoring fibrils) arise from the underside of lamina densa, extend down into the dermis, hooking around dermal type I and III collagen fibers, and then loop back up to reattach to lamina densa (or anchoring plaques in dermis) ➔ firmly anchors the lamina densa (and all aforementioned structures) to the papillary dermis
○ BMZ also functions as a permeability barrier: heparan sulfate proteoglycan (negatively charged) in lamina densa is a major contributor
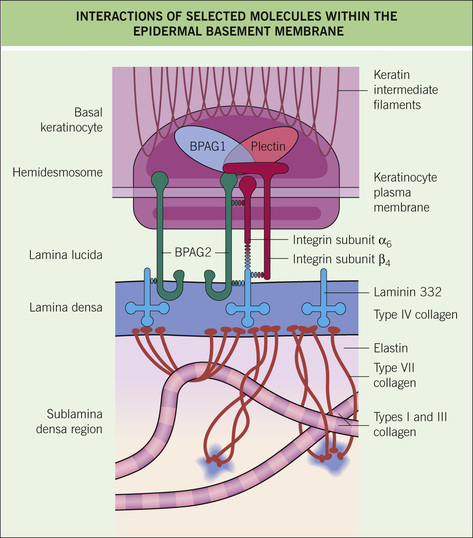
Table 1-2
Basement Membrane Zone Proteins
| Protein | Site | Source | Family | Function | Disease State |
BPAg1 (230 kD) | Hemidesmosome/keratinocyte | Keratinocyte | Plakin | Binds keratins and integrins; intracellular/part of attachment plaque | BP, EB simplex |
BPAg2 (180 kD) | Hemidesmosome/ keratinocyte ➔ lamina lucida Amino terminus is intracellular and carboxy terminus is extracellular –NC16A domain is closer to amino terminus but is extracellular | Keratinocyte | Collagen (XVII) | Transmembrane protein and one of the anchoring filaments; interacts with BPAg1, laminin 5, β4 integrin, and plectin | N16A Terminus: BP, pemphigoid gestationis, linear IgA bullous disease Carboxy Terminus: Cicatricial pemphigoid |
| α6β4 Integrin | Hemidesmosome/ keratinocyte ➔ lamina lucida | Keratinocyte | Integrin | Interacts with keratins, laminin 5, plectin, BPAg1, BPAg2; part of the anchoring filaments | Ocular cicatricial pemphigoid (antibodies to β4), EB with pyloric atresia (85%) |
| Laminin 332 (laminin 5, epiligrin) | Lamina lucida ➔ Lamina densa | Keratinocyte | Laminin | Connects other anchoring filaments (BPAg2 and α6β4 integrin) to collagen VII; part of the anchoring filaments | Antiepiligrin pemphigoid (a/w malignancy), JEB-Herlitz |
| Plectin | Hemidesmosome | Keratinocyte | Plakin | Binds keratins and integrins; intracellular/part of attachment plaque | EB with muscular dystrophy, EB with pyloric atresia (15%) |
| Nidogen (entactin) | Lamina densa | Unclear | Nidogen | Adaptor between laminin 1 and collagen IV in lamina densa; stabilizes proteins of lamina densa | |
| Collagen IV | Lamina densa | Unclear | Collagen | Anchors laminins in lamina densa ➔ structural support; also a component of anchoring plaques in dermis, which attach collagen VII to collagen I and III | Goodpasture disease, Alport syndrome |
| Collagen VII | Sublamina densa | Fibroblasts | Collagen | Major component of anchoring fibrils | Dystrophic EB, bullous lupus, EB acquisita |
| Heparan sulfate proteoglycan | Lamina densa | Fibroblasts | Proteoglycans | Contribute to matrix of and give an overall negative charge (creating a permeability barrier) to the basement membrane |
Table 1-3
Protein Components of the Epidermis (Including Nonepidermal Keratins)
| Protein | Site of Synthesis | Function | Disease State |
| Keratin 1 | Suprabasal keratinocytes (produced in spinous layer) | Primary keratinocyte cytoskeleton | Epidermolytic ichthyosis (preferred new name for EHK), epidermolytic and nonepidermolytic (Unna-Thost) PPK, ichthyosis hystrix of Curth-Macklin* |
| Keratin 2 | Granular layer | Superficial epidermolytic ichthyosis (Siemens) | |
| Keratin 3 | Cornea | Meesmann’s corneal dystrophy | |
| Keratin 4 | Mucosal epithelium | White sponge nevus | |
| Keratin 5 | Basal keratinocytes | EBS, Dowling-Degos disease* | |
| Keratin 6a | Outer root sheath of hair | Pachyonychia congenita I* | |
| Keratin 6b | Nail bed epithelium | Pachyonychia congenita II | |
| Keratin 9 | Palmoplantar suprabasal keratinocytes | Vorner (epidermolytic) PPK | |
| Keratin 10 | Suprabasal keratinocytes (produced in spinous layer) | Epidermolytic ichthyosis* | |
| Keratin 11 | Granular layer | ||
| Keratin 12 | Cornea | Meesmann’s corneal dystrophy | |
| Keratin 13 | Mucosal epithelium | White sponge nevus | |
| Keratin 14 | Basal keratinocytes | EBS, Naegeli-Franceschetti-Jadassohn syndrome, dermatopathia pigmentosa reticularis | |
| Keratin 16 | Outer root sheath of hair | Pachyonychia congenita I* | |
| Keratin 17 | Nail bed epithelium | Pachyonychia congenita II, steatocystoma multiplex | |
| Keratin 19 | Stem cells of basal layer | ||
| Keratin 71, 73, 74 | Hair inner root sheath | Wooly hair | |
| Keratin 32, 35, 82, 85 | Hair cuticle | ||
| Keratin 17, 33, 34, 36, 37, 75, 81 | Hair medulla | Pseudofolliculitis barbae | |
| Keratins 31–38, 81, 83, 85, 86 | Hair cortex | Monilethrix (KRT81, KRT83, KRT86 most commonly; also DSG4) | |
| Filaggrin/profilaggrin | Granular layer | Aggregates keratin, flattening granular layer cells. Degraded in the stratum corneum into urocanic acid and pyrrolidone carboxylic acid, which help block/absorb UV radiation. Urocanic acid is also a component of natural moisturization factor – helps keep stratum corneum hydrated/moist | Ichthyosis vulgaris, atopic dermatitis |
| Loricrin | Granular layer | Most abundant component of cornified cell envelope. Cross-linked to involucrin by transglutaminase 1**. | Vohwinkel syndrome with ichthyosis (NO deafness) Decreased in psoriasis |
| Involucrin | Granular layer | Component of cornified cell envelope. Proteins are cross-linked together by transglutaminase 1 ➔ strong border | Increased in psoriasis |
○ Located below the epidermis, derived from mesoderm, and divided into papillary dermis (superficial) and reticular dermis (deep)
♦ Fibroblasts-create extracellular matrix and are involved in wound healing
♦ Mononuclear phagocytes – discussed on p. 23
♦ Mast cells – discussed on p. 23
♦ Glomus cells – specialized smooth muscle cells derived from Sucquet-Hoyer canals, which allow for blood shunting from arterioles to venules (bypassing capillaries); found mainly in the palms/soles
➔ Overproduction ➔ glomus tumor (favors acral sites because of ↑glomus cell density)
♦ Dermal dendritic cells – bone marrow-derived APC that resides within dermis; highly phagocytic
♦ Provides structure and support to the dermis; essential for water retention and for signal transduction
♦ Synthesized by dermal fibroblasts
♦ Composed of collagens, elastin, fibrillins, fibulins, integrins, laminins, glycoproteins, and proteoglycans
➔ Collagens are triple helices formed by amino acid chains where every third residue is glycine (Gly-X-Y), with a high likelihood of proline and hydroxyproline/hydroxylysine in the X and Y positions, respectively
⟐ Accounts for 75% of dry weight of the skin; #1 component of the dermis
⟐ Collagen I is the primary collagen (85%) of the ECM; type III (10%; important and prevalent in blood vessels, fetal skin, GI tract, new scars, and keloids) and V are also present
⟐ Lysyl hydroxylase and proline hydroxylase catalyze crosslinking of collagen; vitamin C-dependent process (deficiency ➔ scurvy)
⟐ Defects in collagen and/or collagen cross-linking result in most forms of Ehlers-Danlos syndrome: COL1A1/2 (EDS arthrochalasia type, and osteogenesis imperfecta); COL3A1 (EDS vascular type); COL5A1/2 (classical EDS); Lysyl hydroxylase/PLOD1 gene (EDS kyphoscoliosis type)
⟐ Matrix metalloproteinases degrade collagen
⟐ Retinoids ➔ ↑collagen production
⟐ Corticosteroids and UV ➔ ↓collagen production
➔ Elastic fibers provide resilience from stretching and modulate TGF-β and BMP signaling
⟐ Account for 4% of dry skin weight
⟐ 90% elastin (core) and 10% fibrillin (surrounds elastin); elastin contains high levels of desmosine and isodesmosine ➔ these crosslink with fibrillin via lysyl oxidase (copper necessary for function)
⟐ Notable defects in elastic tissue: Fibrillin 1 mutation (Marfan’s syndrome); Fibrillin 2 mutation (Congenital contractural arachnodactyly); Fibulin 5 (Cutis laxa; gene defect results in decreased desmosine); LEMD3 mutation (Buschke-Ollendorf syndrome; defect results in increased desmosine); ABCC6 mutation (Pseudoxanthoma elasticum; mutation results in fragmentation and calcification of elastic fibers)
⟐ Elaunin fibers run horizontal/parallel in reticular dermis and oxytalan fibers run vertical/perpendicular to DEJ in papillary dermis; mnemonic: “stand (= vertical) up-high (= high in dermis) with OXYgen (= OXYtalan)”
⟐ UV radiation ➔ damage of elastic fibers
♦ All aforementioned fibers are set in a matrix of proteoglycans and glycosaminoglycans (GAGs) that retain large quantities of water (up to 1000× their volume!) = ground substance
➔ Most important GAGs = hyaluronic acid, chondroitin sulfate, dermatan sulfate, and heparan sulfate
➔ GAGs are broken down by lysosomal hydrolases
♦ Cutaneous vasculature important for metabolic support of the skin and maintenance of body temperature
♦ Two vascular plexuses: superficial (➔ vessels of the reticular dermis) and deep (➔ follicles, glands)
♦ VEGF is the primary mediator of vasculogenesis
➔ ↑VEGF: most cancers, psoriasis, POEMS syndrome, and other diseases with increased vasculature
♦ Lymphatic vessels collect fluid and proteins from interstitium and direct it into the lymph circulation
♦ Prox1, Podoplanin (D2-40), LYVE-1, and VEGFR-3 are lymphatic vessel markers
♦ Nerves of the skin are responsible for detecting touch/pressure, pain, itch, and other sensations
♦ Cutaneous sensory nerves are divided into free nerve endings and corpuscular nerve endings (round/globular collection of neural and other cells)
⟐ Itch and pain: A-δ (larger; myelinated) and C-polymodal nociceptor afferent fibers (smaller; unmyelinated)
⟐ End in the epidermis/superficial dermis
➔ Specialized nerve receptors (corpuscular nerve endings)
⟐ Krause end bulbs: genitalia, perianal region, and vermillion lips; mnemonic “Krazy Krause ends on erotic areas”
⟐ Meissner’s corpuscle: superficial (dermal papillae) mechanoreceptor of digits; fast adapting; suited for pressure/light touch
⟐ Pacinian corpuscle: deep (deep dermis/fat) mechanoreceptor of palmoplantar skin, nipples, and genital region; fast adapting; suited for vibration and deeper pressure
⟐ Merkel nerve ending: superficial (basal epidermis) mechanoreceptor most concentrated in fingertips, lips, and external genitalia; slow adapting; suited for pressure/touch
⟐ Ruffini corpuscle: deep (fat) mechanoreceptor most concentrated around fingernails; slow adapting; suited for sustained pressure
♦ Innervation of cutaneous appendages:
➔ Adrenergic control: vascular smooth muscle, apocrine glands, and arrector pili contraction
➔ Cholinergic control: eccrine glands
♦ Secretory exocrine gland primarily responsible for thermoregulation and waste excretion
♦ Found on all cutaneous surfaces except: external auditory canal, lips, glans penis, clitoris, and labia minora
♦ Highest concentration = palms and soles
♦ Controlled by hypothalamus; innervated by postganglionic sympathetic fibers, which synapse with muscarinic acetylcholine receptors on the glands
♦ Isotonic sweat secreted in secretory gland ➔ NaCl reabsorbed in duct ➔ hypotonic sweat is delivered to surface
➔ ↑rate of sweating ➔ more isotonic solution (less time to reabsorb in duct)
➔ Maximal rate of sweating ~ 3 L/hr
♦ Components (deep to superficial): secretory coil (deep dermis), intradermal/straight duct (eosinophilic cuticle seen on histology), and acrosyringium (intraepidermal portion; spiral duct that opens onto the skin surface)
♦ Stains for S100, keratin, and CEA
♦ Secretory exocrine glands with unclear function in humans, though in animals they mediate sexual attraction through pheromone release
➔ Activity begins around puberty
♦ Located primarily in anogenital skin, axillae, external ear canal, vermillion border, periumbilical region, eyelid margin, and breast
♦ Empty into follicular infundibulum (above sebaceous duct)
♦ Secretory control unclear ➔ glands noninnervated, but do have β-adrenergic receptors, which are likely stimulated by circulating catecholamines
♦ Secretory products released through decapitation secretion: cholesterol and cholesterol esters, triglycerides, squalene, and fatty acids
➔ Lipofuscin = pigmented mixture of lipids and proteins ➔ responsible for yellow-brown color of chromhidrosis
♦ Initially odorless secretions ➔ later modified by surface bacteria ➔ results in body odor
♦ Ectopic or modified apocrine glands: mammary glands, ceruminous glands of the external auditory canal, and Moll’s gland of the eyelids
➔ These empty directly to the surface
♦ Secretory exocrine glands found primarily on the scalp, face, and upper anterior trunk (“seborrheic areas”)
♦ Functions include water retention and innate immune defense
♦ Consist of sebocytes, which contain lipid vacuoles
♦ Normally associated with hair follicles and empty into inferior portion of the infundibulum
♦ Pubertal androgen production is major signal for sebaceous gland maturation (under adrenergic control)
➔ Transient maternal androgen stimulation present in infancy
♦ Other endocrine factors stimulating maturation and sebum production: MSH, CRH, and substance P
♦ Secretory products released through holocrine secretion (entire cell lyses to release contents):
➔ Triglycerides (#1 component; ≈50%) > wax esters (#2) > squalene (#3)
➔ Others: cholesterol esters, cholesterol, antimicrobial peptides, androgens, and cytokines
♦ Ectopic sebaceous glands: Meibomian glands on eyelid tarsal plate, Fordyce spots (vermillion lip/oral mucosa), Montgomery tubercles (areolae/nipples), Tysons glands (labia minora/prepuce), and Zeis glands (eyelid margin, close to Moll’s gland)
♦ Epithelial-derived appendage important for temperature regulation, protection of other structures (nasal mucosa, eyes, and ears), social and sexual cues, and tactile sensory input
➔ Lanugo – fine hairs shed late in gestation and during the first month of life
➔ Vellus – fine hairs over face, trunk, and extremities early in life
➔ Terminal – coarse, darker hairs of scalp, eyebrows, and eyelashes; postpubertal androgens induce switch to terminal hairs in other sites
♦ Hair density: ~100,000 hairs on scalp; more in blonde and fewer in red-haired individuals
♦ Anatomy: Table 1-4
Table 1-4
Hair Anatomy
| Portion of Hair | Description |
| Hair bulb | Lowermost portion of the hair follicle |
| Hair matrix | Rapidly proliferating keratinocytes that terminally differentiate to produce the hair shaft |
| Infundibulum | Region extending from the skin surface down to the point where the sebaceous gland opens into the hair follicle; ORS displays cornification similar to that of the interfollicular epidermis (i.e., contains keratohyaline granules) |
| Isthmus | Region located between the opening of the sebaceous gland, down to the site of insertion of the arrector pili muscle; ORS displays trichilemmal keratinization (no inner root sheath and IRS is shed before this point) |
| Lower hair follicle | Region located between hair bulb to proximal isthmus; encapsulates dermal papilla; has inner and outer root sheaths; critical line of Auber is the widest area |
| Arrector pili muscle | Inserts at the level of the bulge; pulls up hair (“goose bumps”) |
| Bulge | Segment of the outer root sheath located at the level of arrector pili muscle insertion; major seat of epithelial stem cells of the hair follicle |
| Secondary hair germ | Additional seat of epithelial and also of melanocyte stem cells; located between club hair and dermal papilla in telogen hair follicle |
| Connective tissue sheath (CTS) | Special mesenchymal follicular sheath that is tightly attached to the hair follicle basement membrane and is continuous with the follicular dermal papilla |
| Follicular dermal papilla (DP) | Onion-shaped, closely packed, specialized fibroblast population with inductive and morphogenic properties; hair cycle-dependent fibroblast trafficking occurs between CTS and DP; volume of DP determines size of hair bulb and, thus, hair shaft diameter |
| Inner root sheath (IRS) | Packages and guides the hair shaft; cornifies normally; stains red secondary to citrulline; not present in telogen hairs; is present in lower hair follicle but not in the isthmus/infundibulum |
| Outer root sheath (ORS) | Merges distally into the epidermis and proximally into the hair bulb; provides slippage plane, nutrition, regulatory molecules, and stem cells |
| Critical line of Auber | Widest section of the hair bulb and where most mitotic activity essential for hair growth occurs |
| Follicle pigmentary unit | Melanin-producing hair follicle melanocytes located up and around the upper one-third of the DP; transfer pheomelanosomes or eumelanosomes to differentiating hair follicle keratinocytes in the precortical matrix; goes largely into apoptosis during each catagen phase, regenerated from melanocyte stem cells in hair germ during anagen |
♦ Follicular layers (outer to inner): glassy membrane, outer root sheath, inner root sheath (Henle’s layer, Huxley’s layer, and cuticle), and hair shaft (cuticle, cortex [where most hair keratins located], and medulla)
➔ Cuticle helps keeps hair intact – damage ➔ split ends (trichoptilosis)
➔ Cuticle from hair shaft and inner root sheath merge
♦ Dermal papilla: mesenchymal structure (from embryonic mesoderm) containing vasculature; contributes to hair cycle regulation
♦ Hair cycle: Table 1-5
➔ Exogen: phase of active shedding of club hair between telogen and anagen
➔ Kenogen: subphase of telogen in which no shaft is present
Table 1-5
Hair Growth Cycle
| Anagen | Catagen | Telogen (Club Hairs) | |
| Phase activity | Growth (~0.4 mm/day or ~1 cm/month on scalp); follicular melanocytes only active in anagen phase) | Regression (melanocytes in matrix apoptose; inner root sheath lost) | Resting |
| Duration | 2–6 years | 2–3 weeks | 3 months |
| Percentage (%) of scalp hairs | 85–90 | 1–2 | 10–15 |
♦ Color of hair 2° to hair melanocytes in anagen bulb/matrix (melanin unit = 1 melanocyte: five keratinocytes; melanocytes only produce pigment in anagen phase!)
➔ Eumelanin (brown/black hair pigment) vs pheomelanin (red/blonde hair pigment)
♦ Hair follicle stem cells reside in bulge and contribute to hair cycling, tissue regeneration, and wound healing; follicular melanocytes can migrate to interfollicular areas in disease states (e.g., vitiligo) to assist with repigmentation
➔ As with interfollicular stem cells in the basal layer of skin (see p. 1), also produce transient amplifying cells with restricted mitotic capacity
♦ Disulfide bonding via cysteine residues determines curliness of hair – these bonds are broken when hair is straightened, but subsequently reform with time
♦ Appendageal structure important for protection and function of fingertips, for proper function of the feet, ability to scratch, and aesthetic appearance
♦ Plate is composed of keratin-producing onychocytes
♦ Important anatomic structures: Table 1-6
Table 1-6
Nail Anatomy
| Nail Location | Description |
| Proximal nail fold | Superficial layer continuous with skin, deep layer continuous with nail matrix |
| Eponychium (cuticle) | Located between nail plate and nail matrix; acts as seal against the environment |
| Nail matrix | At the proximal end of nail unit, generates the plate. Proximal matrix➔ superficial portion of nail plate; distal matrix ➔ ventral portion of plate. Melanocytes found in nail matrix |
| Lunula (distal nail matrix) | Junction between matrix and bed |
| Nail plate | Hard, functional unit of nail, composed primarily of keratins; strong attachment to nail bed |
| Nail bed | Extends from lunula to onychodermal band. Provides support for nail plate. Very minimal contribution to nail plate synthesis |
| Onychodermal band | Red/pink transverse band marking end of bed |
| Hyponychium | Continuous with ventral edge of free nail plate and distal fingertip skin |
| Lateral nail folds | Guide growth of nail plate |
➔ Fingernails: 2 to 3 mm/month (~6 months to grow out)
➔ Toenails: 1 mm/month (~12 months to grow out)
1.2 Embryology
• Skin structures derived from two of three primary germ layers
■ Ectoderm: epidermis, adnexal structures, Merkel cells, melanocytes (neural crest), and nerves (neuroectoderm)
■ Mesoderm: fibroblasts, LCs, vessels, and inflammatory cells
■ 5 weeks: outer periderm and inner basal epidermal layer
■ 8 weeks: epidermal stratification
■ 9–12 weeks: melanocytes, LCs, and Merkel cells migrate into the epidermis
■ Late second trimester: terminal differentiation, with full stratification
• Basement membrane, dermis, and subcutis
■ 6–8 weeks: fibroblasts appear beneath the epidermis
■ 9 weeks: distinct border between epidermis and dermis (DEJ present)
■ 9–12 weeks: primordial vasculature formed
■ 16–18 weeks: initial fat formation in subcutis
■ 20 weeks: mature thickness of dermis and dermal ridges present
■ 9–12 weeks: initial follicle development on the eyebrows, scalp, upper lip, and chin – spreads caudally and ventrally; epidermal placodes (derived from ectoderm) induce underlying dermal papilla formation (derived from mesoderm)
■ 18–20 weeks: hair canal fully formed
■ 24–28 weeks: initiation of cycling through anagen, catagen, and telogen
○ Sonic hedgehog is an important molecule for telogen to anagen transition
■ 8–10 weeks: nail bed demarcation
■ 12 weeks: proximal nail folds formed
■ 17 weeks: nail plate formed, covers nail bed by week 20
■ 10 weeks: eccrine gland anlage formation on palms and soles
■ 14–16 weeks: eccrine primordia bud down, glands begin to develop
■ 22 weeks: initiation of truncal eccrine gland formation; eccrine glands and ducts nearly mature on volar skin
■ Apocrine gland formation initiated later than eccrine glands, at 22 weeks
■ Sebaceous gland formation parallels hair follicle development (derived from outer root sheath)
■ Neural crest-derived cells, migrate under the direction of KIT and KIT ligand
■ 12 weeks: melanocytes present in epidermis
■ 12–16 weeks: melanin production begins
■ 16–20 weeks: melanocytes proliferate and become fully functional (transfer melanosomes to keratinocytes)
■ Epidermal stem cells responsible for maintenance, repair, and renewal of epidermis
■ Keratinocyte stem cells located within the bulge region of the hair follicle and at the base of rete ridges of interfollicular epidermis
■ Complete renewal of epidermis every 40 to 56 weeks
■ Stem cells are multipotent with unlimited capacity to divide
■ Asymmetric division gives rise to transient amplifying cells, which divide rapidly to produce terminally differentiated cells
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree